Clear Sky Science · en
A common 19 bp APOE enhancer deletion is protective against Alzheimer’s disease in African Americans
Why this discovery matters
Alzheimer’s disease robs memories and independence from millions of older adults, but it does not strike everyone with the same intensity. One of the strongest known genetic risk factors, a version of a gene called APOE, is especially dangerous in people of European or East Asian ancestry. Yet African Americans who carry this same high‑risk version do not develop Alzheimer’s as often or as early. This article uncovers a small stretch of missing DNA that helps explain why—and points to new ways of separating brain risk from heart and cholesterol problems.
A risky gene with a puzzling exception
Scientists have known for decades that a particular form of the APOE gene, called epsilon‑4, greatly increases the odds of developing late‑onset Alzheimer’s disease. People who inherit two copies of this version face some of the highest known genetic risk. At the same time, APOE also helps manage fats and cholesterol in the body, so changes in this gene influence both brain health and blood lipids. Curiously, African Americans and other people with African ancestry who carry two copies of APOE‑ε4 do not see the same sky‑high Alzheimer’s risk observed in people of European ancestry, suggesting that additional nearby DNA differences might buffer the danger.
Finding a tiny missing piece of DNA
The researchers used long‑read genome sequencing, a technology that reads long stretches of DNA at once, to examine the region around APOE in people from diverse backgrounds. They focused on insertions and deletions—places where DNA is added or missing—rather than on single‑letter changes that have already been heavily studied. In people of African ancestry, they identified a small 19‑base‑pair deletion located just beyond the end of the APOE gene, inside a regulatory element bound by a microglial transcription factor called SPI1. This deletion is fairly common in Africans and African Americans, often sitting on the same chromosome as APOE‑ε4, but it is almost absent in Europeans. That pattern made it a strong candidate for explaining the ancestry‑specific risk difference.
Evidence that the deletion protects the brain
To test whether this tiny deletion changes Alzheimer’s risk, the team analyzed genetic and clinical data from thousands of African American participants in the Alzheimer’s Disease Sequencing Project and the NIH All of Us program. After carefully adjusting for age, sex, ancestry, and APOE type, they found that carriers of the deletion had lower odds of Alzheimer’s disease than non‑carriers with the same APOE‑ε4 background. Among people with two APOE‑ε4 copies and local African ancestry at this region, those with the deletion developed Alzheimer’s roughly three years later, on average, than those without it. When all ancestries were pooled, the protective effect of this small deletion was comparable in strength to the well‑known protective APOE‑ε2 variant.
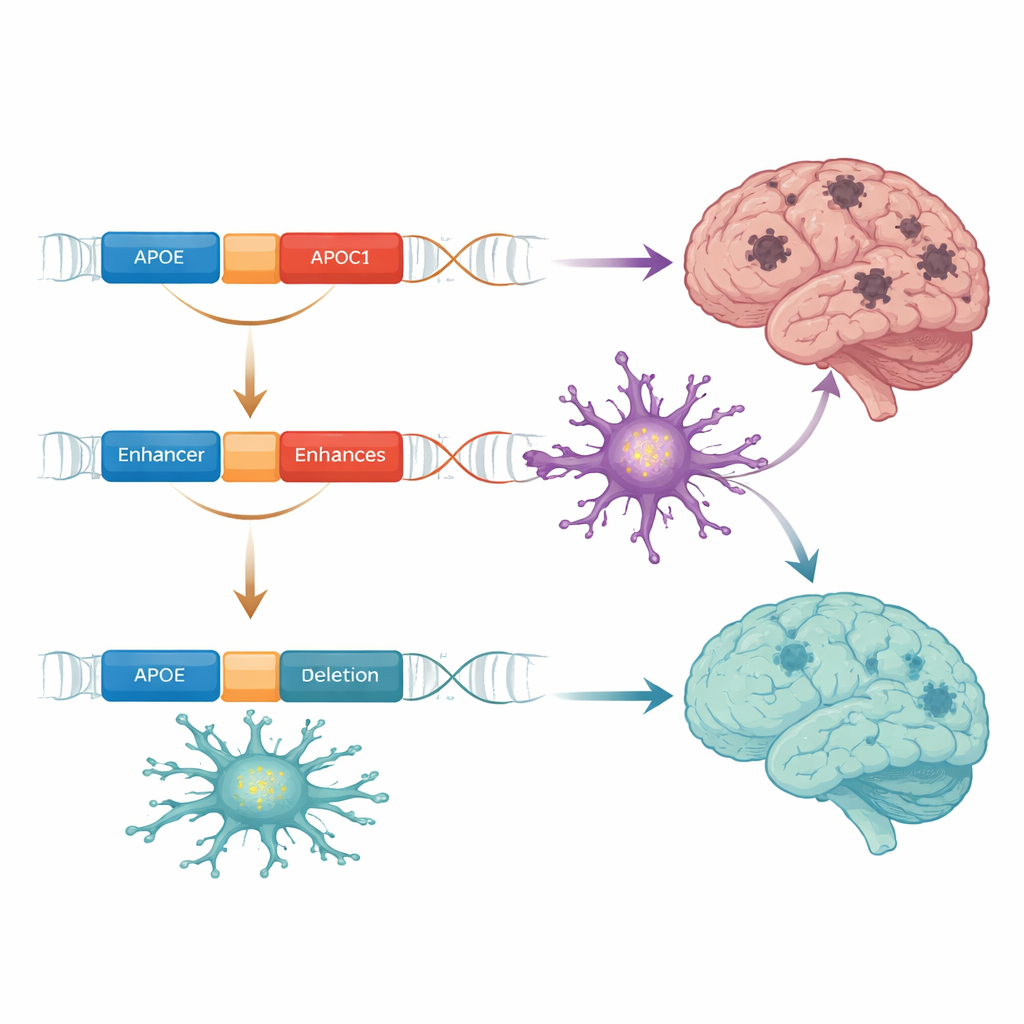
How the deletion may change brain cells and cholesterol
Next, the scientists asked how a tiny missing segment of DNA could have such an impact. They inserted the normal and deleted sequences into reporter constructs and introduced them into microglia‑like cells grown in the lab. The intact sequence, which includes the SPI1 binding site, acted as a brake on nearby gene activity—reducing the output of a linked reporter gene. When the 19‑base segment was missing, this repression disappeared. Further experiments showed that boosting SPI1 in these cells did not change APOE levels directly, but instead reduced expression of a neighboring gene called APOC1 and altered a long non‑coding RNA spanning the region between APOE and APOC1. The deletion therefore appears to reshape how this enhancer communicates with APOC1 and, indirectly, with APOE in microglia, the brain’s immune cells that help clear toxic proteins.
Untangling brain risk from blood fat levels
Because APOE also controls blood lipids, the researchers used a phenome‑wide association study to see how APOE variants and the deletion affect hundreds of health outcomes in the All of Us cohort. The high‑risk APOE‑ε4 variant increased both Alzheimer’s risk and rates of high cholesterol and high blood fats, while the APOE‑ε2 variant showed the opposite pattern. In contrast, the 19‑base deletion, once the effects of APOE type were accounted for, was linked to protection from Alzheimer’s and related dementias but had little independent effect on lipid traits. Other variants in the same intergenic region showed the reverse pattern—strong effects on cholesterol with little impact on brain disease. Together, these findings suggest that DNA between APOE and APOC1 can decouple neurological risk from cardiovascular and lipid effects.
What this means for future patients
In everyday terms, this study shows that some African Americans carry a small, naturally occurring DNA change that takes the edge off one of the most dangerous Alzheimer’s risk genes. By weakening a repressive switch that links APOE to its neighbor APOC1 in microglia, the 19‑base deletion appears to delay or reduce disease, even though it sits on an otherwise high‑risk APOE‑ε4 background. Understanding this protective mechanism may help researchers design treatments that mimic its effect—lowering Alzheimer’s risk without worsening cholesterol problems—and highlights why including diverse populations in genetic research is crucial for finding both risk‑raising and risk‑reducing variants.
Citation: Brutman, J.N., Busald, T., Nizamis, E. et al. A common 19 bp APOE enhancer deletion is protective against Alzheimer’s disease in African Americans. Nat Commun 17, 2237 (2026). https://doi.org/10.1038/s41467-026-68808-3
Keywords: Alzheimer’s disease, APOE gene, African ancestry, genetic protection, microglia