Clear Sky Science · en
Electrochemical and photocatalytic generation of cis-olefin-bridged carbodication for umpolung [4+1] cycloaddition
Turning a Chemical Weakness into a Strength
Chemists are always looking for faster, cleaner ways to stitch together complex molecules, especially those that could power next‑generation electronics and lighting. This study reveals a clever way to flip the usual reactivity of certain carbon frameworks, using gentle electricity or visible light to create highly charged intermediates that normally would be too unstable. These fleeting species then react efficiently with simple building blocks like amines and water to form rigid, three‑dimensional ring systems that are promising for advanced optoelectronic materials.
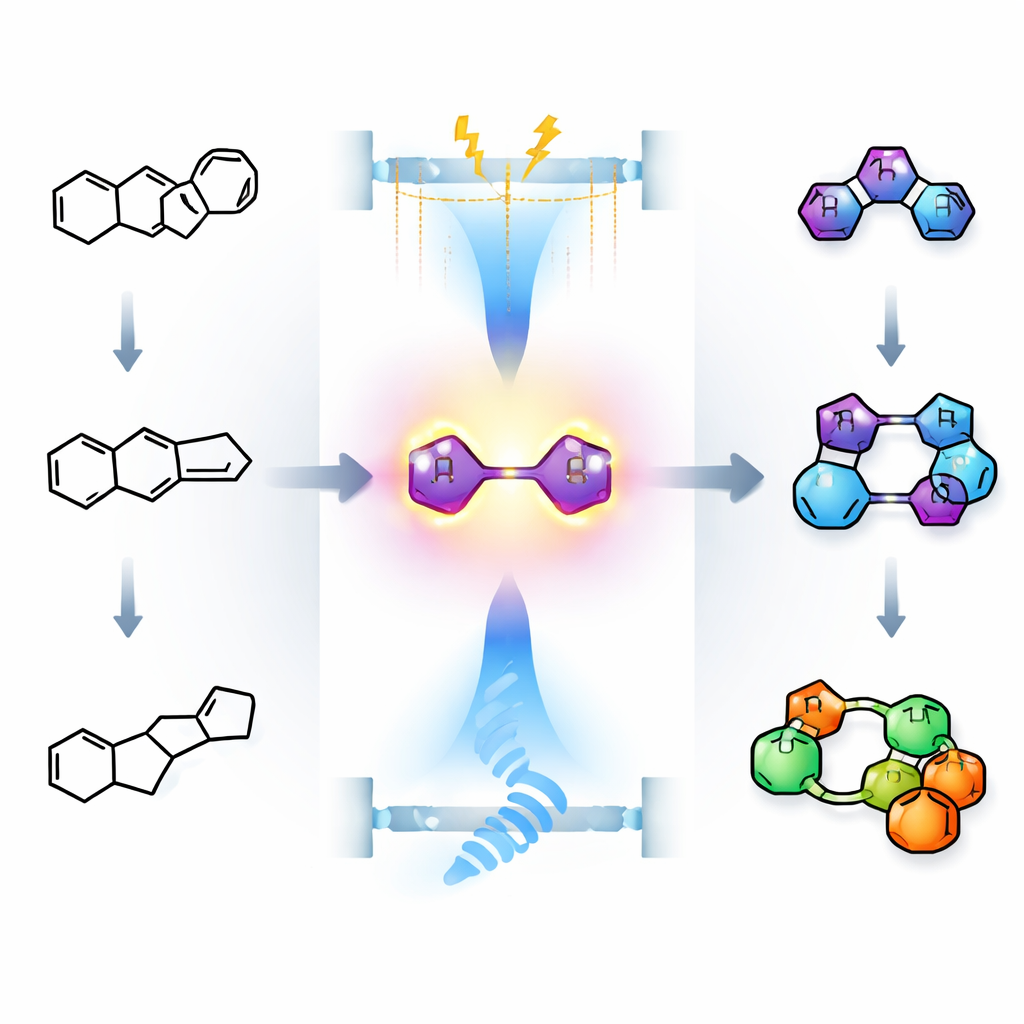
Highly Charged Carbon as a Useful Tool
At the heart of the work are “carbodications” – molecules in which two carbon atoms carry positive charge at the same time. Such species are extremely eager to react and have mostly been used with carbon‑based partners. When heteroatoms such as nitrogen (in amines) or oxygen (in water) are present, they tend to interfere with the strong acids or harsh oxidants traditionally needed to make carbodications, shutting the chemistry down. The authors set out to show that, under much milder conditions, these highly charged molecules can in fact be tamed and directed to react cleanly with amines and water.
Gentle Electricity and Light to Build New Rings
The team designed a special flat molecule: a 1,3‑diene clamped between two large sulfur‑containing ring systems called thioxanthenes. When a small electric current is passed through a solution of this molecule with a supporting salt, the diene portion is selectively oxidized by two electrons to give a “cis‑bridged” dication – a bent structure in which the two positively charged carbon centers are held close together on the same side of the bridge. Careful electrochemical measurements and quantum‑chemical calculations showed that this cis form is strongly favored in the presence of a fluorine‑containing counter‑ion, which helps spread out charge and stabilize the structure. Under these conditions, a wide variety of primary amines can then add to the dication, triggering a [4+1] cycloaddition that closes a new five‑membered ring and produces rigid “dispiro” products with a central partially saturated nitrogen‑containing ring.
Water Joins the Game Under Blue Light
Using water as the partner is trickier, because water itself is easily broken down when current flows, outcompeting the desired reaction. To sidestep this, the researchers switched to a light‑driven strategy. They used a common ruthenium‑based photocatalyst together with a persulfate oxidant in a mixture of organic solvent and water, illuminated with blue light. In this setup, the excited photocatalyst and the radical species derived from persulfate oxidize the same thioxanthene‑diene to the very same cis dication as before, but now without directly electrolyzing water. Water then attacks the dication in a stepwise fashion to give a closely related dispiro product, this time with an oxygen‑containing five‑membered ring at the center. The authors confirmed the structures of both nitrogen and oxygen products by X‑ray crystallography and showed that many differently substituted starting dienes can be transformed this way.
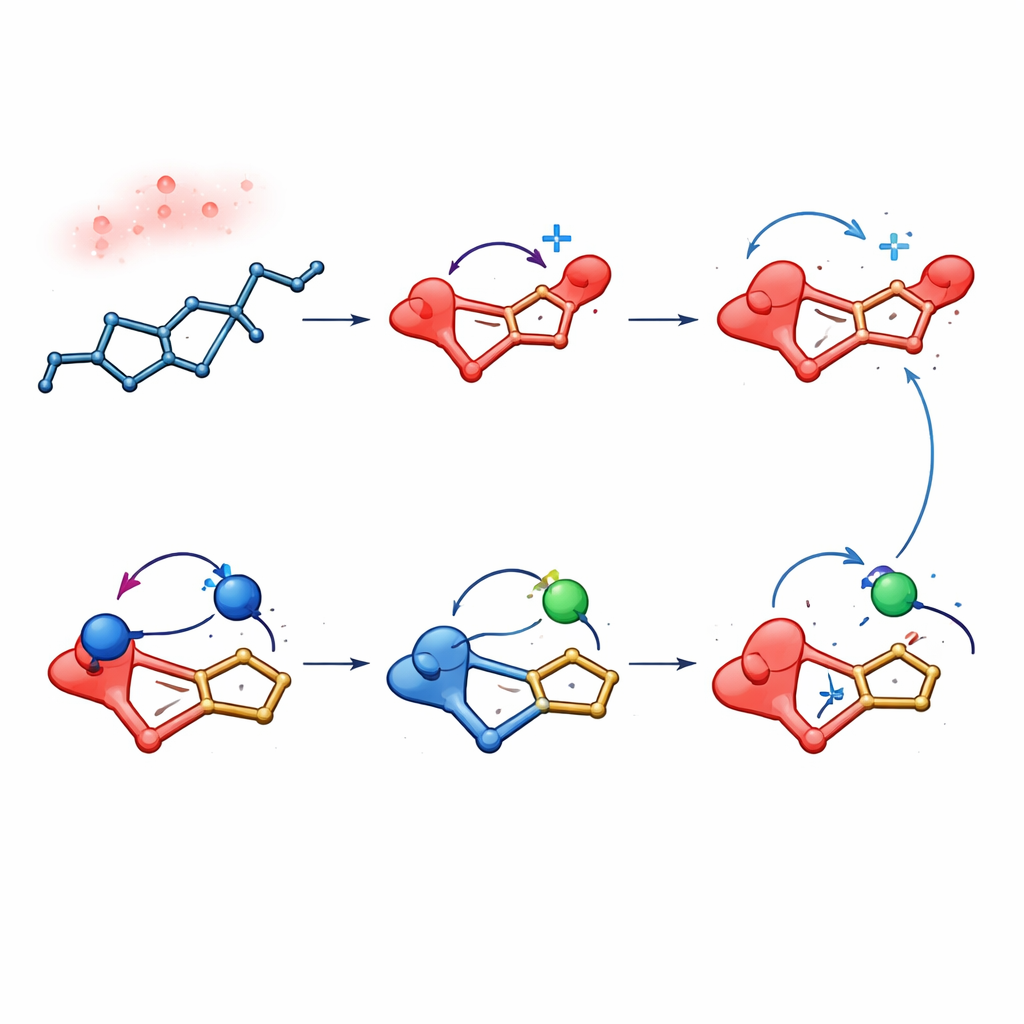
How Subtle Interactions Steer Reactivity
Beyond demonstrating new reactions, the study dissects why they work so well. The electrochemical process depends on a fluorinated co‑solvent, which weakens the tendency of amines to oxidize and instead makes the diene easier to oxidize, ensuring that the desired dication forms first. Calculations further suggest that fleeting hydrogen‑bond contacts between the amine’s N–H group and fluorine atoms from the electrolyte lower the energy barriers for key bond‑forming steps. In both the electric and light‑driven versions, the resulting products share a characteristic electronic pattern: their highest‑energy electrons are localized on the outer thioxanthene units, while the lowest‑lying empty levels sit on the central new ring, a layout that is attractive for charge‑transport and light‑emitting functions.
From Curious Intermediate to Future Devices
Overall, the work turns a once‑awkward, highly charged intermediate into a practical synthetic linchpin. By generating a cis‑bridged carbodication under mild electrochemical or photocatalytic conditions, the authors unlock a new type of [4+1] cycloaddition that joins simple amines or even plain water to complex aromatic scaffolds in a single step. The resulting dispiro compounds are closely related to materials already known to function as efficient hole‑transporters and emitters in devices like organic LEDs and perovskite solar cells. This makes the new reactions not only a conceptual advance in reactive‑intermediate chemistry, but also a promising route toward tailor‑made building blocks for future optoelectronic technologies.
Citation: Matsuyama, H., Yokoyama, K., Sato, T. et al. Electrochemical and photocatalytic generation of cis-olefin-bridged carbodication for umpolung [4+1] cycloaddition. Nat Commun 17, 2270 (2026). https://doi.org/10.1038/s41467-026-68836-z
Keywords: carbodication, electrosynthesis, photoredox catalysis, cycloaddition, optoelectronic materials