Clear Sky Science · en
Chemically guided single-cell transcriptomics reveals sulfotransferase-mediated scaffold remodeling in securinine biosynthesis
Why a Shrub’s Chemistry Matters
Securinega alkaloids are potent molecules found in a small ornamental shrub, Flueggea suffruticosa, long studied for possible treatments for cancer and neurological diseases. Yet, until now, no one really knew how this plant actually built these intricate compounds. This study combines modern single-cell gene reading with clever chemical experiments to reveal, step by step, how the plant assembles and reshapes these molecules—and uncovers a surprising new job for a common type of enzyme along the way. 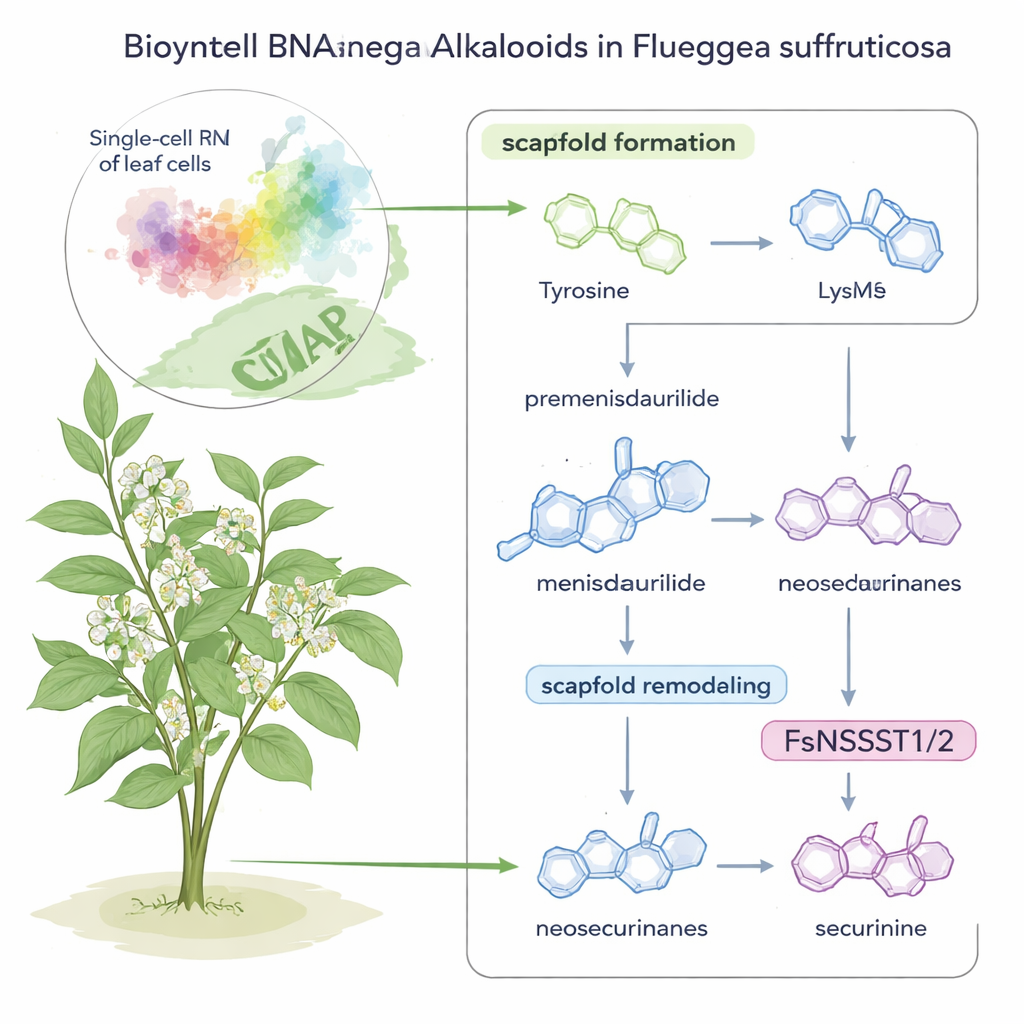
Building Blocks from Everyday Amino Acids
The story begins with two familiar nutrients: the amino acids tyrosine and lysine. In F. suffruticosa, tyrosine is transformed into an unusual ring-shaped molecule called premenisdaurilide, which is then reduced to menisdaurilide by a newly discovered enzyme the authors name FsMS (for “menisdaurilide synthase”). In parallel, lysine is converted into a small nitrogen-containing ring called 1-piperideine by another enzyme, FsPS, discovered in earlier work. When menisdaurilide and 1-piperideine meet in slightly basic, water-like conditions, they spontaneously fuse to form “neosecurinane” alkaloids—intermediate structures on the road to the final medicinal compounds.
Watching Chemistry Happen in Real Time
To prove that these proposed steps are not just theoretical, the team synthesized versions of the suspected intermediates tagged with heavy carbon atoms. Feeding these labeled molecules to plant extracts let the scientists trace exactly where the atoms ended up. They saw labeled menisdaurilide converted into labeled neosecurinanes, and those into the known alkaloids allosecurinine and securinine, confirming that these intermediates really lie along the natural pathway. Importantly, some of the key ring-forming reactions occurred even in boiled extracts, showing that parts of the route can proceed without enzymes, driven simply by the chemistry of the molecules themselves.
Zooming In on the Right Cells
Knowing which molecules appear where is only half the puzzle; identifying the genes that control each step requires knowing which cells are doing the work. The researchers sequenced RNA from thousands of individual cells taken from F. suffruticosa leaves, clustering them into distinct cell types based on gene activity patterns. One cluster, associated with the leaf’s veins, stood out: it strongly expressed the known pathway genes and many enzymes connected to tyrosine, lysine, and sulfur metabolism. By examining which genes varied in lockstep with the pathway enzymes within this cluster, the team zeroed in on FsMS and two sulfotransferases, FsNSST1 and FsNSST2, as prime candidates for the missing steps. 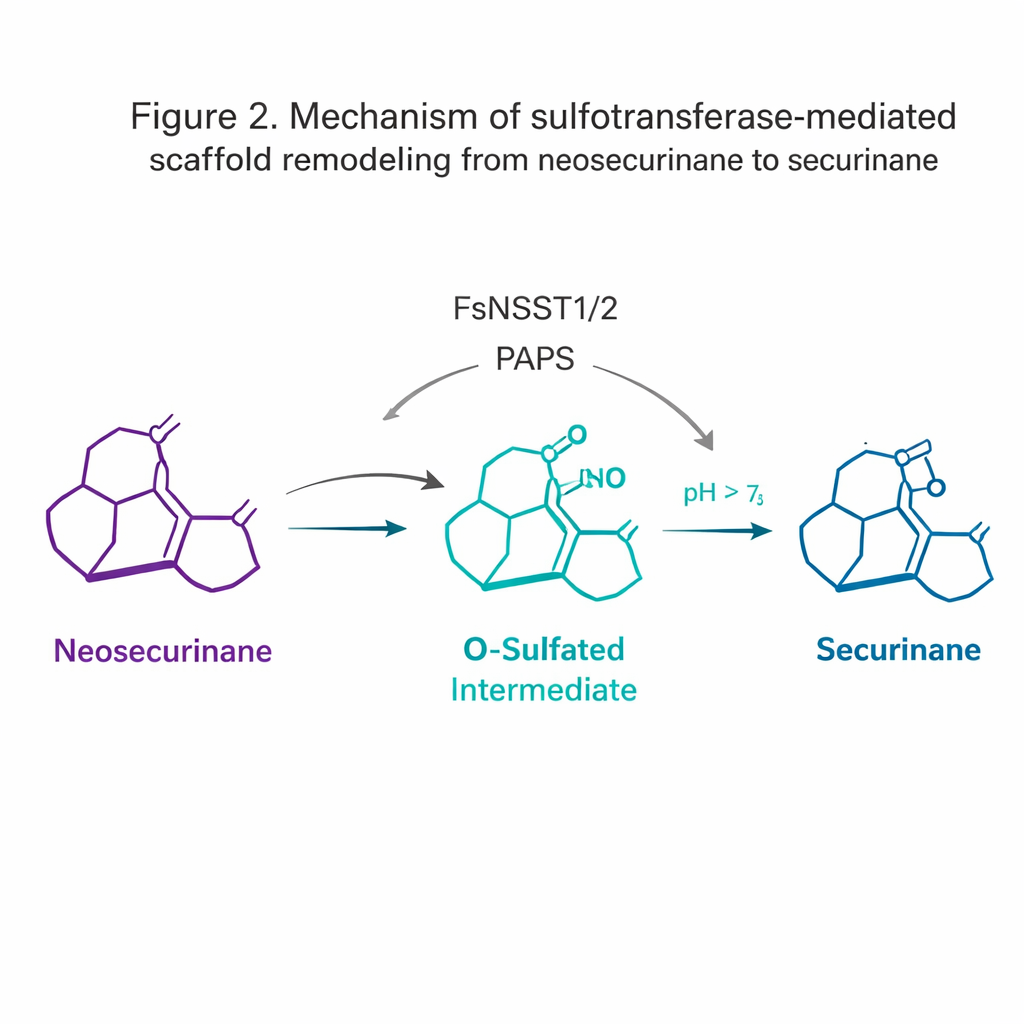
An Enzyme That Remodels the Molecular Scaffold
The most surprising discovery came from the sulfotransferases. Sulfotransferases usually tack a sulfate group onto molecules to improve their solubility or mark them for breakdown. Here, FsNSST1 and FsNSST2 instead act as switchmakers: they attach sulfate to the neosecurinane scaffold, briefly turning it into a high-energy “O-sulfated” intermediate. This activated form then undergoes a spontaneous 1,2-amine shift—a small rearrangement in which a nitrogen atom migrates—converting the [2.2.2] bicyclic “neosecurinane” framework into the [3.2.1] “securinane” framework. This subtle reshaping step is what gives rise to the distinctive tetracyclic core of the biologically active alkaloids.
Why This Pathway Matters
For a non-specialist, the key message is that the plant assembles securinega alkaloids in two main phases: first, it builds a preliminary ring system from everyday amino acids; then, it chemically remodels that scaffold into a more complex form using a sulfate “trigger.” By combining cell-by-cell gene profiling with isotope-labeled chemistry, the authors map this route in detail and reveal that sulfotransferases—enzymes found across all forms of life—can do more than just decorate molecules; they can initiate wholesale rearrangements of their shape. Understanding this pathway not only clarifies how a promising class of drug-like molecules is made in nature, but also opens the door to engineering crops or microbes to produce new securinega-inspired medicines.
Citation: Choung, S., Kang, G., Kim, T. et al. Chemically guided single-cell transcriptomics reveals sulfotransferase-mediated scaffold remodeling in securinine biosynthesis. Nat Commun 17, 1954 (2026). https://doi.org/10.1038/s41467-026-68816-3
Keywords: plant alkaloids, biosynthesis, single-cell transcriptomics, sulfotransferase, natural product chemistry