Clear Sky Science · en
YL1004 is a SARS-CoV-2 papain-like protease inhibitor with immunomodulatory and antiviral activity in mice
Why a new COVID-19 pill still matters
The world has vaccines and several antiviral pills for COVID-19, yet the coronavirus keeps evolving, dodging immunity and developing resistance to existing drugs. This study introduces YL1004, an experimental oral medicine that targets a different weak spot in SARS-CoV-2 and also helps the body’s own defenses fight back. In mice, it not only cut virus levels from multiple variants, including a drug‑resistant strain, but also prevented death from otherwise lethal infections.
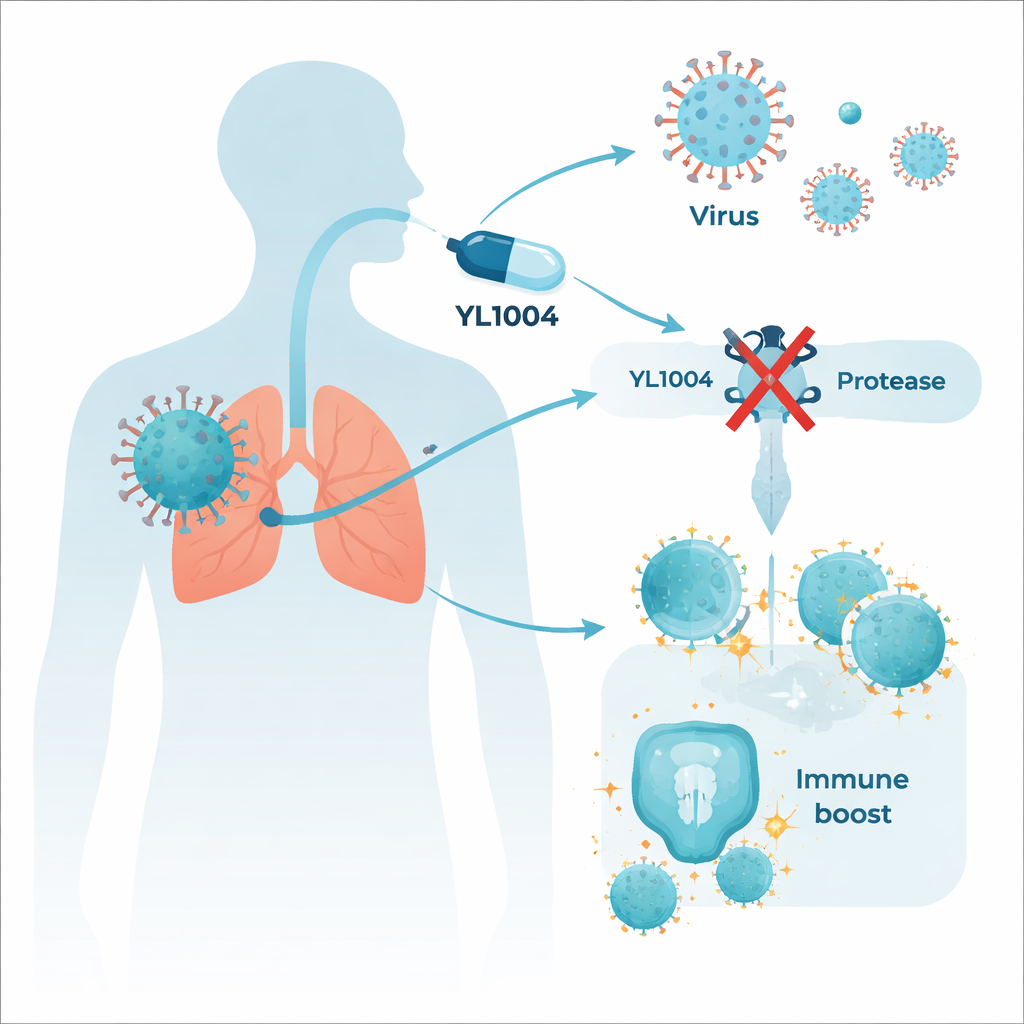
A viral tool with a double role
YL1004 is designed to block a viral enzyme called papain-like protease, or PLpro. This enzyme acts like a molecular scissors: it cuts long viral proteins into working parts the virus needs to copy itself. But PLpro has a second, more devious job. It removes small chemical tags from our own immune proteins, effectively muffling alarm signals that would normally call immune cells into action. Because of this dual role, shutting down PLpro can both slow virus replication and restore early antiviral defenses.
Designing a precise molecular lock
The researchers began by screening about 35,000 compounds for their ability to interfere with PLpro in a test-tube assay. They found a promising “tricyclic” chemical shape and then systematically refined it, guided by comparisons with older PLpro blockers. Dozens of variations were made, tuned, and tested for potency, safety in cells, and how they behaved in animals. YL1004 emerged as the best candidate: it strongly inhibited PLpro at very low concentrations, showed low toxicity in several human and monkey cell types, and had favorable “drug-like” features such as good stability in liver tests, decent solubility, and the ability to cross cell layers efficiently.
Seeing how the drug grips the virus
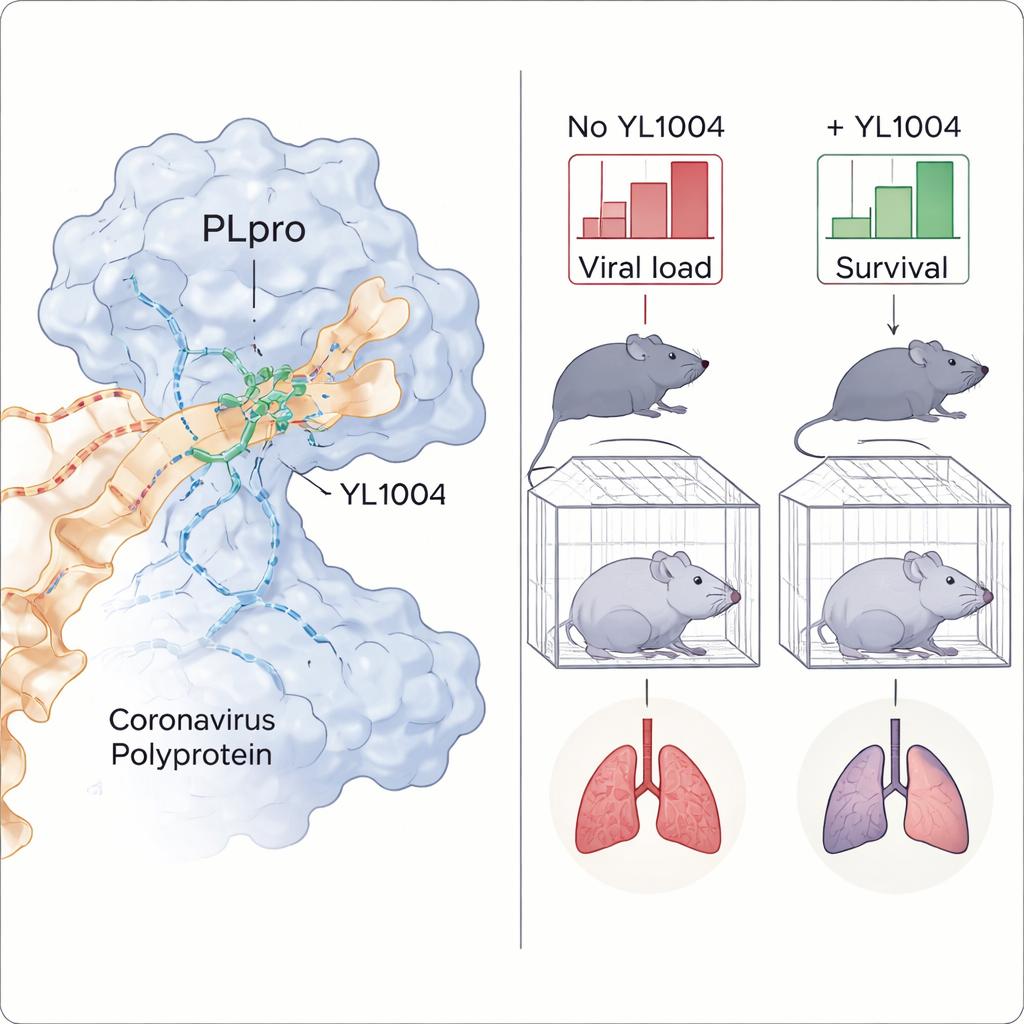
To understand why YL1004 works so well, the team solved a 3D crystal structure of the drug bound to PLpro. The images revealed that YL1004 nestles into a pocket near a flexible loop on the enzyme surface and forms multiple tight contacts. Parts of the molecule form hydrogen bonds—specific directional attractions—with several amino acids, while other ring-shaped regions create a broad hydrophobic, or water‑repelling, interface that locks the drug in place. Compared with a classic PLpro inhibitor, YL1004 makes more and stronger contacts, explaining its enhanced grip. Structural comparisons with similar human enzymes also showed why YL1004 spares them, reducing the chance of off‑target side effects.
Boosting antiviral defenses and beating variants
In cells, YL1004 reversed the enzyme’s ability to strip regulatory tags from immune signaling proteins, thereby restoring key warning pathways such as interferon signaling and NF-κB, which are central to early antiviral defense. Genome‑wide RNA analyses confirmed that PLpro expression normally suppresses many immunity‑related and stress‑response genes, while adding YL1004 turned these pathways back on. When the drug was tested against live virus in human lung‑derived cells, it sharply reduced both viral genetic material and infectious particles from the original SARS‑CoV‑2 strain and several major variants, including Delta and Omicron sublineages JN.1 and KP.3. Notably, it remained active against a virus engineered to resist nirmatrelvir, the main component of the antiviral pill Paxlovid.
Protecting infected mice from lethal disease
The most striking results came from experiments in genetically engineered mice that are highly vulnerable to SARS‑CoV‑2. After oral dosing, YL1004 reached blood levels high enough, and for long enough, to match or exceed those needed to block the virus in cell culture. When mice were infected with an Omicron JN.1 strain, treatment significantly lowered virus counts in the nose and lungs and reduced tissue damage and viral protein staining in lung sections. In a tougher test, mice challenged with a lethal dose of the Delta variant were treated with either YL1004 or a comparison PLpro inhibitor. All animals receiving YL1004 survived with milder weight loss, whereas most untreated mice died, and the older compound performed less well.
What this could mean for future COVID-19 care
To a non-specialist, the takeaway is that YL1004 is a next‑generation experimental COVID‑19 pill that strikes the virus from two angles at once: it blocks a key enzyme the virus needs to reproduce and lifts the brake the virus puts on our innate immune system. In laboratory and mouse studies, it worked across multiple variants, including one that sidesteps an existing oral drug, and showed encouraging safety and dosing properties. While human trials are still needed, YL1004 and similar PLpro inhibitors could become valuable additions to our toolkit—especially for high‑risk patients and in the face of future variants that erode the effectiveness of current treatments.
Citation: Nan, J., Shuai, H., Qiao, J. et al. YL1004 is a SARS-CoV-2 papain-like protease inhibitor with immunomodulatory and antiviral activity in mice. Nat Commun 17, 2035 (2026). https://doi.org/10.1038/s41467-026-68795-5
Keywords: SARS-CoV-2, COVID-19 antivirals, papain-like protease, immune modulation, drug resistance