Clear Sky Science · en
A bacterial defense system targeting modified cytosine of phage genomic DNA
How Bacteria Outsmart Invading Viruses
Viruses that infect bacteria, called phages, are locked in a constant arms race with their microbial hosts. Many of these phages rewrite the chemical letters in their DNA to slip past bacterial defenses. This study uncovers a previously hidden bacterial counter-move: a protein system named CMoRE that can spot and destroy phage DNA carrying these special chemical tweaks. Beyond revealing a new twist in microbe–virus warfare, CMoRE could become a precise tool for detecting subtle DNA marks linked to human diseases.
A Hidden Tag on DNA Letters
Both phages and animals sometimes alter the basic DNA letter cytosine by adding small chemical groups to it. In many T-even phages, including the classic T4 that infects E. coli, cytosine is replaced with a modified version called 5-hydroxymethylcytosine (5hmC), which can then be further decorated to form 5-glucosyl-hydroxymethylcytosine (5ghmC). These changes help phages dodge common bacterial defenses that normally cut unmodified “foreign” DNA while sparing the bacterium’s own genome. In mammals, a related mark, 5hmC, is now seen as a key epigenetic signal involved in gene control, brain function, and cancers—but it is very rare and hard to measure accurately.
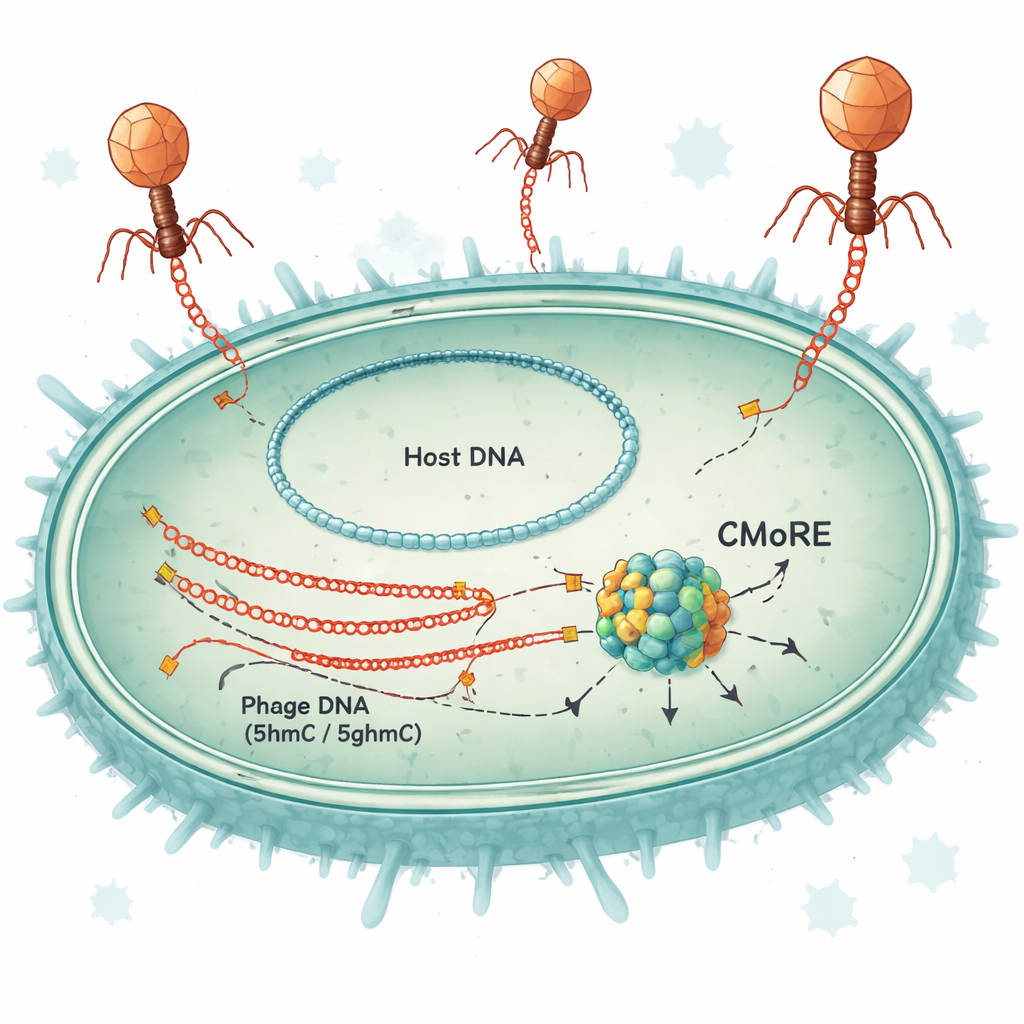
A Single-Protein Security System
The researchers studied a defense gene originally found in certain strains of E. coli and related bacteria. When they inserted this gene—now renamed CMoRE—into lab strains that normally lack it, the bacteria became almost completely resistant to several T-even phages, including T2, T4, and T6. Under heavy viral attack, cells carrying CMoRE continued to grow, showing that the system protects without sacrificing the host in a “suicide” response. Tests in liquid culture and on solid plates demonstrated that phage infection dropped by up to about one hundred thousand-fold, while bacteria without CMoRE remained vulnerable.
Precision Targeting of Modified Viral DNA
To see what CMoRE actually cuts, the team purified the protein and exposed it to DNA from different phages and from bacteria. CMoRE selectively chopped up DNA from T-even phages but left bacterial DNA essentially untouched. When they made test DNA fragments built with different versions of cytosine, CMoRE ignored normal cytosine and a common methylated form (5mC), yet efficiently degraded DNA containing 5hmC or 5ghmC. A mutant T4 phage whose genome used unmodified cytosine became completely resistant to the defense, confirming that the chemical modification—not a particular sequence—is what CMoRE recognizes. Sequencing of DNA fragments after cutting showed that CMoRE acts like a restriction enzyme: it binds two modified cytosines with a characteristic spacing and makes clean cuts that produce short overhangs at the DNA ends.
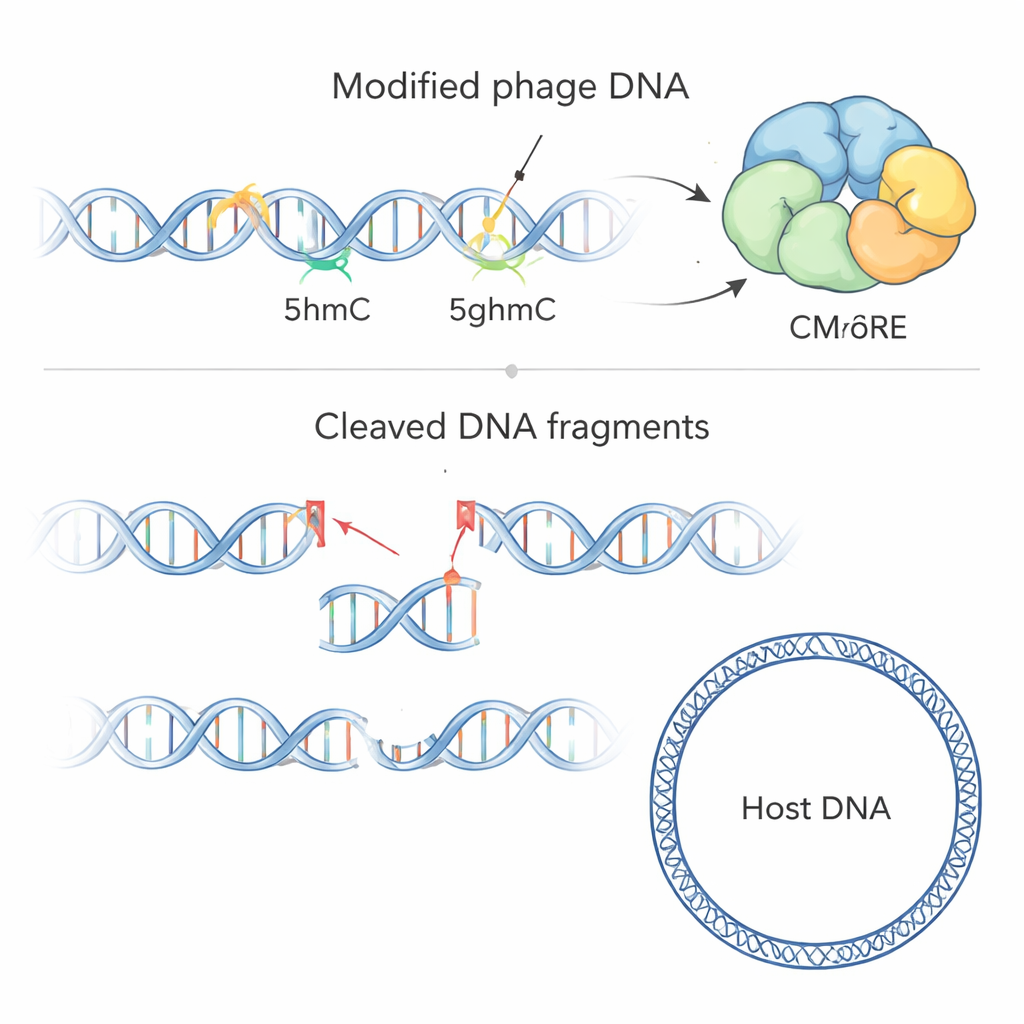
The Shape and Safety Switch of CMoRE
Using X-ray crystallography, the authors solved high-resolution structures of CMoRE from two bacterial species. The protein is made of two linked parts: an N-terminal “blade” that performs the DNA cutting, belonging to the GIY-YIG nuclease family, and a C-terminal “sensor” that grips the modified cytosine. Four copies of CMoRE assemble into a compact tetramer, and disrupting this assembly largely eliminates antiviral activity. The cutting domain carries a distinctive “GIYxY–YIG” motif and an unusual loop rich in negative charge that hangs over the active site like a lid. When the researchers neutralized this loop, CMoRE became overactive, started attacking normal bacterial DNA, and slowed cell growth—evidence that the loop acts as a built-in safety catch, helping the protein discriminate strongly in favor of phage DNA marked with 5hmC or 5ghmC.
From Microbial Warfare to Medical Tools
By scanning thousands of microbial genomes, the team found hundreds of related CMoRE systems spread across many bacterial groups, all sharing the same key features: the extra tyrosine in the catalytic motif and the negatively charged safety loop. This suggests CMoRE is a widely used strategy in bacterial warfare against chemically disguised phages. Because CMoRE can cleanly distinguish 5hmC and 5ghmC from the nearly identical 5mC, and because the protein is stable and easy to handle in the lab, it may also serve as a highly selective “molecular scalpel” for mapping 5hmC in mammalian genomes. That could improve tools for detecting disease-linked epigenetic changes, offering a practical payoff from understanding how bacteria survive their microscopic enemies.
Citation: Liu, R., Tang, D., Niu, M. et al. A bacterial defense system targeting modified cytosine of phage genomic DNA. Nat Commun 17, 1920 (2026). https://doi.org/10.1038/s41467-026-68792-8
Keywords: bacteriophage defense, DNA modification, 5-hydroxymethylcytosine, restriction enzyme, epigenetics