Clear Sky Science · en
Chemistry-driven autonomous nanopore membranes
Why tiny, self-adjusting holes matter
Every cell in your body relies on tiny gateways that open and close to let specific ions pass, controlling everything from nerve impulses to muscle motion. Engineers have long wanted to build artificial versions of these ion channels using solid materials, but making and controlling such minute openings—just a few atoms wide—has been extremely difficult. This paper reports a way to let chemistry itself repeatedly build and erase ultra-small pores in a solid membrane, automatically and on demand, using only a simple voltage. The result is a man-made membrane whose nanopores "breathe"—opening and closing on their own—much like natural ion channels.
Turning a nanopore into a tiny chemical workshop
The researchers start with a silicon nitride (SiNx) membrane containing a single, lithographically defined nanopore about 100 nanometers wide. This pore connects two liquid compartments filled with different salt solutions. By applying a voltage across the membrane, ions are driven into the pore where they can react and form a solid layer of metal phosphate inside it. In one typical setup, manganese ions (Mn2+) on one side and phosphate ions on the other meet in the pore and precipitate as manganese phosphate, gradually plugging the opening. Reversing the voltage dissolves this solid back into the solution, reopening the pore. In electrical measurements, this shows up as a strong diode-like behavior: current flows easily in one voltage direction but is almost blocked in the other, and this behavior remains highly stable over hundreds of cycles. 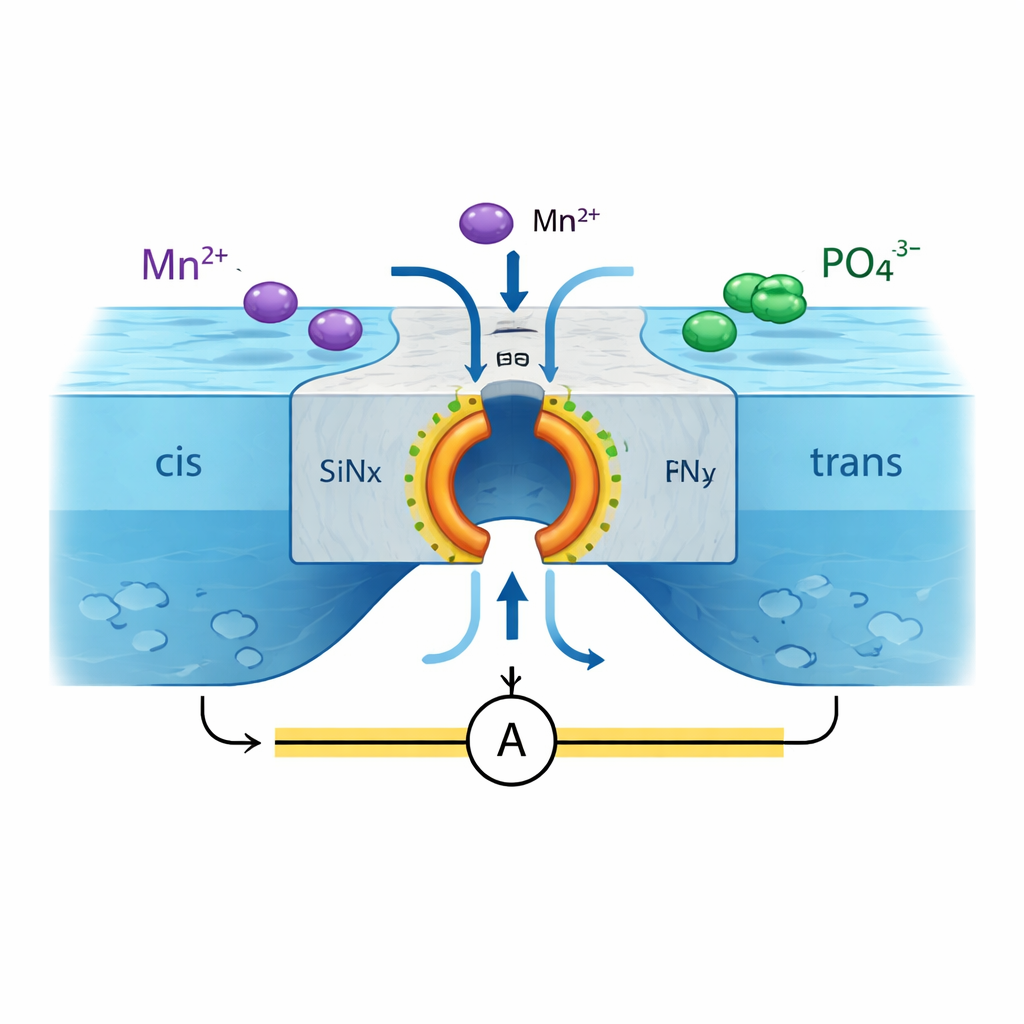
Self-driven pore opening and closing
Once the nanopore is coated with this reactive layer, something remarkable happens under a constant voltage. Rather than staying either fully open or fully blocked, the membrane begins to “breathe.” The phosphate film completely seals the larger nanopore, so almost no current flows. Then, as parts of the film slowly dissolve, a tiny sub-nanometer hole suddenly pierces the layer, letting ions rush through and causing a sharp spike in current. The electric field in that small opening then accelerates local precipitation, which reclogs the hole and brings the current back down. This cycle—dissolve, pierce, reprecipitate—repeats on its own, creating a sequence of current spikes that closely resemble the spontaneous firing of biological ion channels.
Using chemistry to tune behavior
The team shows that the character of this breathing can be controlled by changing the ions and acidity in the surrounding solutions. Different metal ions such as magnesium, calcium, manganese, or aluminum form phosphate layers that dissolve and re-form at very different rates. Some leave the pore mostly open, others seal it permanently, and a few create complex bursting patterns where many small spikes lead up to occasional giant surges in current when the film ruptures. The acidity (pH) also matters: more acidic conditions favor dissolution and allow larger pores to open, while less acidic conditions encourage faster replugging and smaller pores. By carefully tuning pH, the researchers can adjust the average pore diameter from about 2 to 7 nanometers with sub-nanometer precision, all without physically machining the membrane.
Ion traffic at the edge of the possible
Because the pores created in the film are so small—approaching the size of individual dehydrated ions—the way ions move through them carries signatures of extreme confinement. The authors test different negatively charged ions that carry water shells of different thickness, such as fluoride, chloride, and iodide. Fluoride, which is small and tightly wrapped in water, can still slip through the tiniest pores once its hydration shell is partly stripped away, leading to distinct, voltage-dependent current peaks that indicate a well-defined pore size of around 0.4 nanometers. Larger ions like iodide are partially excluded and even produce negative-going pulses when they momentarily block the entrance. By creating arrays of larger “parent” nanopores that each host many of these transient sub-nanometer pathways, the team can gather vast statistics on such events and tease out the subtle physics of ion dehydration and crowding. 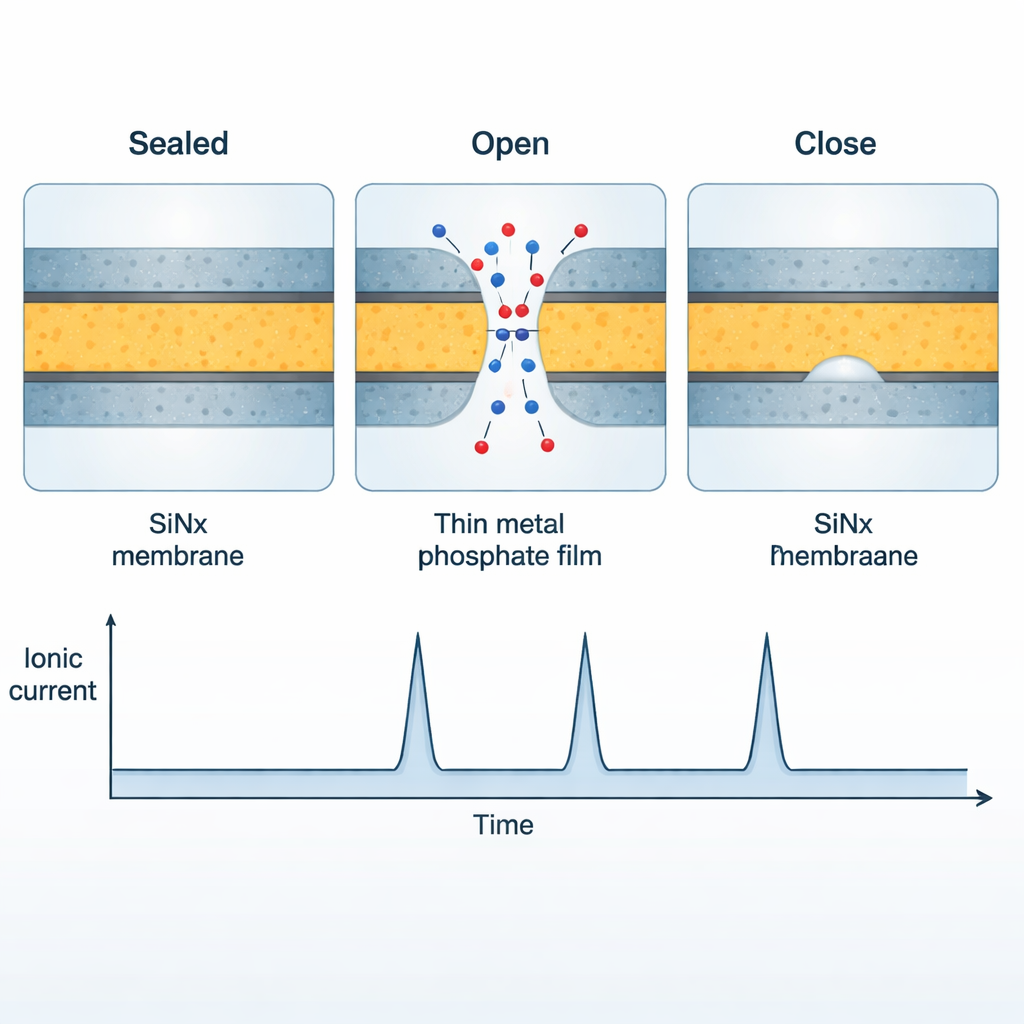
From artificial ion channels to future devices
In essence, the authors have developed a "chemically controlled break-membrane" method: instead of carving atomically precise pores once and for all, they let reversible reactions repeatedly build and remove them inside a larger template pore. Although the exact shapes of these tiny channels cannot yet be imaged directly, electrical data strongly suggest that ions are traveling through conduits only slightly wider than the ions themselves. This offers a powerful new way to study how fluids and ions behave when squeezed to almost unimaginable smallness, potentially improving technologies such as single-molecule sensing, ion-based information processing, and nanoscale chemical reactors. For non-specialists, the key message is that we are learning to harness simple chemistry and voltage to give solid membranes a life-like ability to open and close their own molecular doorways—bringing artificial ion channels a step closer to reality.
Citation: Tsutsui, M., Hsu, WL., Garoli, D. et al. Chemistry-driven autonomous nanopore membranes. Nat Commun 17, 1496 (2026). https://doi.org/10.1038/s41467-026-68800-x
Keywords: nanopores, ion transport, nanofluidics, solid-state membranes, single-molecule sensing