Clear Sky Science · en
ALKB-1-dependent tRNA methylation is required for efficient paternal mitochondrial elimination
Why fathers’ mitochondria quietly disappear
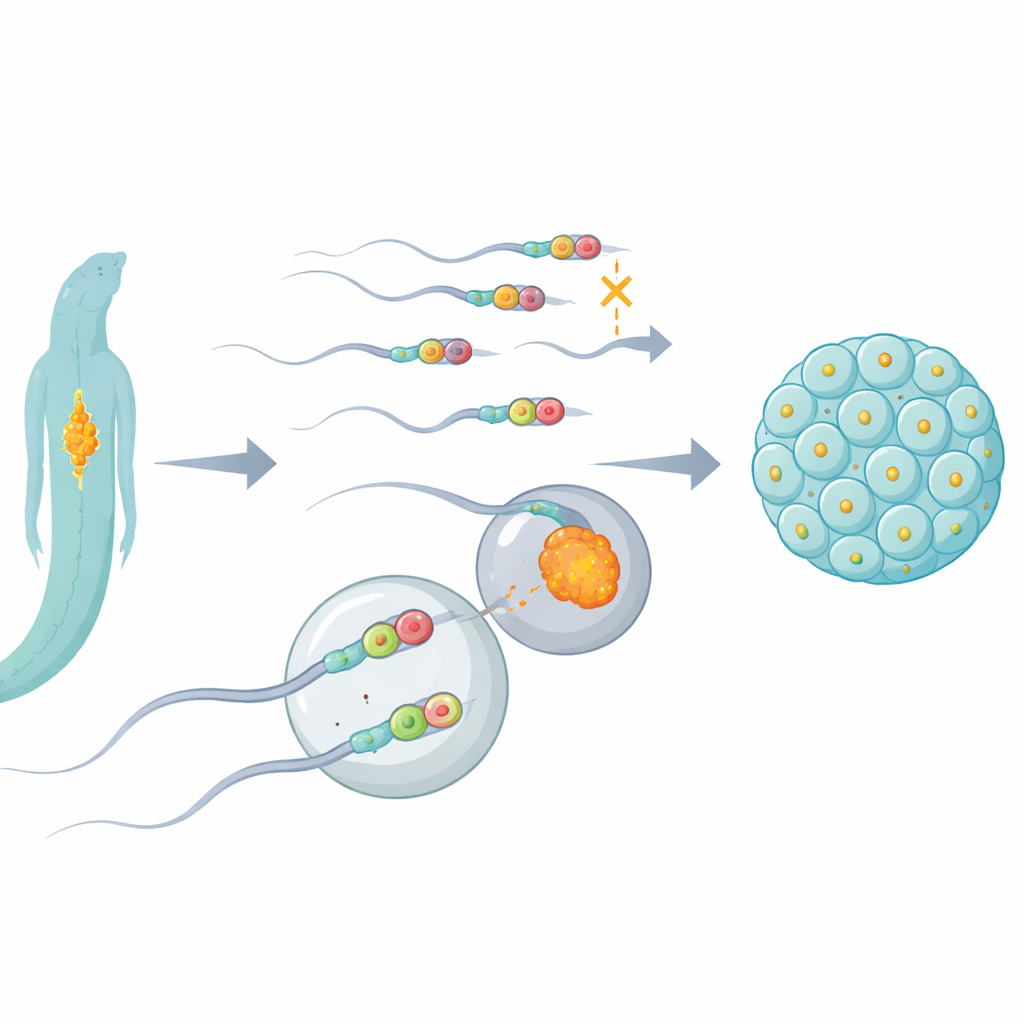
Every human, worm, and most other animals inherit their cellular power plants—mitochondria—almost entirely from their mothers. Fathers’ mitochondria enter the egg with the sperm but are swiftly removed, a biological cleanup that helps prevent damaged mitochondrial DNA from being passed on. This study in tiny roundworms uncovers a molecular “stress checkpoint” that helps decide whether paternal mitochondria are efficiently cleared, offering clues that may one day relate to certain infertility and mitochondrial diseases.
The power factories we inherit from mom
Mitochondria generate the energy that keeps cells alive, and they carry their own small loop of DNA. Over a lifetime, this mitochondrial DNA can accumulate harmful mutations. If both parents routinely passed on their mitochondria, these defects could mix and build up across generations. To avoid this, most animals rely on maternal inheritance: the embryo keeps the mother’s mitochondria and eliminates almost all of the father’s soon after fertilization. Although several disposal routes are known—such as cellular recycling systems and DNA-cutting enzymes—scientists have not fully understood how subtle chemical marks on DNA and RNA might influence this paternal mitochondrial elimination.
A molecular eraser with a key reproductive job
The researchers focused on a protein called ALKB-1 in the nematode Caenorhabditis elegans, a workhorse of genetics. ALKB-1 is a kind of molecular eraser that can remove methyl groups—tiny chemical tags—from DNA and RNA. By selectively disabling ALKB-1 or reducing its levels, the team tracked what happened to sperm mitochondria and their DNA after fertilization. They found that when ALKB-1’s chemical “eraser” activity was lost, paternal mitochondria and their DNA lingered abnormally long in developing embryos, instead of being rapidly cleared. This delay occurred mainly when ALKB-1 was missing in sperm, and went hand-in-hand with sperm that contained unusually high numbers of mitochondria and mitochondrial DNA.
How tinkering with tRNA chemistry upsets mitochondria
Digging deeper, the scientists asked which chemical marks ALKB-1 really matters for. Tests on DNA suggested that changes in DNA methylation alone could not explain the clearance problem. Instead, attention turned to tRNAs, small adapter molecules that help translate genetic messages into proteins. In normal worms, ALKB-1 removes a specific mark—called m1A—from a subset of tRNAs. Without ALKB-1, these marks piled up. This subtle shift in tRNA chemistry broadly altered how efficiently many proteins were made: protein production in the cell fluid increased, while protein synthesis inside mitochondria faltered. The result was a mismatch between proteins destined for mitochondria and what the organelles could handle, leading to clumps of mismanaged proteins and signs of mitochondrial stress.
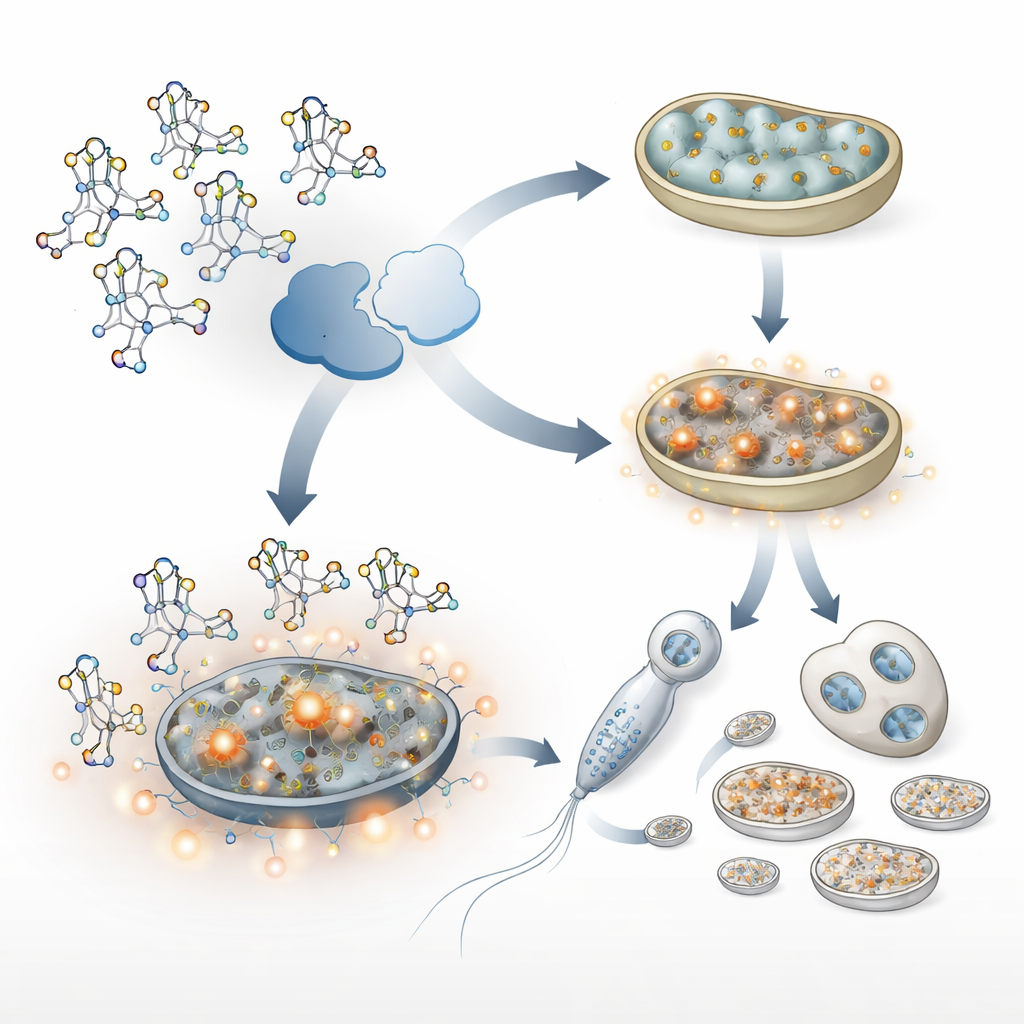
Stress signals that protect—and inadvertently preserve—paternal mitochondria
Stressed mitochondria often produce more reactive oxygen species, chemically aggressive byproducts of metabolism. In the ALKB-1–deficient worms, these reactive molecules rose sharply. That, in turn, switched on two key stress-response programs: one run by a factor similar to human Nrf2 (called SKN-1 in worms), and another known as the mitochondrial unfolded protein response. Together, these pathways boosted the production of new mitochondria and increased mitochondrial DNA copying, particularly in sperm. While this response helps cells cope with damage, it also had an unintended side effect: more paternal mitochondria and mitochondrial DNA accumulated and were harder to clear after fertilization. Blocking these stress pathways, or reducing oxidative damage with antioxidants, restored more normal removal of paternal mitochondria.
When cleanup fails, fertility and embryos suffer
The delayed removal of paternal mitochondria was not just a microscopic curiosity. Male worms lacking proper ALKB-1 activity sired fewer offspring, and the embryos they helped create died more often. Yet their sperm formed in normal numbers and looked structurally intact, suggesting that the main problem lay in mitochondrial quality control rather than in making sperm at all. The study proposes that ALKB-1–controlled tRNA marks act as an epigenetic checkpoint: when this system works, mitochondrial protein balance is maintained, stress remains in check, and paternal mitochondria are safely eliminated. When it fails, stressed and overabundant paternal mitochondria linger, harming fertility and early development. Although these experiments were done in worms, they illuminate a fundamental principle—that tiny chemical edits to RNA can steer how we inherit our cellular power plants, and may ultimately influence reproductive health in more complex animals, including humans.
Citation: Luo, Z., Li, Y., He, C. et al. ALKB-1-dependent tRNA methylation is required for efficient paternal mitochondrial elimination. Nat Commun 17, 2144 (2026). https://doi.org/10.1038/s41467-026-68813-6
Keywords: mitochondrial inheritance, paternal mitochondria, tRNA methylation, oxidative stress, male fertility