Clear Sky Science · en
Near-unity CO2-to-ethylene photoconversion over low coordination single-atom catalysts
Turning a Greenhouse Gas into a Useful Fuel
Carbon dioxide is often cast as the villain of climate change, but what if we could turn this waste gas into valuable fuels using only sunlight and simple materials? This study shows how carefully arranging single metal atoms in a solid can make solar-powered reactors that transform carbon dioxide into ethylene, a key building block for plastics and chemicals, with almost perfect efficiency.
Why Ethylene Matters for Everyday Life
Ethylene is one of the world’s most important industrial molecules. It underpins the production of plastics, solvents, and many everyday products. Today, ethylene is mostly made from fossil fuels at high temperatures, releasing large amounts of carbon dioxide. A process that instead starts from carbon dioxide and runs on sunlight could both cut emissions and recycle a major greenhouse gas. The challenge is that turning carbon dioxide into multi‑carbon products like ethylene is much harder than making simple one‑carbon products such as carbon monoxide or methane, because it requires two carbon fragments to meet and bond in just the right way on a catalyst surface.
A New Kind of Atomically Tuned Surface
The researchers tackled this problem using a family of materials known as metal sulfides. On their own, these materials tend to hold onto reactive carbon fragments too weakly, so the fragments float away before they can pair up. The team redesigned zinc sulfide by inserting isolated manganese atoms into its lattice and deliberately removing nearby sulfur atoms, creating what they call low‑coordination manganese single‑atom sites. At these spots, a manganese atom is connected to fewer neighbors than usual and sits next to a tiny sulfur vacancy, subtly reshaping the local electronic landscape.
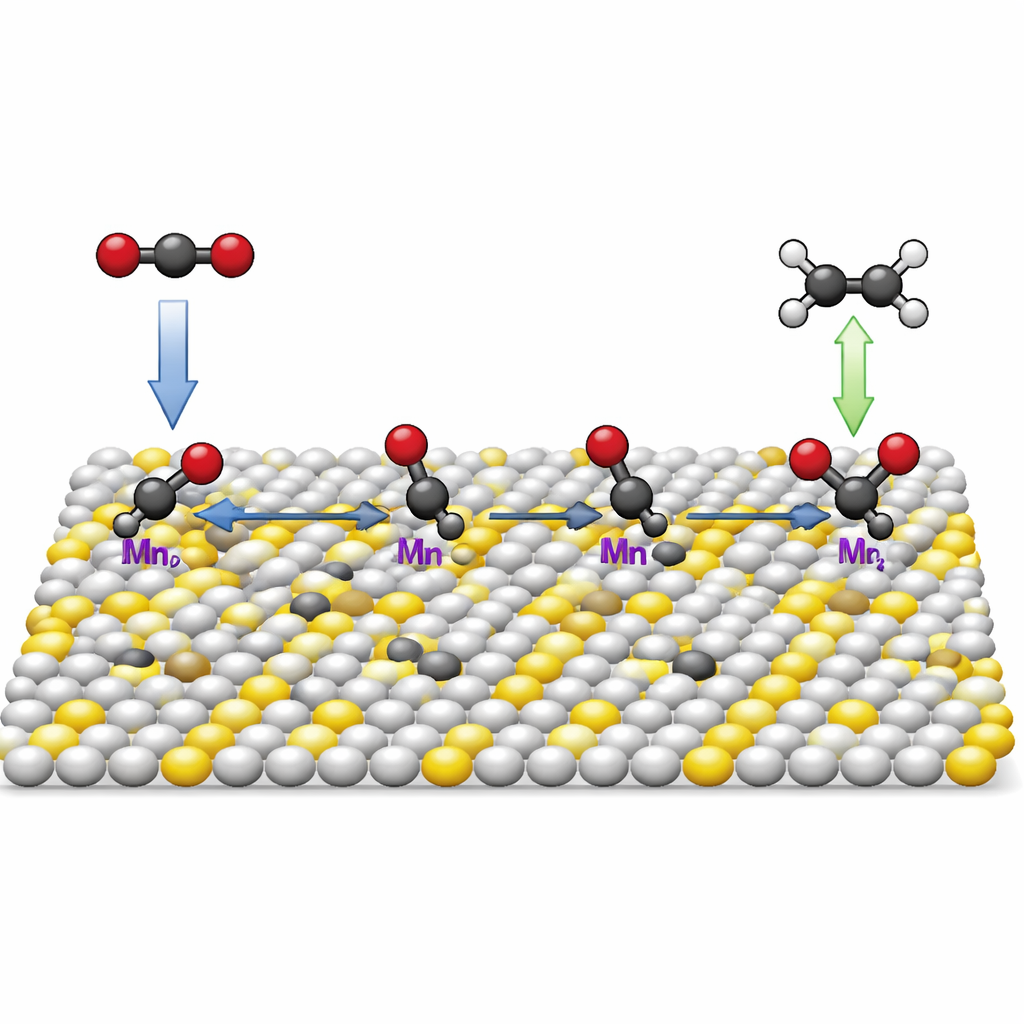
How the Catalyst Grabs and Joins Carbon
Through computer simulations and in‑situ infrared measurements taken while the reaction was running, the authors showed that these special manganese sites bind key carbon‑based intermediates much more strongly and selectively than ordinary zinc sulfide. In particular, the surface grips carbon monoxide fragments and their hydrogenated cousins just firmly enough to keep them in place, but not so tightly that they cannot move or react. This balance allows one fragment to be partially hydrogenated into a *CHO species and then couple asymmetrically with a neighboring *CO fragment to form a *COCHO unit, a crucial two‑carbon stepping stone that leads onward to ethylene.
Sunlight In, Clean Fuel Out
When tested under simulated sunlight in water without any added helper chemicals, the optimized manganese‑doped zinc sulfide produced ethylene with remarkable performance: 99.1% of the carbon‑based gas products were ethylene, and the production rate was nearly 59 times higher than that of plain zinc sulfide. Competing reactions, such as generating hydrogen gas or simple one‑carbon products, were strongly suppressed. The catalyst remained stable over more than 200 hours of continuous operation, and similar low‑coordination designs using other metals also boosted ethylene production, showing that this design principle is broadly applicable.
What This Means for a Carbon‑Smart Future
In simple terms, the study demonstrates that carefully "unbalancing" how a single metal atom sits in a solid can dramatically change what that surface does with carbon dioxide. By giving manganese atoms fewer neighbors and nearby empty spots, the researchers created tiny reaction hot spots that favor joining carbon atoms into ethylene rather than making simpler, less useful molecules. While scaling such photocatalysts to industrial levels will require further advances, this atomic‑level engineering offers a promising route toward future solar refineries that convert waste carbon dioxide and water into valuable multi‑carbon fuels and chemicals.
Citation: Tang, Z., Wang, Y., Qin, T. et al. Near-unity CO2-to-ethylene photoconversion over low coordination single-atom catalysts. Nat Commun 17, 2081 (2026). https://doi.org/10.1038/s41467-026-68830-5
Keywords: CO2 conversion, photocatalysis, single-atom catalysts, ethylene fuel, solar fuels