Clear Sky Science · en
Enhanced methane chlorination via RuO2-gas convection electrode with in-situ generated dynamical three-phase boundaries
Turning a Common Gas into Useful Goods
Methane is often talked about as a troublesome greenhouse gas, but it is also a rich raw material that could be turned into everyday products like rubber, paints, and medicines. Today, a key step in that chain is making chloromethane, a basic building block for many industrial chemicals. The problem is that the standard manufacturing route is hot, energy-hungry, and depends on relatively costly ingredients. This study explores a cooler, cleaner way to turn methane and salty water into chloromethane using electricity and a specially designed electrode, potentially helping industry cut both emissions and energy use.
Why Chloromethane Production Needs a Rethink
Chloromethane is a workhorse molecule in chemical manufacturing, especially for making organosilicon compounds used in sealants, coatings, and other materials, as well as products in rubber, paint, and pharmaceutical industries. Demand is growing into the millions of tons per year, particularly in China. Today it is mostly produced by reacting methanol with hydrogen chloride at high temperatures and pressures. That process consumes a lot of energy, relies on methanol whose price can swing sharply, and involves corrosive chemicals that wear down equipment. A more sustainable route would use abundant methane directly, plus milder chlorine sources such as salty wastewater, and operate near room temperature.
The Challenge of Taming an Inert Gas
Using methane directly is not straightforward. Its tightly bound hydrogen atoms make it one of the toughest molecules to activate, normally requiring temperatures of several hundred degrees Celsius. In liquid-based systems there is an additional hurdle: methane barely dissolves in water, so only a small amount reaches a catalyst surface at any moment. Earlier light-driven and electricity-driven approaches could make chloromethane, but production rates were modest and the catalysts often degraded. The central question the authors tackle is how to both activate methane efficiently and keep a steady supply of it in contact with reactive chlorine species at ambient conditions.
A New Electrode That Mixes Gas and Liquid on Demand
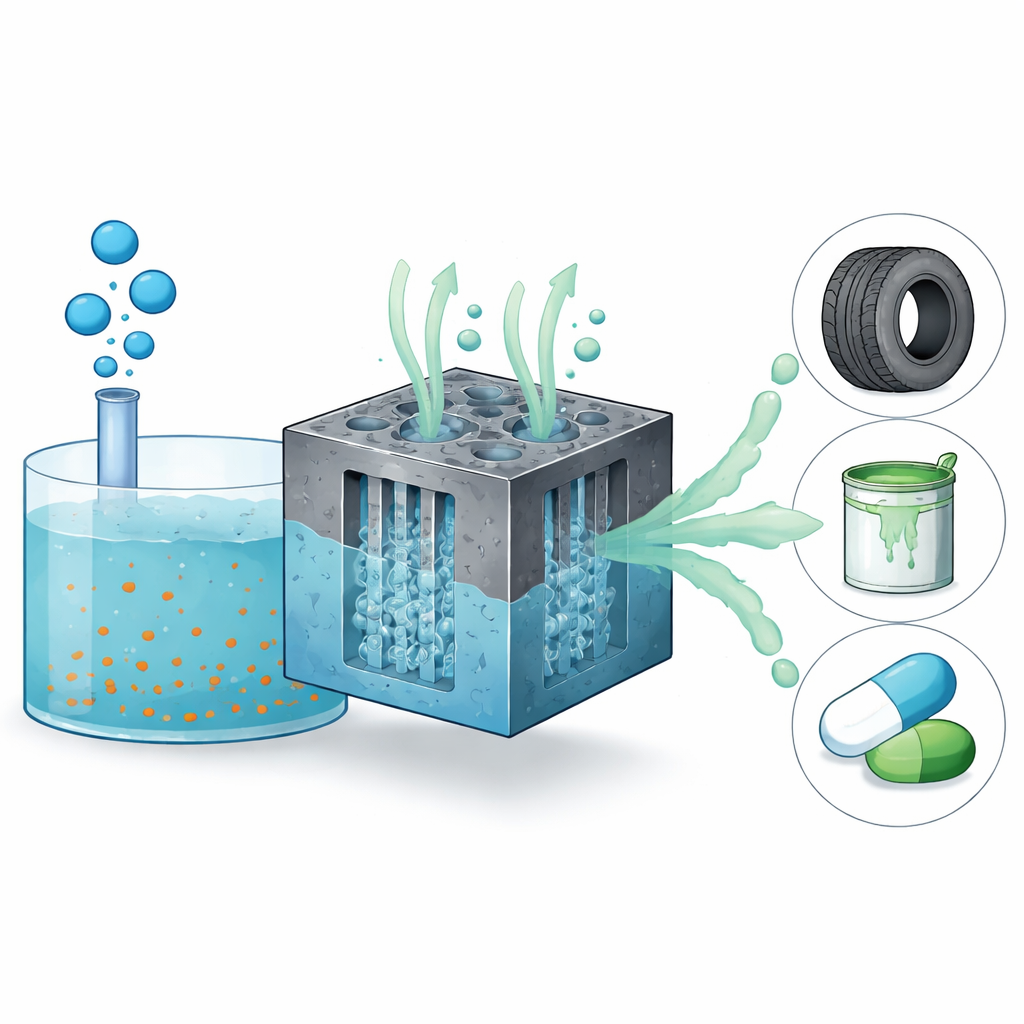
The researchers combined two advances: a catalyst that excels at making reactive chlorine on its surface, and an electrode structure that forces gas and liquid to mingle where that catalyst sits. They used ruthenium oxide, a known industrial material for chlorine-generating reactions, to create surface-bound chlorine species that can strip hydrogen from methane and form chloromethane. Instead of a standard gas diffusion electrode, where methane simply seeps through a thin layer and dissolves slowly, they built a three-dimensional gas convection electrode. In this design, methane gas and salty liquid flow through different directions in a porous carbon foam coated with catalyst and a thin water-loving layer. Pressure differences cause gas and liquid to repeatedly interpenetrate the pores, continually forming fresh contact zones among gas, liquid, and solid.

How the New Design Boosts Production
Computer fluid simulations and mass-transfer modeling show that this gas convection electrode creates dynamic, volume-filling three-phase boundaries instead of just a thin reaction front. Swirling flows and bubbles constantly renew the gas–liquid interface, keeping methane concentrations near the catalyst close to their physical limit rather than falling off with distance. Electrochemical tests confirm the benefit: compared with a conventional gas diffusion electrode using the same catalyst, the new system increases chloromethane production per electrode area by about nineteen times and maintains high selectivity for the desired product. It also suppresses a competing side reaction that simply makes chlorine gas, improving the efficiency with which electrical current is turned into useful chemical bonds. The setup runs stably for at least fifteen hours with little catalyst loss, and scaling up catalyst loading further raises output.
What This Could Mean for Industry and the Environment
To a non-specialist, the key takeaway is that the team has built a kind of “mini chemical factory” where gas and liquid are steered through a porous block so they meet and react far more effectively than before. By pairing this smart flow control with a robust catalyst, they show that chloromethane can be produced from methane and salty solutions at room temperature with impressive rates and efficiency. While more engineering work is needed before this approach reaches full industrial scale, it points to a promising way to turn methane emissions and hypersaline wastewater into a valuable chemical feedstock, potentially lowering energy use, equipment corrosion, and environmental impact in one step.
Citation: Fu, Z., Zhou, Y., Cao, Z. et al. Enhanced methane chlorination via RuO2-gas convection electrode with in-situ generated dynamical three-phase boundaries. Nat Commun 17, 2221 (2026). https://doi.org/10.1038/s41467-026-68845-y
Keywords: methane conversion, chloromethane, electrocatalysis, gas convection electrode, saline wastewater reuse