Clear Sky Science · en
Cellular and transcriptional trajectories of neural fate specification in sea anemone uncover two modes of adult neurogenesis
How a simple sea creature keeps making new brain cells
Most animals, including humans, make most of their neurons early in life and then largely stop. Yet some seemingly simple creatures, such as sea anemones, continue to add new nerve cells throughout adulthood. This study explores how the starlet sea anemone (Nematostella vectensis) constantly renews and reshapes its diffuse nervous system, revealing strategies that may echo ancient solutions to growing and repairing brains.
A growing body needs a growing nerve net
Sea anemones do not have a brain; instead, they have a web-like nerve net spread through their body wall. As these animals grow or shrink depending on food availability, their nerve net must scale up or down to match. Using a light-sensitive fluorescent protein as a time stamp, the authors marked existing neurons in juvenile adults and then watched for newly born cells that appeared over the next week. They found abundant new neurons scattered along the body axis, not confined to a special growth zone. These fresh cells appeared in both the mouth-facing and opposite ends of the animal, showing that adult neurogenesis is spatially widespread and ongoing.
Tracing the origins of new nerve cells
To discover where these new neurons come from, the researchers combined live imaging with single-cell RNA sequencing, a technique that reads which genes are active in thousands of individual cells. They focused on cells carrying fluorescent “reporters” controlled by three key genes: FoxL2, SoxC and Elav. FoxL2 marks a broad pool of dividing, multipotent progenitor cells; SoxC is switched on briefly as cells start to specialize; and Elav is strongly active in peptidergic neurons, which communicate mainly with small signaling peptides. The team showed that a population of FoxL2-positive, Piwi1-positive progenitor cells behaves like stem cells: they self-renew while continuously generating diverse descendants, including neurons, stinging cells called cnidocytes, and secretory cells. SoxC and Elav, in contrast, light up only as cells leave this progenitor pool and commit to particular neural fates.
Two different paths to adult neurons
By aligning the reporter timings with each cell’s gene expression signature, the study uncovered two distinct routes to mature neural cells. Peptidergic neurons follow a direct path: they arise from progenitors that already bear a “peptidergic-committed” molecular profile and then quickly differentiate into a variety of neuron subtypes. New members of each subtype appear in proportion to how common that subtype already is, suggesting that the system maintains a balanced mix of neuron types rather than favoring particular ones. In this lineage, there is no clear series of intermediate waystations; instead, closely related neuron subtypes emerge in parallel from similarly primed progenitor cells. 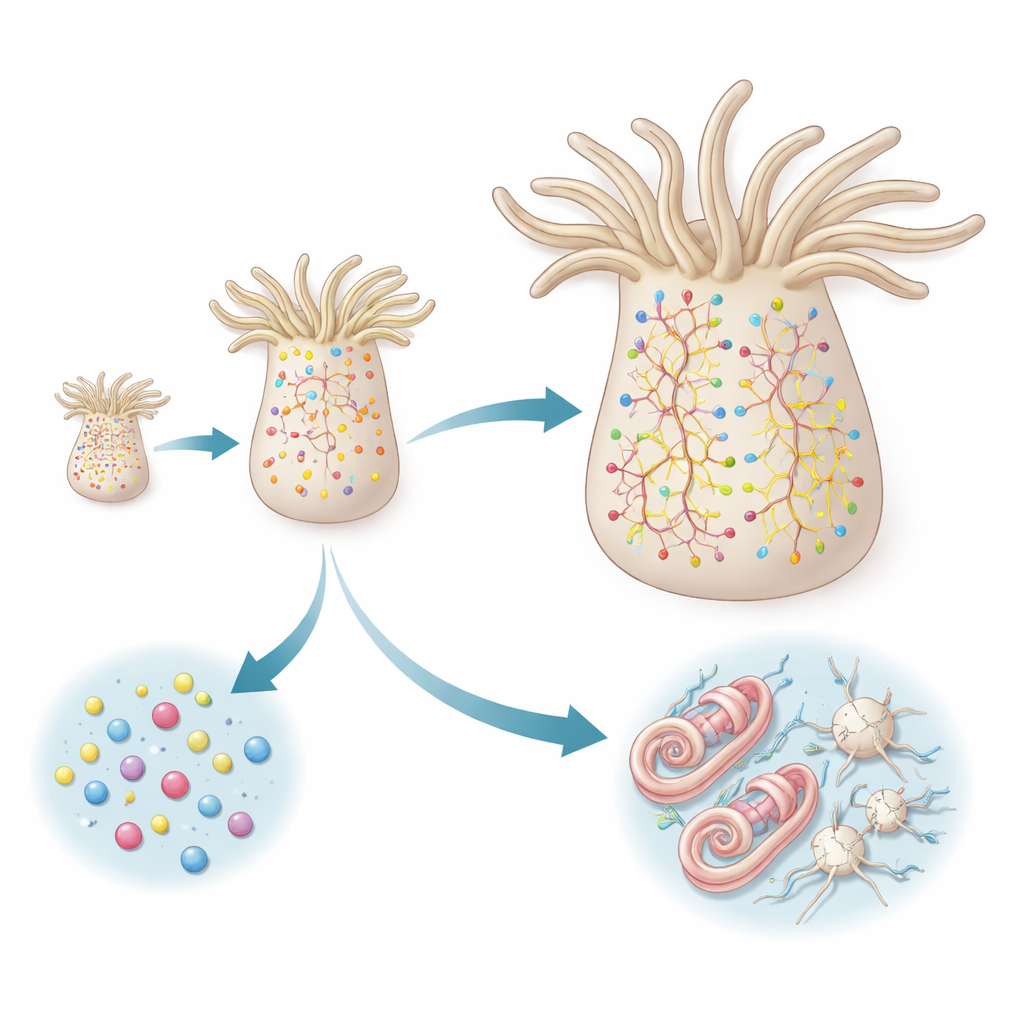
The special case of stinging cells
Cnidocytes, the stinging cells unique to cnidarians, take a more stepwise route. They first pass through a “capsule-building” phase, during which they construct their characteristic pressurized organelles that can fire a microscopic harpoon. This stage relies on a specific set of regulatory genes and structural proteins that are mostly absent from other cell types. Only after this intermediate program is completed do cnidocytes switch on a second, more conventional neural program, adding ion channels and other components needed for electrical signaling. Genetic data from mutants that stall at the early stage reinforce this two-phase model: blocking the transition prevents the later neural features from appearing. 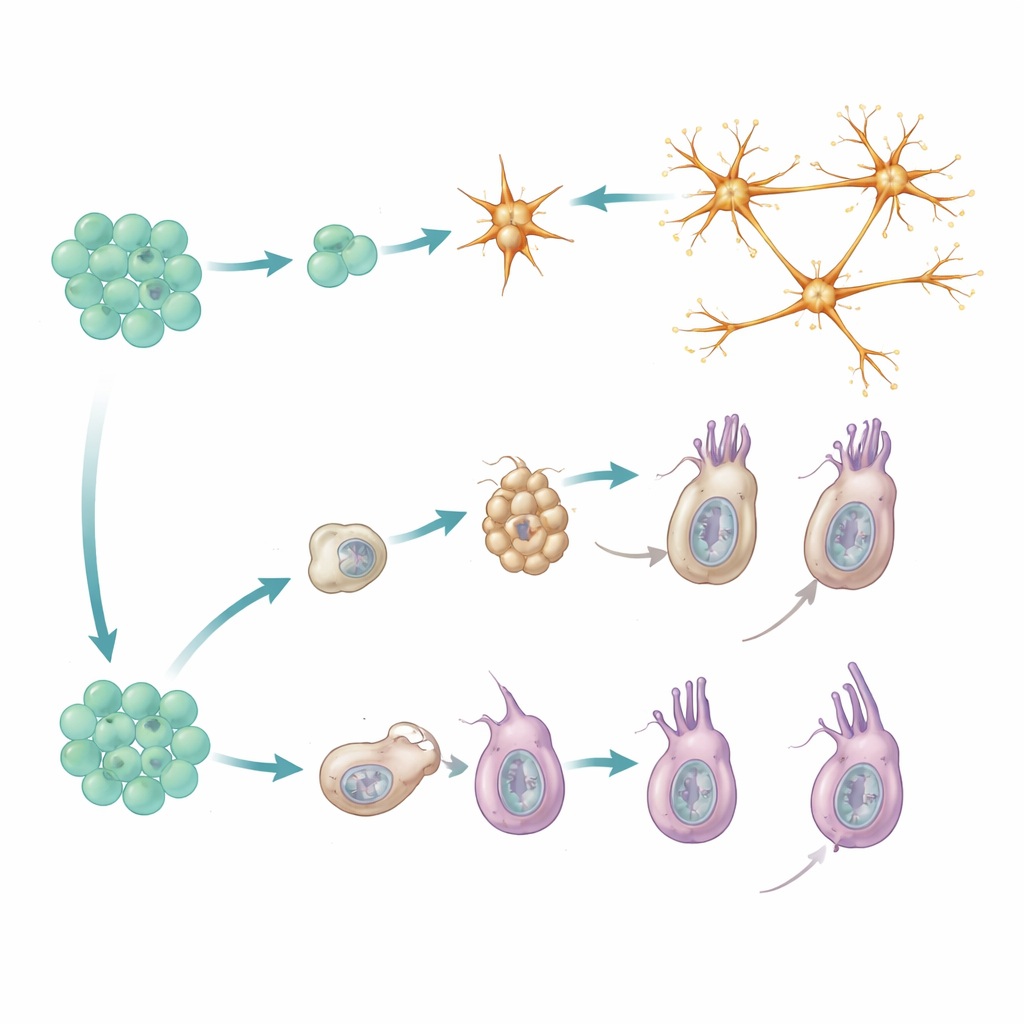
Shared rules and ancient roots
Despite their different routes, peptidergic neurons and cnidocytes rely on overlapping families of regulatory genes to lock in their identities. Broad neural features are associated with bHLH and bZIP transcription factors, while the fine-grained identity of each neuron subtype is specified by unique combinations of homeodomain and zinc-finger genes—a “code” reminiscent of how neuron types are defined in more complex animals, including worms and vertebrates. The authors argue that these modular strategies—stem-like progenitors, transient SoxC activity during commitment, and subtype-defining homeodomain codes—may represent deeply conserved principles of neurogenesis. In sea anemones, they support lifelong renewal and scaling of the nerve net; in other animals, related mechanisms may underlie both limited adult neurogenesis and the remarkable regenerative abilities seen in some lineages.
Citation: Plessier, F., Marlow, H. Cellular and transcriptional trajectories of neural fate specification in sea anemone uncover two modes of adult neurogenesis. Nat Commun 17, 2611 (2026). https://doi.org/10.1038/s41467-026-68802-9
Keywords: adult neurogenesis, sea anemone, neural progenitor cells, cnidocytes, single-cell transcriptomics