Clear Sky Science · en
Physiological basis of resolution acuity in vision
Why sharp eyesight matters
Being able to read fine print, recognize a face across the room, or see a distant road sign all depend on how finely our eyes and brain can resolve detail. For decades, scientists have known that the tiny cone cells at the center of the retina set a physical limit on how sharp vision could be, but it was unclear whether the brain’s early wiring actually used information cone by cone. This study pinpoints how signals from single cones are passed forward in the primate visual system, revealing how close our everyday vision comes to the physical limits set by the eye itself.
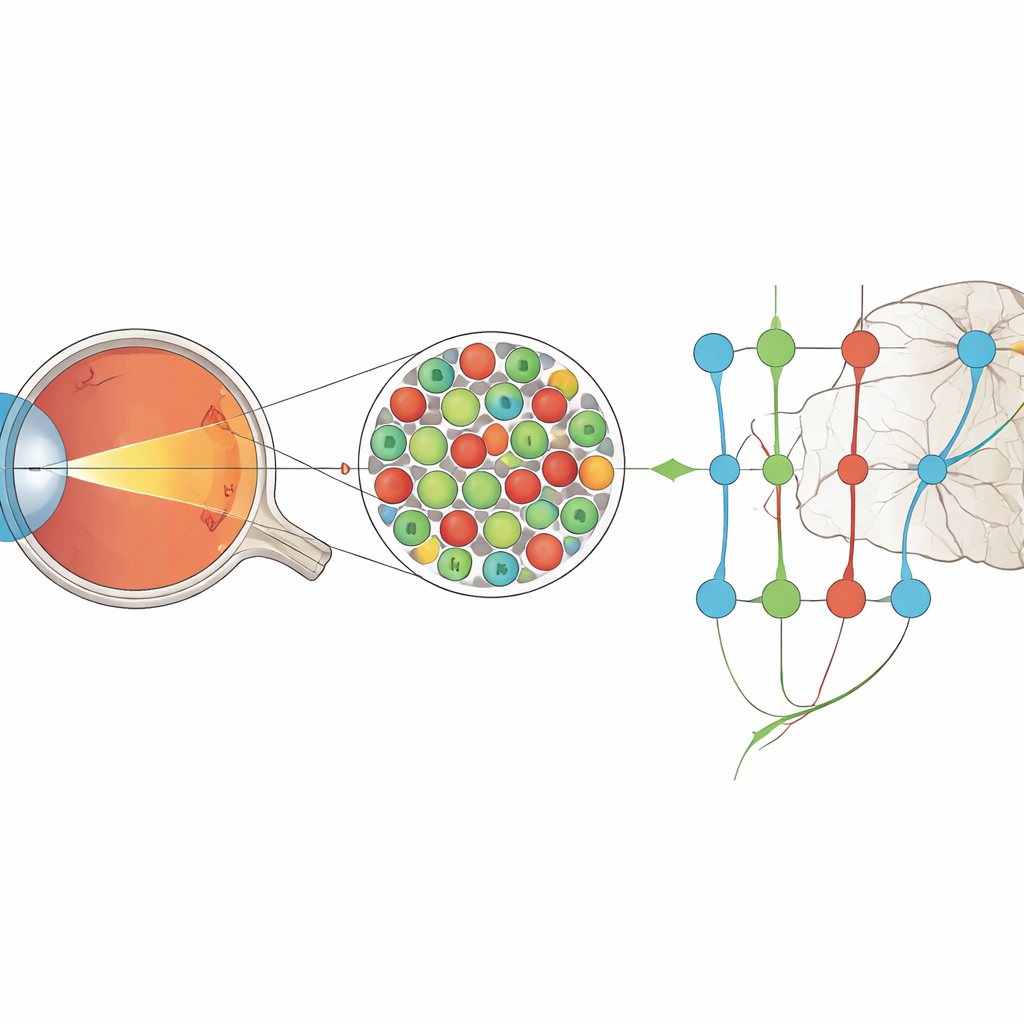
The eye’s finest grid of light sensors
In humans and other primates, the sharpest vision comes from the fovea, a small pit near the center of the retina packed with cone photoreceptors. These cones sit in a near-perfect grid, each one covering a tiny patch of the visual world. Anatomy suggested that, near the fovea, each cone could connect almost privately to its own dedicated output cell in the retina, and from there to the brain. Yet previous physiological measurements made it seem as if early visual neurons were sampling over several cones at once, implying that some of the fine detail was lost before signals ever reached the cortex.
Building a microscopic projector in the eye
To resolve this mismatch, the authors used a highly specialized instrument called an adaptive optics scanning laser ophthalmoscope. This system compensates for the eye’s optical imperfections in real time, allowing them to both image individual cones and project tiny, precisely controlled spots of colored light directly onto the cone mosaic. Working with anesthetized macaque monkeys, they recorded electrical activity from neurons in the lateral geniculate nucleus (LGN), a relay station that passes signals from the retina to the visual cortex. They presented rapidly flickering “noise” movies in red and green light whose pixels were smaller than a single cone, while simultaneously tracking exactly which cones were being illuminated.
Finding neurons driven by single cones
By averaging the visual patterns that preceded each neural spike, the team reconstructed the “receptive field” of each LGN neuron—the tiny region of the cone mosaic that most strongly drove it. They then overlaid these receptive fields onto high-resolution images of the cones. For most parvocellular LGN neurons, which are specialized for fine detail and color, the centre of the receptive field lined up with just one cone. As the researchers moved farther from the fovea, both cone size and receptive field size increased together, preserving this one-cone match. A smaller fraction of cells showed contributions from two or three neighboring cones, consistent with known electrical coupling and slight convergence in retinal circuits.
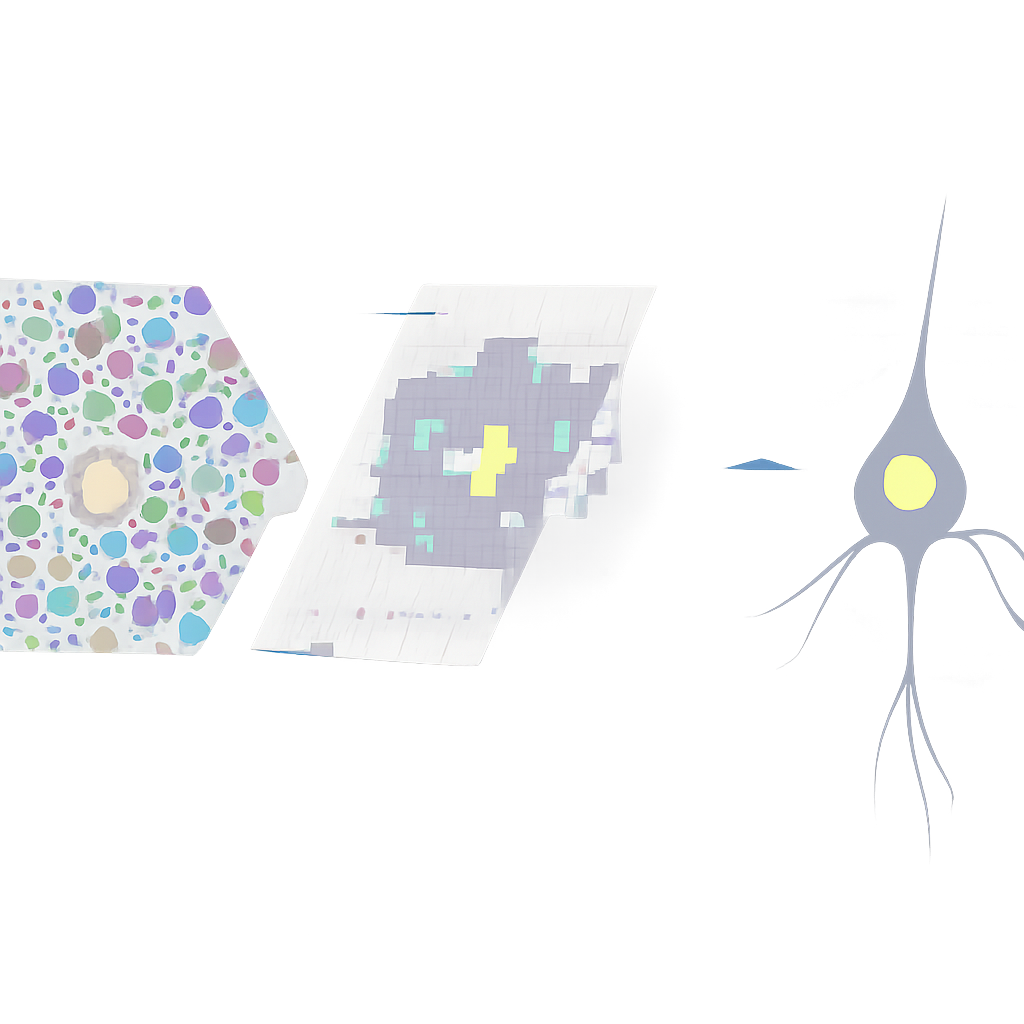
Pushing vision to the limits of the cone grid
The team went further, combining a detailed physical model of how light spreads and is absorbed in cone outer segments with their experimental data. Running large-scale simulations, they tested whether the shapes and sizes of the measured receptive fields were more consistent with input from one, two, or three cones. About three quarters of the mapped LGN neurons were best explained by a single-cone centre, even after accounting for optical blur, tiny eye movements, and measurement noise. When they probed some of these same neurons with high-contrast drifting gratings, the cells responded robustly to spatial frequencies beyond 20 cycles per degree—roughly four times higher than earlier estimates made without adaptive optics—matching what would be expected if the system were sampling at the spacing of individual cones.
What this means for everyday sight
These findings show that, near the centre of gaze, the early visual pathway transmits information at nearly the finest resolution physically available from the cone mosaic. In other words, signals reaching the cortex already carry cone-by-cone detail, and limits on ordinary visual acuity largely reflect cone spacing and optical blur, not early pooling of signals. This framework helps separate basic resolution from “hyperacuity” tasks, like judging tiny offsets between lines, which must rely on higher-level processing that goes beyond the physical sampling grid. The results also underscore the importance of good optical correction—through natural optics, glasses, or surgery—because when the image on the retina is sharp, the neural wiring is poised to use it at the ultimate limit set by the cones themselves.
Citation: Ramsey, K.M., Tellers, P., Meadway, A. et al. Physiological basis of resolution acuity in vision. Nat Commun 17, 2467 (2026). https://doi.org/10.1038/s41467-026-68851-0
Keywords: visual acuity, cone photoreceptors, lateral geniculate nucleus, fovea, adaptive optics