Clear Sky Science · en
Comparative multi-omic analysis reveals conserved and derived mechanisms of fin and limb regeneration
Why growing back lost body parts matters
Many animals can regrow lost body parts, from lizard tails to salamander limbs. Understanding how they do this is more than a curiosity: the same rules that let a fish rebuild a fin or a salamander replace a limb could one day inform new treatments for severe injuries in people. This study compares different animals that are unusually good at regeneration, looking deep inside their cells to discover which repair tricks are ancient and shared, and which are newer inventions.
Different animals, different limbs, same big question
The researchers focused on three species: the axolotl salamander, which can regrow full limbs; the common zebrafish, famous for its fin repair; and Polypterus, a primitive ray-finned fish that can regenerate not just the thin outer fin rays but the entire fin, including internal bones and muscles. By comparing these animals, the team asked whether there is a common “toolkit” for rebuilding complex body parts that dates back to early vertebrate evolution. They used modern genomic methods that read which genes are active in thousands of individual cells and map where those cells sit inside the tissue.
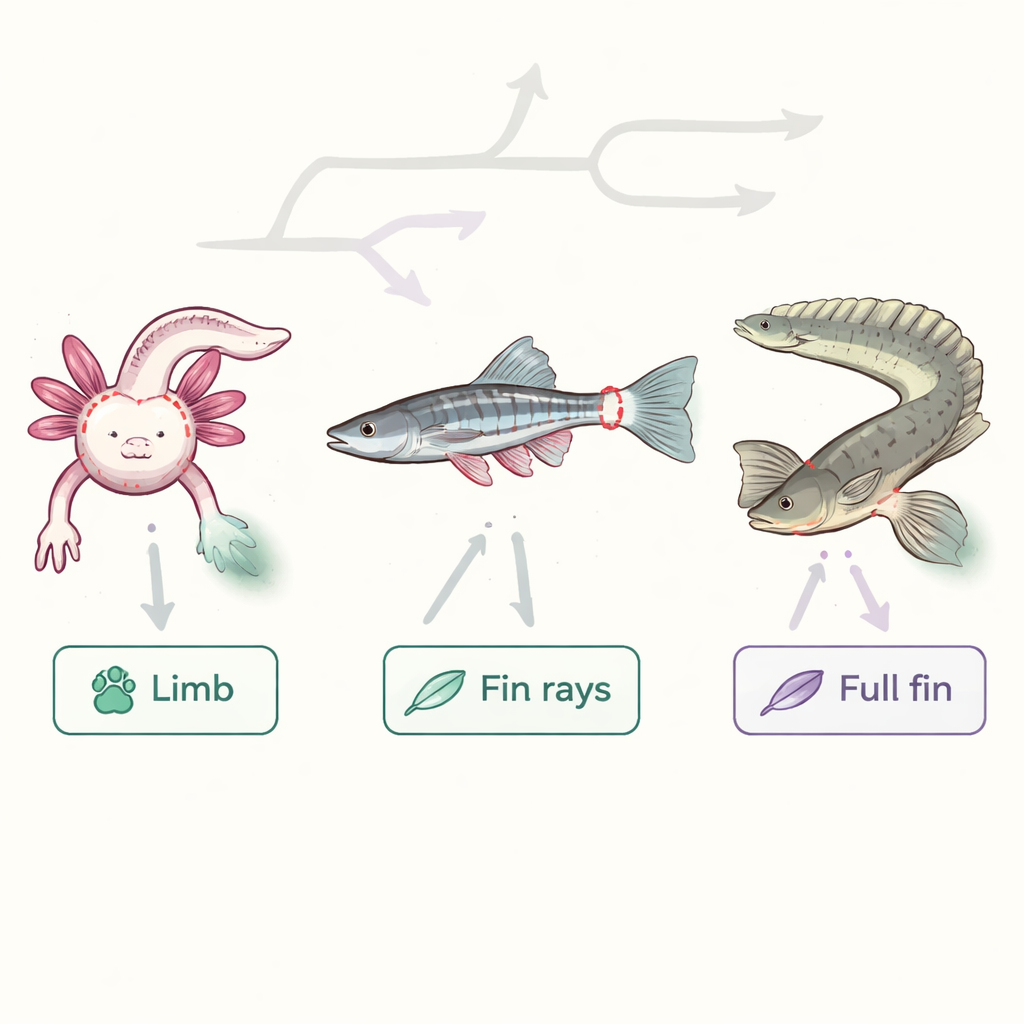
Mapping the cells of a regrowing fin
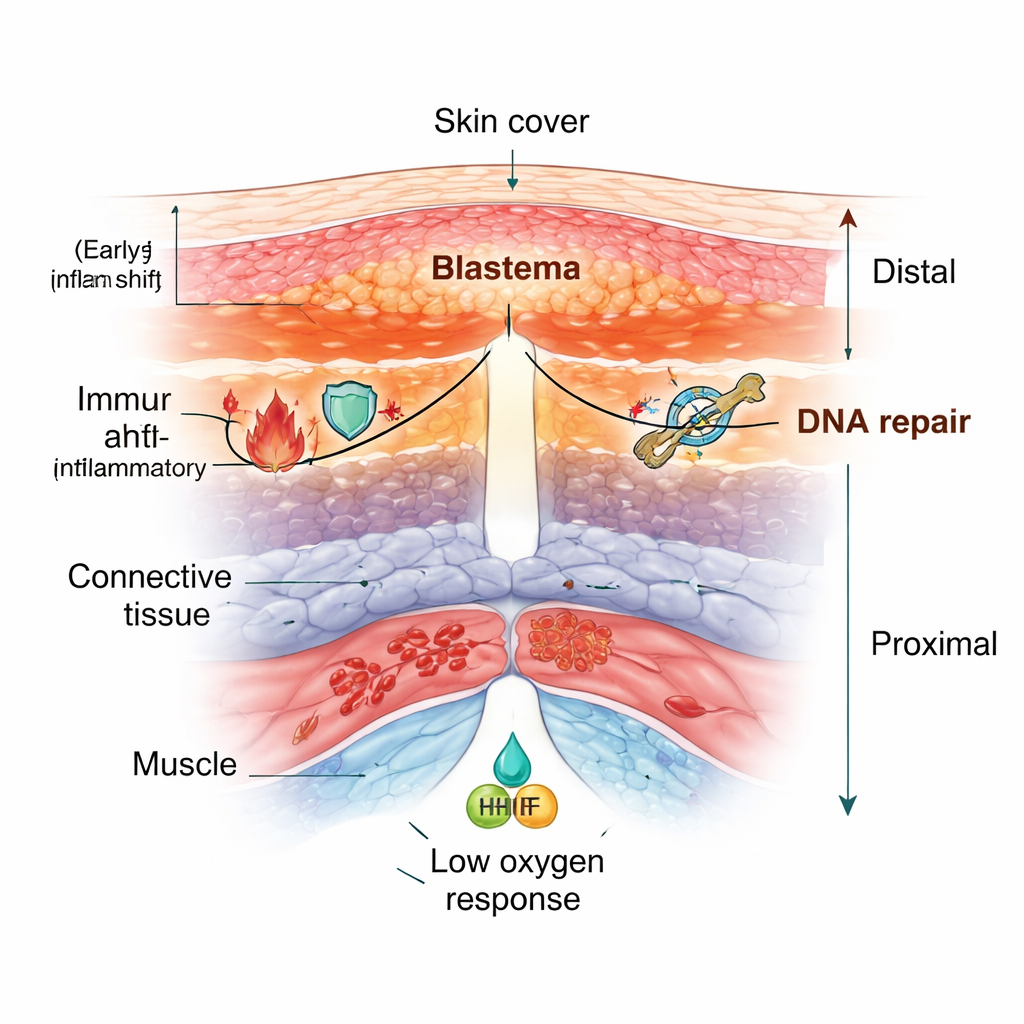
In Polypterus, the scientists sampled fins before injury and at several days after amputation. They found more than thirty distinct cell groups, including different layers of skin, immune cells, blood vessels, muscle, connective tissue, and dividing “blastema” cells—the mass of cells that drives new growth. As the fin healed, quiet adult tissues gave way to an active repair zone: immune cells flooded in, skin thickened into a specialized wound covering, and connective tissue cells streamed toward the cut site to build the blastema. Similar patterns were seen when they examined axolotl limbs and zebrafish fins, showing that this reshuffling of cell types is a shared feature of appendage regeneration.
Old building plans and new twists
Closer inspection revealed that the growing tip is not uniform. In both Polypterus fins and axolotl limbs, the connective tissue beneath the wound skin split into two zones along the length of the limb: a distal region near the tip rich in fast-dividing, matrix-producing fibroblasts, and a more proximal region closer to the body with cells more like stabilizing, contractile support cells. The skin over the wound also reactivated a genetic program normally used in embryos to build the “apical ectodermal ridge,” a signaling strip crucial for limb outgrowth. That program appeared both in the wound skin and in nearby connective tissue, suggesting that adult regeneration reuses ancient developmental instructions but distributes them across several tissues.
Stress signals, oxygen control, and the immune switch
Across species, injured fins and limbs showed strong activation of DNA damage and repair genes, as if cells were checking and fixing their genomes before entering the intense growth phase. The immune response also followed a similar script: an early wave of pro-inflammatory signals helped clear damaged tissue, followed by a rise in anti-inflammatory signals that favored tissue rebuilding instead of scarring. Another shared theme was a “low oxygen” response. Cells stabilized hypoxia-sensitive factors and boosted genes that support glycolysis, a metabolic pathway that works even when oxygen is scarce. In Polypterus and axolotl, there was also a striking expansion of red blood cells near the injury that carried a special oxygen-sensing gene variant, hinting that blood cells may help tune the healing environment. In Polypterus and zebrafish, even the wound skin turned on a myoglobin gene—usually found in muscle—that may help buffer oxygen and harmful reactive molecules during regrowth.
Control switches in the genome
To find the DNA switches that turn regeneration genes on and off, the team measured which parts of the genome opened up after fin injury in Polypterus. Hundreds of regions became more accessible, many lying near genes already known to be active in the wound skin and blastema. These regions were enriched for binding sites of AP-1 transcription factors, proteins that act as master switches for gene networks. Similar factors have been implicated in zebrafish and axolotl regeneration, suggesting that a conserved regulatory logic operates across very different animals and appendages.
What this means for future healing
For a general reader, the key message is that fin and limb regeneration are not magical one-offs; they rely on a shared set of cellular players and genetic circuits that evolved long ago. Highly regenerative animals combine this ancient toolkit with species-specific tweaks—such as extra myoglobin genes or unusual blood cell behaviors—to fine-tune repair. By charting these common and unique strategies, the study brings us closer to understanding why some vertebrates can regrow complex structures while others, including humans, cannot, and points to molecular pathways that might one day be harnessed to improve healing in our own bodies.
Citation: F. Sousa, J., Lima, G., Perez, L. et al. Comparative multi-omic analysis reveals conserved and derived mechanisms of fin and limb regeneration. Nat Commun 17, 1922 (2026). https://doi.org/10.1038/s41467-026-68801-w
Keywords: limb regeneration, fin regeneration, wound healing, stem cells, evolution