Clear Sky Science · en
Priming versus propagating: distinct immune effects of alpha- versus beta-particle emitting radiopharmaceuticals when combined with immune checkpoint inhibition in mice
Turning Radiation into an Immune Ally
Cancer doctors increasingly try to enlist the immune system to fight tumors, but many cancers still ignore or resist these drugs. This study asks a timely question: can different types of targeted radiation be used not just to shrink tumors directly, but to coach the immune system to do a better job—and does the choice of radioactive drug change how that coaching works?
Two Flavors of Precision Radiation
The researchers focused on radiopharmaceutical therapy, a “search‑and‑destroy” approach where a radioactive payload is attached to a molecule that naturally homes to tumors. Once injected, it circulates through the body and delivers radiation to cancer deposits wherever they hide, something ordinary external beam radiation cannot easily do for widespread disease. The team compared two major categories: alpha‑particle emitters, which release extremely dense bursts of energy over a very short distance, and beta‑particle emitters, which send out lower‑density radiation that travels farther through tissue. All were attached to the same tumor‑seeking compound, NM600, so that only the radiation type—not the targeting—differed.
Pairing Radiation with Immune Checkpoint Blockers
On their own, checkpoint inhibitor drugs like anti‑PD‑L1 and anti‑CTLA4 can lift the brakes on immune cells, but they work best when the tumor is already visible to the immune system. In mouse models of melanoma, prostate cancer, and colorectal cancer, the scientists gave a carefully chosen low radiation dose from either alpha‑ or beta‑based NM600, with or without a combination of checkpoint inhibitors. They also varied when the immune drugs were given—before, shortly after, or well after the radioactive treatment—to see how timing affected outcomes such as tumor growth, survival, and long‑term immune memory.
When Beta Radiation Shines: Boosting an Existing Response
In an “immune‑hot” colorectal cancer model that already responds to immunotherapy, the best results came from beta‑emitting NM600, especially when the immune drugs were started early or at an intermediate time point. Tumors shrank more, mice lived longer, and cured animals often rejected the same cancer when it was re‑introduced months later. Detailed immune profiling showed that beta‑based treatment plus checkpoint blockade did not so much create a new response as amplify one that was already there: existing tumor‑specific killer T cells expanded, became more activated, and produced more attack signals. In short, beta emitters were excellent at propagating an ongoing immune response.
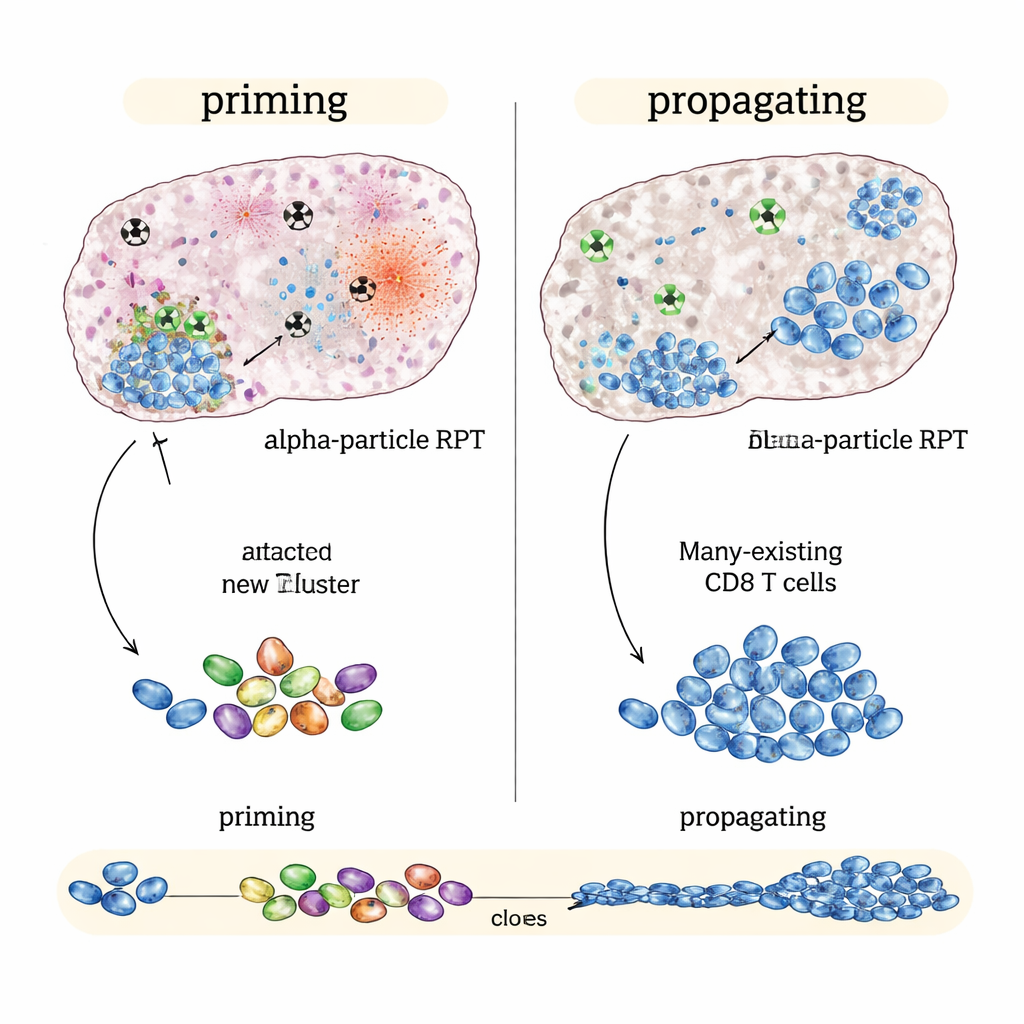
When Alpha Radiation Shines: Sparking New Immune Attack
In contrast, in “immune‑cold” tumors such as a melanoma and a prostate cancer model that barely respond to checkpoint drugs alone, the alpha‑emitting version performed better than beta emitters at the same average tumor dose. Alpha‑based therapy plus immune drugs slowed tumor growth more and further extended survival. Single‑cell genetic analyses suggested why: alpha radiation caused intense, localized damage that seemed to generate strong alarm signals inside the tumor while sparing nearby immune structures. This pattern was linked to broader, more diverse T‑cell responses and signs that new tumor‑recognizing killer T cells were being formed and linked to long‑lived memory cells—evidence of immune priming rather than mere amplification.
Why the Type and Timing of Radiation Matter
Across the different models, early or intermediate timing of checkpoint inhibitors—roughly coinciding with when radiation‑induced danger signals peak—consistently outperformed delayed treatment. The work suggests a practical rule of thumb: in cancers already visible to the immune system, low‑dose beta‑based radiopharmaceuticals may be ideal partners to checkpoint inhibitors because they propagate and strengthen existing immunity. In more immune‑resistant cancers, high‑impact alpha emitters may be better suited to prime fresh T‑cell responses and turn a “cold” tumor “hot.” For patients, this means that not all radioactive drugs are interchangeable; matching the isotope and schedule to the tumor’s immune personality could make combination radio‑immunotherapy both more effective and more durable.
Citation: Kerr, C.P., Jin, W.J., Liu, P. et al. Priming versus propagating: distinct immune effects of alpha- versus beta-particle emitting radiopharmaceuticals when combined with immune checkpoint inhibition in mice. Nat Commun 17, 2044 (2026). https://doi.org/10.1038/s41467-026-68834-1
Keywords: radiopharmaceutical therapy, alpha vs beta radiation, immune checkpoint inhibitors, cancer immunotherapy, tumor microenvironment