Clear Sky Science · en
Negative electrode degradation induced by two-stage zinc plating and its recovery in zinc batteries
Why zinc batteries matter for everyday life
Storing electricity safely and cheaply is essential for everything from backing up solar panels on a home to stabilizing the power grid. Zinc‑metal batteries are attractive candidates: zinc is abundant, non‑toxic, and works in water‑based electrolytes that are far safer than the flammable liquids in many lithium batteries. Yet these promising batteries still fade and short‑circuit too quickly. This article uncovers a hidden two‑step growth process on the zinc electrode that causes this damage, and it presents a chemical “self‑healing” strategy that keeps the batteries running far longer.
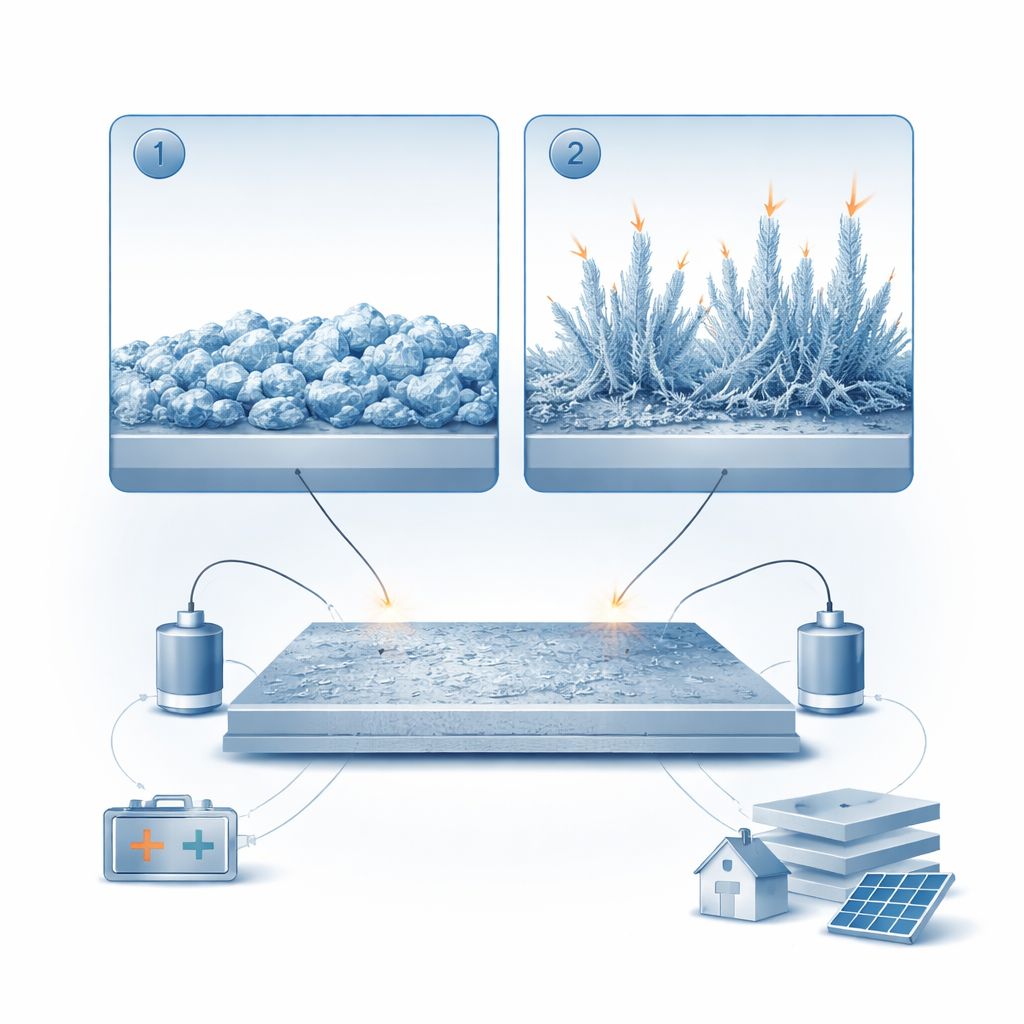
From smooth metal to zinc “moss”
The negative electrode in these batteries is made of zinc metal, which repeatedly plates (adds zinc) and strips (removes zinc) during charging and discharging. Using a transparent, microscope‑equipped cell, the researchers watched how zinc builds up on a metal surface over time. They discovered that zinc does not grow in just one way but in two distinct stages. First, it forms relatively dense, lumpy crystals that make a shiny, compact layer. Then, as plating continues, thin, filament‑like structures sprout from sharp edges and tips. This second, “mossy” zinc stage fills the space between electrodes and eventually connects them, risking internal short circuits.
How mossy zinc becomes dead zinc
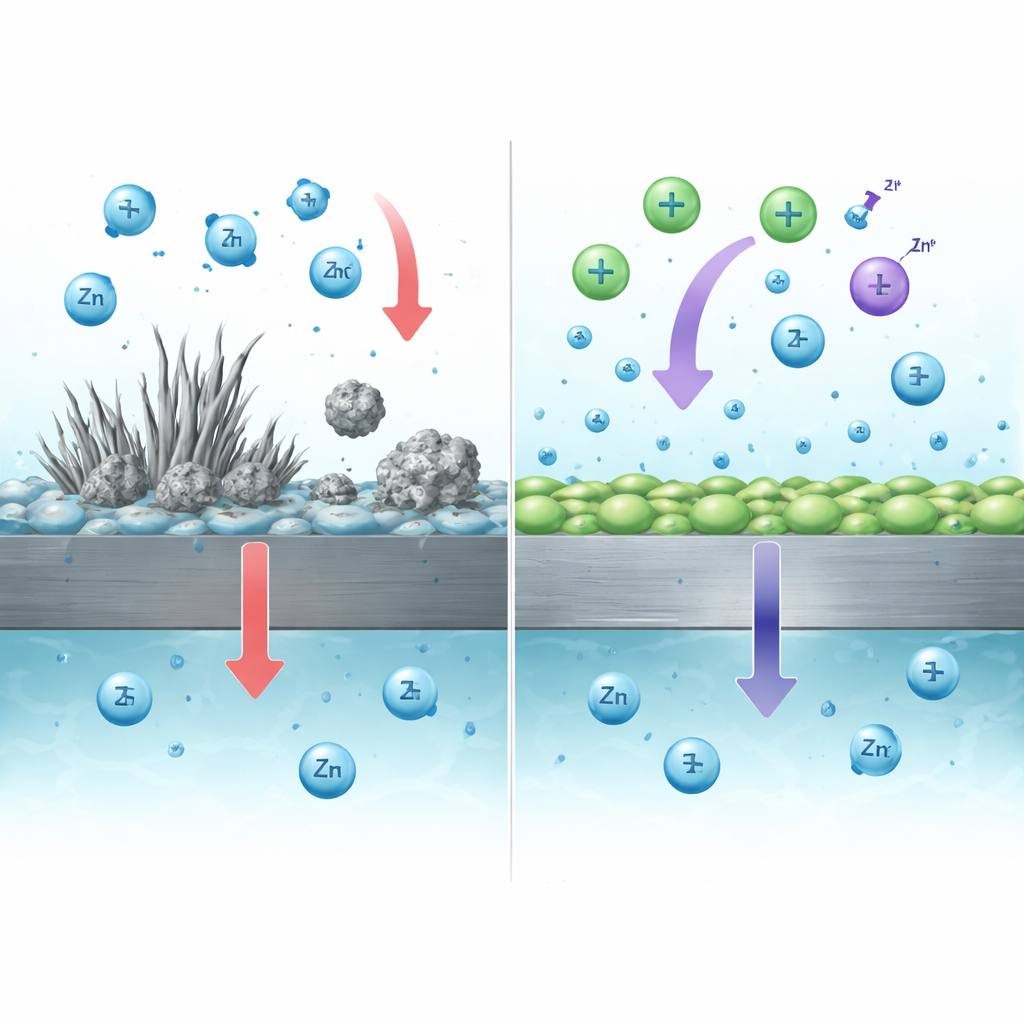
The team combined direct imaging, electron microscopy, and computer simulations to understand why this mossy layer appears. The sharp protrusions on the lumpy zinc concentrate the electric field—an effect similar to lightning rods concentrating electrical discharge. That concentration pulls more zinc ions to the tips, feeding rapid, whisker‑like growth. During the reverse process, when zinc is stripped away, the mossy filaments dissolve first and can lose electrical contact with the underlying metal. What is left behind is “dead” zinc: small, electrically isolated pieces that no longer participate in the battery reaction but still contain valuable active material, leading to capacity loss and rough, unstable surfaces.
Designing a smarter electrolyte
Knowing that mossy growth comes from local ion build‑up at protruding sites, the researchers designed an electrolyte additive that tackles both the formation of mossy zinc and the wasteful dead zinc it produces. They used a salt called acetylcholine iodide, which supplies positively charged organic cations and iodide anions in the same molecule. According to sensitive mass measurements and infrared spectroscopy, the cations strongly and selectively adsorb on the zinc surface, forming a thin positively charged layer that evens out the zinc‑ion flux. This promotes smooth, flat zinc plating instead of filament growth, keeping the surface compact and more resistant to corrosion and hydrogen gas formation.
Reviving lost zinc to extend battery life
The iodide part of the additive plays a different but complementary role. During charging, iodide is partially converted into a mild oxidizing species (I3−) that can react with dead zinc particles and with insulating zinc‑containing by‑products that form on the surface. These reactions turn electrically isolated zinc back into dissolved zinc ions, which can then re‑plate onto the electrode in subsequent cycles. Experiments showed that dead zinc immersed in an iodine‑containing solution dissolved to nearly the theoretical extent expected, and full cells using the dual‑ion additive recovered more charge than would be possible from freshly plated zinc alone—clear evidence that previously lost zinc was being “recycled” inside the battery.
What this means for real batteries
By combining field‑smoothing cations with a zinc‑recycling anion, the new electrolyte enabled zinc electrodes with an average Coulombic efficiency of about 99.7% and stable operation for over 1400 hours at high current and capacity—conditions relevant to grid‑scale storage. Symmetric zinc cells and practical zinc–iodine pouch cells maintained low voltage losses and retained more than 96% of their capacity after hundreds to tens of thousands of cycles, depending on the test. For a layperson, the key message is that the researchers have identified exactly how zinc electrodes degrade—through a two‑stage mossy growth that produces dead metal—and have shown a chemical recipe that both prevents this harmful structure and revives lost material. This dual approach brings safe, water‑based zinc batteries much closer to long‑lasting, large‑scale use in homes and power networks.
Citation: Gan, H., Liu, D., Zhang, Y. et al. Negative electrode degradation induced by two-stage zinc plating and its recovery in zinc batteries. Nat Commun 17, 2067 (2026). https://doi.org/10.1038/s41467-026-68844-z
Keywords: zinc metal batteries, electrode degradation, mossy zinc, electrolyte additives, grid energy storage