Clear Sky Science · en
cGAS-IFN-I responses by extracting nuclear DNA from dying cells via nucleocytosis
How Our Immune System Reads DNA from the Dead
When our cells die during infections, cancer, or everyday wear and tear, their remains must be cleaned up without provoking too much inflammation. This paper reveals a surprising way that immune cells can actually tunnel into the nuclei of dying cells, pull out their DNA, and use it as a danger signal. Understanding this hidden cleanup-and-alarm system could change how we think about antiviral drugs, autoimmunity, cancer therapy, and drug side effects.
A Hidden DNA Alarm Inside Immune Cells
Our cells contain a sensor called cGAS that detects DNA in the wrong place—floating in the cell fluid rather than safely packed in the nucleus or mitochondria. When cGAS encounters such DNA, it switches on a powerful alarm program that produces type I interferons, key antiviral and immune-regulating proteins. Scientists have known that viruses can deliver their genetic material into cells to trigger this pathway, and that bits of our own DNA can sometimes leak out and cause autoimmune disease. But a basic puzzle remained: how do large amounts of our own DNA, especially from dead cells, ever reach cGAS without simply being digested in cellular “stomachs” called lysosomes?
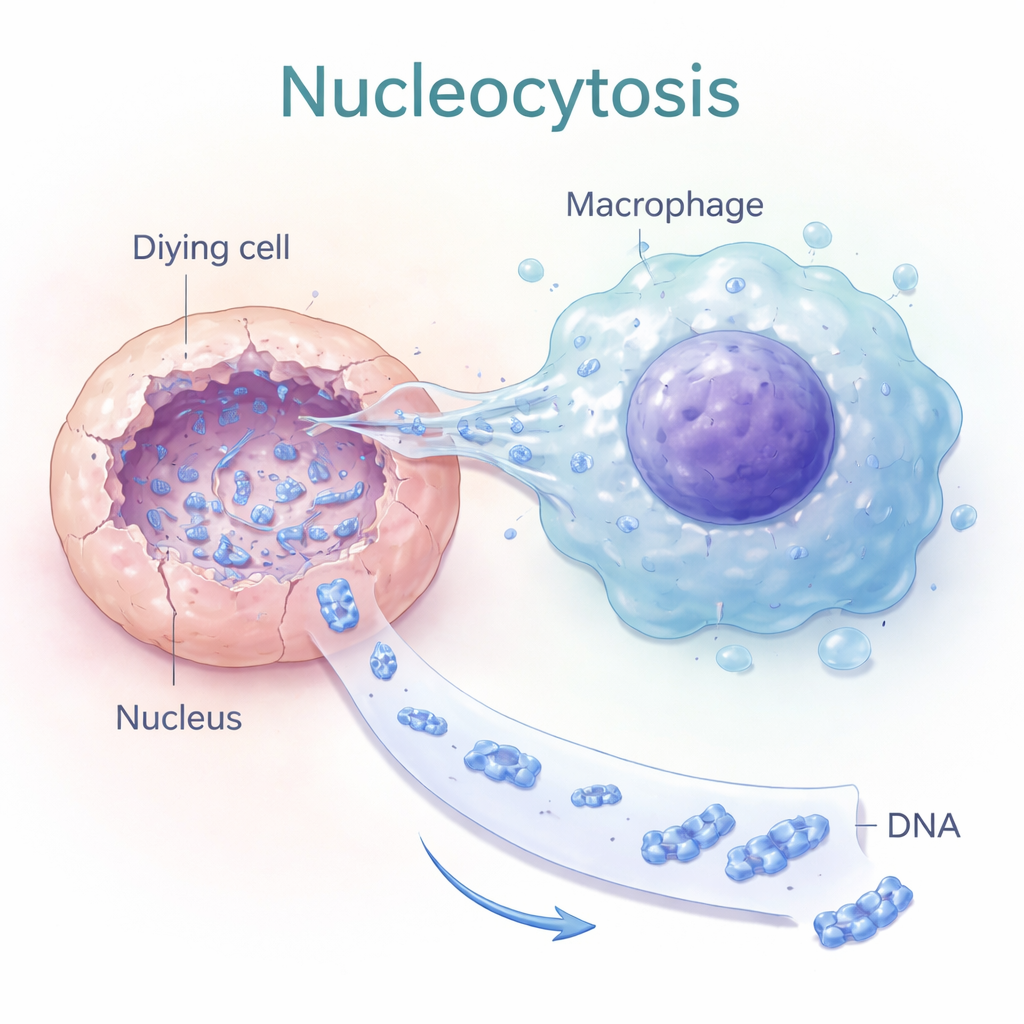
Nucleocytosis: Tunneling into Dead Cell Nuclei
The authors discovered a process they call “nucleocytosis,” in which certain macrophages—immune cells that normally engulf debris—do something much more targeted. Instead of swallowing an entire dead cell, they extend thin, finger-like protrusions directly into the dying cell’s nucleus. Live-cell imaging showed DNA intensity dropping inside the dead cell’s nucleus while increasing inside the attached macrophage, indicating active extraction of nuclear DNA. This transfer depended on the cell’s internal skeleton (actin) and on signaling molecules that control cell shape, suggesting that nucleocytosis is a purposeful mechanical behavior, not a passive leak.
When Antiviral Drugs Turn On the DNA Alarm
During the COVID-19 pandemic, drugs like hydroxychloroquine were widely tested as antivirals, yet their precise actions in the body remained murky. The researchers found that hydroxychloroquine and several related “cationic amphiphilic” drugs can strongly activate the cGAS–STING–interferon pathway—but only in a subset of macrophages. These drugs disturb lysosomes by raising their pH and by blocking an enzyme called PPT1, which helps break down fatty tags on proteins. The combined effect pushes some cells into a special form of death in which a protein called calreticulin accumulates in the nucleus. Macrophages then preferentially extend protrusions toward these calreticulin-rich nuclei, extract DNA via nucleocytosis, and trigger intense interferon production in just a few cells—enough to create a strong local antiviral signal without flooding the whole body.
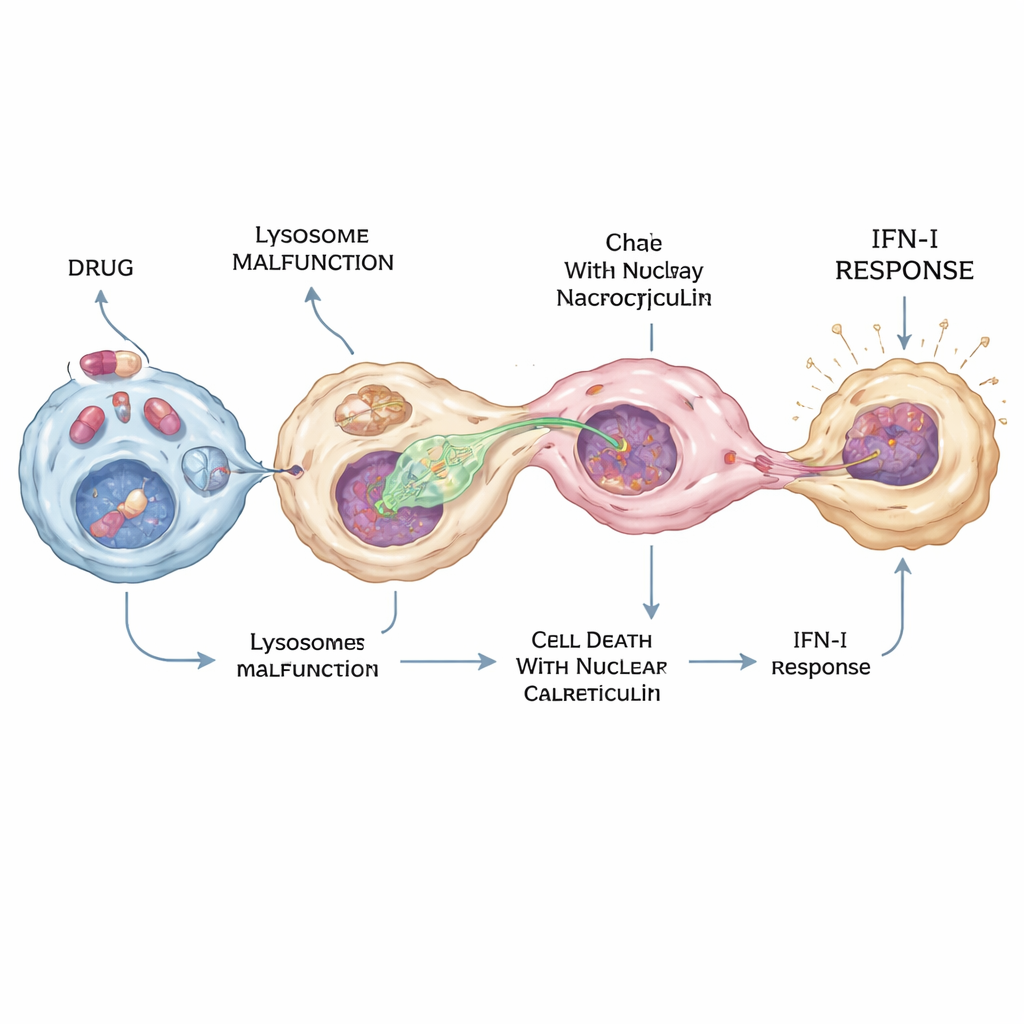
From Local Defense to Disease and Side Effects
By studying both cell cultures and mouse lungs, the team showed that drug-treated macrophages can spark a STING-dependent interferon response that stays largely local—for example, in the lung—rather than becoming systemic. This helps explain why drugs like hydroxychloroquine might show antiviral or anti-tumor benefits in some tissues, but also why they can cause organ-specific side effects such as eye or heart toxicity when used long term. The same nucleocytosis process could plausibly contribute to autoimmune or inflammatory diseases whenever dead cells and lysosomal stress coincide, because it offers a direct route for self-DNA to turn on the cGAS alarm.
Why This Matters for Future Treatments
To a non-specialist, the key message is that immune cells do not just passively eat dead cells; they can actively mine their nuclei for DNA and use it as a signal to call for help. This nucleocytosis pathway explains how our own DNA can safely activate powerful antiviral defenses in a focused, local way, while also highlighting how drugs that disrupt lysosomes or PPT1 might unintentionally flip this switch. By learning to tune nucleocytosis up or down—either by designing smarter antivirals and cancer treatments or by avoiding unwanted self-DNA signals in autoimmune disease—researchers hope to harness this newly uncovered mechanism for safer, more precise therapies.
Citation: Negishi, H., Wada, Y., Shirasaki, Y. et al. cGAS-IFN-I responses by extracting nuclear DNA from dying cells via nucleocytosis. Nat Commun 17, 1658 (2026). https://doi.org/10.1038/s41467-026-68839-w
Keywords: nucleocytosis, cGAS-STING, hydroxychloroquine, type I interferon, macrophages