Clear Sky Science · en
Delivery of peptide coacervates to form stable interaction hubs in cells
Building New “Workstations” Inside Cells
Our cells are packed with tiny workspaces where important jobs—like energy production or stress responses—take place. Over time, or in disease, these natural workstations can falter. This study explores a way to add brand‑new “interaction hubs” into living cells using simple, lab‑made droplets built from short peptides. These synthetic hubs can capture specific proteins, concentrate them, and even help destroy them, suggesting new strategies for future therapies and cell engineering.
Why Cells Need Custom Workspaces
Cells keep order by separating different tasks into distinct compartments. Some are wrapped in membranes, like mitochondria; others are softer, droplet‑like structures that form when certain proteins and RNAs cluster together. These fluid droplets, or condensates, act as reaction centers that speed up or shut down particular biochemical pathways. Scientists have previously engineered such compartments by forcing cells to make special scaffold proteins from inserted genes. While powerful, that method requires gene delivery and limits how much material the cell can produce. The authors of this study set out to bypass DNA entirely and instead build ready‑made compartments outside the cell, then deliver them directly as stable, micron‑sized droplets.
Peptide Droplets That Form Stable Hubs
The team worked with short, disordered peptides known as HBpep and a redox‑responsive variant, HBpep‑SA. Under mildly acidic conditions these peptides remain dissolved, but when the pH is shifted to near‑physiological levels—or when the temperature is lowered—they phase‑separate into soft, gel‑like droplets about 1–5 micrometers across. By tuning peptide concentration, the researchers could control both droplet size and number. Using fluorescence techniques, they showed that peptide molecules inside these droplets move slowly, indicating a gel state that is sturdy enough to survive dilution and handling. When added to cultures of human cancer cells, mouse melanoma cells, and primary human immune cells, the droplets were taken up efficiently and accumulated in the cytoplasm. Larger droplets, in particular, remained intact for at least five days, effectively acting as long‑lived synthetic organelles inside living cells.
Loading and Aiming the Hubs
For these hubs to be useful, they must hold and organize cargo proteins. The researchers first improved loading by attaching a short HBpep‑derived tag to a model protein, GFP. This tag encouraged GFP to partition strongly into the droplets and, interestingly, to concentrate near their surfaces, creating a core‑shell organization. Next, they embedded nanobodies—compact antibody‑like proteins that bind tightly to chosen targets—inside the droplets. A GFP‑binding nanobody allowed the hubs to selectively capture GFP both in test tubes and inside cells. Because the peptide gel is relatively dense, initially most of the captured GFP accumulated at the droplet surface. But when the internal structure was partially loosened, either by changing redox conditions or by helping droplets escape their surrounding endosomal membranes, GFP also began to penetrate the interior. In cells, chemical helpers that promote endosomal escape greatly increased the fraction of hubs that successfully recruited their targets.
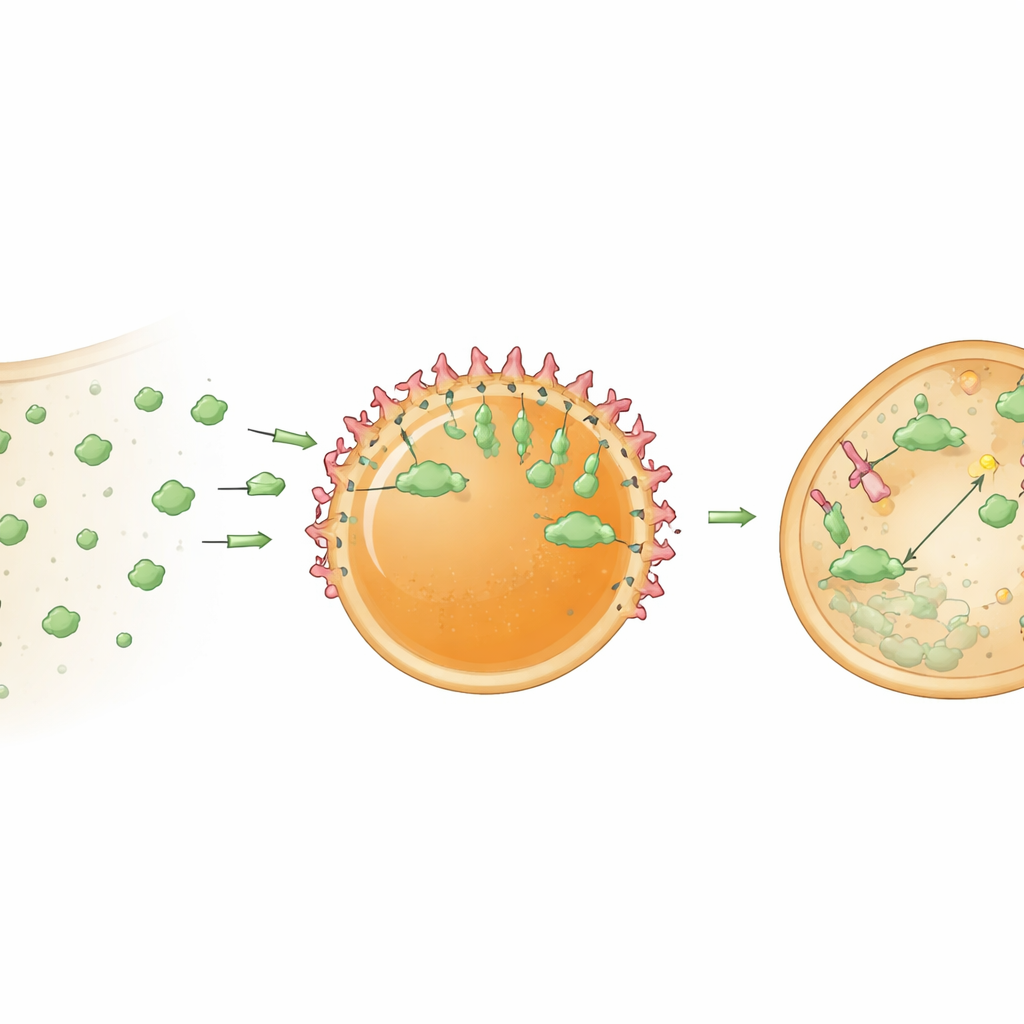
Turning Hubs into Protein Shredders
The authors then upgraded these hubs from passive traps into active processing centers. They loaded the droplets with a bioPROTAC—a fusion protein that combines a target‑binding nanobody with part of an enzyme adaptor that flags proteins for destruction by the cell’s waste‑disposal machinery. When these “degradosome” droplets were delivered into cells stably expressing GFP, cytosolic GFP levels dropped by about 78 percent within a day. Control hubs containing only the nanobody sequestered GFP but did not appreciably lower its overall level, confirming that the strong decrease stemmed from targeted degradation rather than simple capture. The findings suggest that clustering bioPROTACs inside a concentrated microenvironment makes them far more potent than when they are freely dispersed in the cytoplasm.
What This Could Mean for Future Therapies
In plain terms, this work shows that scientists can fabricate peptide‑based droplets in a test tube, pack them with custom protein tools, and then deliver them into living cells where they behave like new, long‑lasting organelles. These synthetic hubs can selectively pull in natural proteins and, when equipped with degradation machinery, can actively remove chosen targets from the cell. Because this platform avoids altering the cell’s genes and is modular in what it can carry, it opens a path toward therapies that restore or rewrite cellular behavior—such as clearing harmful proteins or rewiring faulty signaling—by simply installing new, programmable “workstations” into our cells.
Citation: Tu, W., Theisen, R.Q., Jin, P. et al. Delivery of peptide coacervates to form stable interaction hubs in cells. Nat Commun 17, 2250 (2026). https://doi.org/10.1038/s41467-026-68793-7
Keywords: synthetic organelles, peptide coacervates, intracellular delivery, protein degradation, cell engineering