Clear Sky Science · en
A multi-ancestry genetic reference for the Quebec population
Why Quebec’s DNA Matters to Everyone
The DNA of a population is like a living archive of its history, migrations, and health risks. Quebec offers a rare natural experiment: a mix of long‑standing French‑Canadian founder families and more recent arrivals from places such as Haiti and Morocco, all living in the same environment and health‑care system. This study builds a detailed genetic reference for Quebec, showing how this diversity can sharpen diagnoses, reveal hidden disease risks, and improve the power of medical research far beyond the province itself. 
A Living Map of Quebec’s Origins
The researchers analyzed genetic data from more than 29,000 adults in CARTaGENE, Quebec’s largest population‑based cohort. Using statistical tools that group people by shared DNA, they found patterns that mirror both the province’s history and its geography. Many residents trace their roots to a small number of French settlers who arrived in the 1600s and 1700s. This founder effect left telltale signatures in certain regions, such as Saguenay–Lac‑Saint‑Jean, where some rare disease variants are unusually common. At the same time, Quebec’s modern cities have attracted immigrants from across the globe, creating clusters of genetic ancestry from North and Sub‑Saharan Africa, the Caribbean, Latin America, and elsewhere, often aligning with people’s reported country of birth and language.
Migrations Written in DNA
By zooming in on people whose grandparents were born in the same country, the team could see migration waves preserved in genetic data. For example, Quebec residents of Moroccan descent separated into two clear groups that match historical immigration from Muslim and Jewish communities, each arriving in different decades. Similarly, people with Haitian grandparents show DNA patterns reflecting a mix of African and European origins, echoing the country’s colonial past. The study also confirms that, because CARTaGENE recruited mostly urban residents and excluded those living on reserves, it captures only a small fraction of Indigenous ancestry that existed in the region before European colonization, highlighting an important gap future work must address.
Discovering Rare and Hidden Variants
The team sequenced the entire genomes of more than 2,100 participants, including large subsets of French‑Canadian, Haitian, and Moroccan ancestry. This deep look uncovered over 80 million genetic variants, many of them rare or previously unreported. Some are “high‑impact” changes that can disrupt genes and cause disease. The researchers built public online browsers where doctors and scientists can quickly check how common a variant is in these specific Quebec groups—a crucial step in deciding whether a DNA change in a patient is likely harmful or harmless. They also catalogued structural variants (large deletions or duplications of DNA) and immune‑system gene versions (HLA types) that differ in frequency from those in global reference datasets, including variants tied to drug reactions and autoimmune disease.
From Genealogy to Disease Risk
Quebec’s unusually rich historical records allowed the authors to link modern DNA to family trees going back centuries. They focused on SPG7, a gene where damaging variants cause a form of adult‑onset leg stiffness and weakness. By combining whole‑genome data, ancestry‑aware math, and genealogies, they traced several SPG7 variants to specific founder families who settled early in New France and mapped how carrier frequencies vary from region to region. This reveals that some disease‑causing variants once thought to be unique to Quebec are actually widespread in Europe, while others truly arose and spread locally. Such fine‑scale maps help estimate how many people might carry a given risk variant and where targeted screening could be most useful. 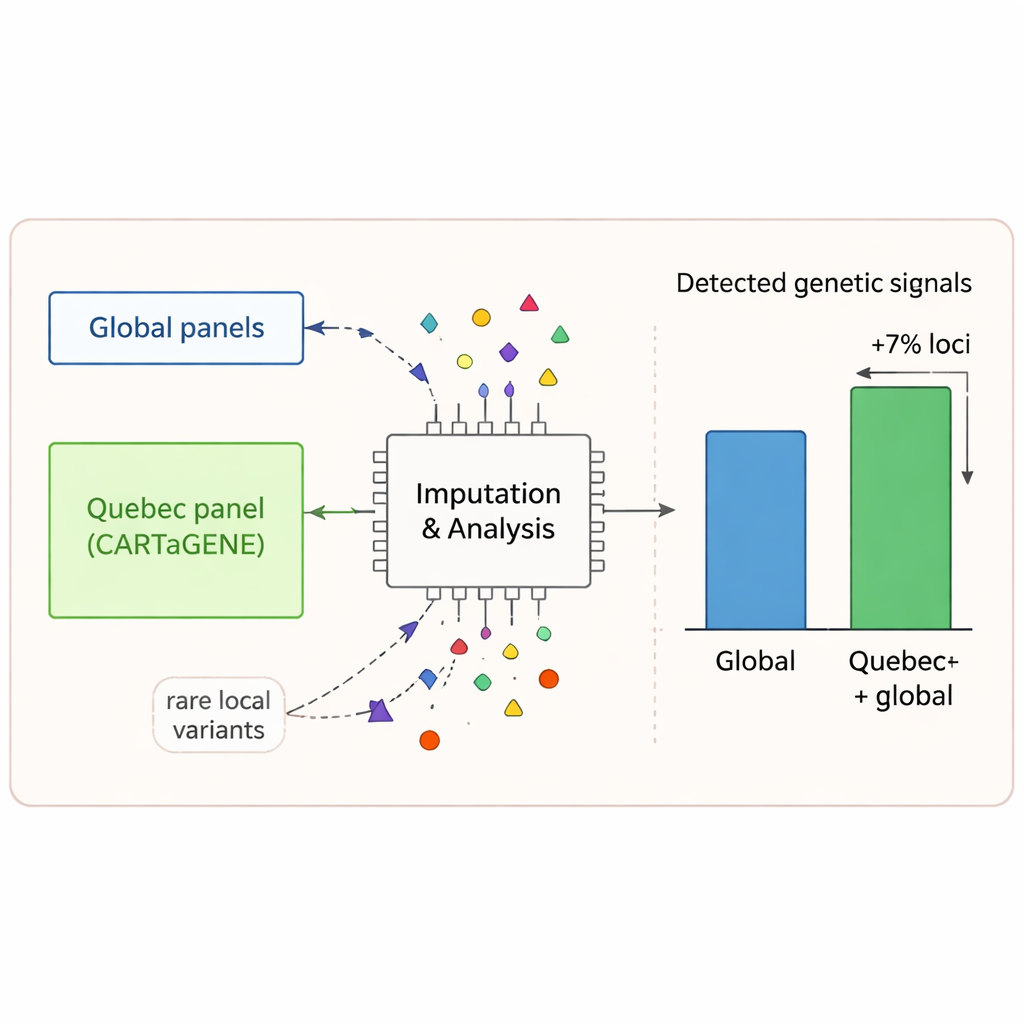
Boosting Discovery with a Local Reference Panel
To make the most of their data, the researchers built a “reference panel” of Quebec haplotypes—long stretches of DNA that tend to be inherited together—and used it to improve a key process called genotype imputation. Imputation fills in missing genetic information in large studies by inferring unseen variants from nearby markers. When they compared this locally tailored panel with a much larger international panel, they found that the Quebec panel recovered many variants that global resources missed, especially those enriched in French‑Canadian, Haitian, or Moroccan ancestry. Genome‑wide association studies on 42 health‑related traits showed that using the Quebec panel alone uncovered more genetic signal than the global panel, and combining both increased the number of associated regions by about 7%. This means a thoughtfully built local resource can reveal disease‑related variants that would otherwise stay invisible.
What This Means for Health and Medicine
For non‑specialists, the take‑home message is that who you are, where your ancestors came from, and where you live all leave fingerprints in your DNA—and these fingerprints matter for diagnosis and treatment. By building a detailed multi‑ancestry genetic reference for Quebec, and making summary results openly available, this work gives clinicians better tools to interpret patients’ genetic tests and gives researchers a richer foundation for studying common conditions such as heart disease, thyroid disorders, and gout. Just as important, it illustrates why genomics must move beyond one‑size‑fits‑all datasets and invest in diverse, locally informative cohorts if precision medicine is to benefit everyone.
Citation: McClelland, P., Femerling, G., Laflamme, R. et al. A multi-ancestry genetic reference for the Quebec population. Nat Commun 17, 1319 (2026). https://doi.org/10.1038/s41467-026-68820-7
Keywords: Quebec genetics, population genomics, founder effects, rare disease variants, precision medicine