Clear Sky Science · en
Safety and biologic activity of a bispecific T cell receptor targeting HIV Gag in males living with HIV: a first-in-human trial
Why this new HIV study matters
People living with HIV today can expect near‑normal lifespans thanks to modern drug cocktails. But these drugs, called antiretroviral therapy (ART), do not remove the virus; they simply keep it in check. Hidden pockets of HIV remain in the body and can restart infection if treatment stops. This study tests a new kind of precision immune therapy—built from a lab‑made T cell receptor—that aims to train the immune system to hunt down those hiding spots safely in people who are already well controlled on ART.
The hidden virus that won’t go away
Even when blood tests show “undetectable” virus, HIV lingers as genetic material tucked inside long‑lived cells, mainly a type of white blood cell called CD4 T cells. These reservoir cells carry silent but intact virus that can wake up again, forcing people to take daily pills for life. Completely erasing every infected cell is extremely challenging and has only been achieved in a handful of people who underwent risky bone‑marrow transplants for cancer. Many researchers instead aim for a “functional cure”: shrinking the reservoir enough that the body’s own defenses can keep HIV down without constant medication.
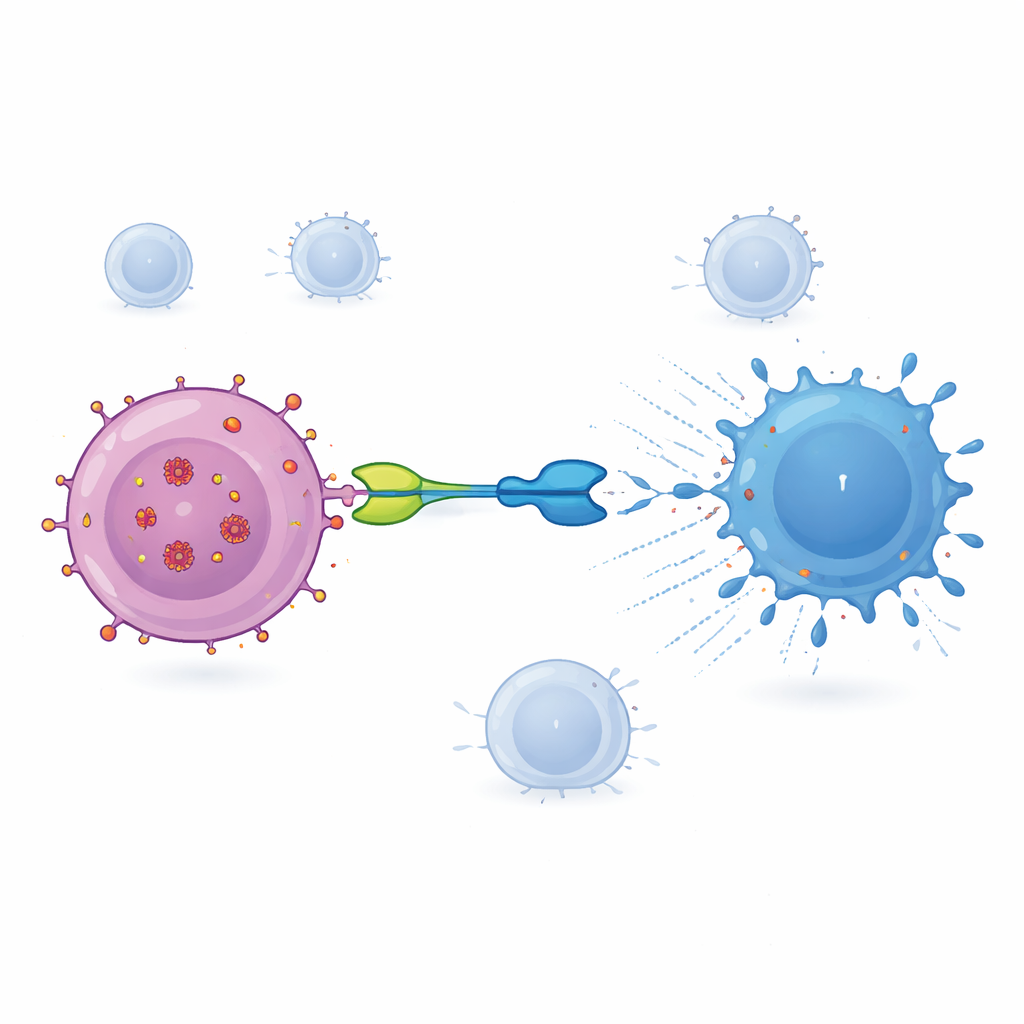
A designer molecule to redirect T cells
The therapy tested here, called IMC‑M113V, is a small protein that works like a molecular adapter between infected cells and the immune system. One end of IMC‑M113V is an engineered T cell receptor that recognizes a tiny HIV fragment from the Gag protein displayed on the surface of infected cells in the context of a common immune marker (HLA‑A*02:01). The other end latches onto CD3, a structure present on all T cells. When IMC‑M113V binds both sides at once, it pulls ordinary T cells into close contact with an HIV‑infected cell and triggers them to kill it. In lab tests, this molecule was extremely sensitive, detecting only a few copies of the viral fragment on a cell’s surface and effectively eliminating cells infected with several common HIV variants, while showing no meaningful activation against a panel of healthy human cells.
First test in people living with HIV
To see whether this approach is safe in humans, the researchers ran an early‑phase trial in twelve adult men living with HIV in the United Kingdom, Belgium and Spain. All participants had well‑controlled infection on ART, high CD4 counts and carried the required HLA type. Each person received a single intravenous dose of IMC‑M113V at one of three low dose levels and was then closely monitored for a month. The main focus at this stage was safety: looking for side effects such as fever, severe inflammation or neurological problems, which can occur with other powerful T cell‑engaging drugs used in cancer treatment.
What the researchers saw in the clinic
Across all doses, IMC‑M113V was generally well tolerated. Half of the participants reported some side effects, mostly mild issues such as fatigue or skin irritation, and no one developed serious problems like cytokine release syndrome or neurotoxicity. Blood tests showed that levels of the drug rose and fell within about a day, with a half‑life of roughly 15–22 hours. At the highest dose (15 micrograms), several participants showed temporary rises in inflammatory molecules, especially interleukin‑6, and signs that their T cells had become activated and more capable of producing cell‑killing proteins. These immune changes were strongest in volunteers whose viruses carried Gag variants that IMC‑M113V can bind particularly tightly, suggesting that the drug was engaging its intended targets in the body. However, after a single dose, there was no measurable reduction in the size of the HIV reservoir in blood, as judged by viral RNA and intact viral DNA inside CD4 cells.
What this means for future HIV treatment
To a non‑specialist reader, the main message is that this study delivers an important first proof of concept: a highly targeted immune “bridge” molecule can be given safely to people with well‑controlled HIV and can wake up their T cells to recognize cells harboring the virus. It did not cure HIV or allow people to stop ART, but that was not the goal of this first‑in‑human trial. The findings support testing higher and repeated doses, possibly combined with drugs that coax more infected cells to reveal themselves, and expanding the approach to cover other immune types beyond HLA‑A*02:01. If future studies confirm that this strategy can reliably shrink viral reservoirs without dangerous side effects, it could become a key part of combination treatments aimed at long‑term, drug‑free control of HIV.
Citation: Vandekerckhove, L., Fox, J., Mora-Peris, B. et al. Safety and biologic activity of a bispecific T cell receptor targeting HIV Gag in males living with HIV: a first-in-human trial. Nat Commun 17, 2207 (2026). https://doi.org/10.1038/s41467-026-68833-2
Keywords: HIV cure, T cell therapy, viral reservoir, bispecific receptor, clinical trial