Clear Sky Science · en
Asymmetrical covalent organic framework mixed matrix membranes for highly efficient gas separation
Turning Waste Gases into Useful Hydrogen
Hydrogen is a promising clean fuel, but it is often produced together with carbon dioxide, a major greenhouse gas. Separating the tiny hydrogen molecules from larger carbon dioxide molecules efficiently and cheaply is a major challenge for future low‑carbon industry. This study reports a new kind of ultra‑thin, robust membrane that can sift hydrogen from carbon dioxide with unusually high speed and accuracy, potentially lowering the energy cost of cleaning up industrial gas streams.
Building a Smarter Filter
The researchers created a hybrid filter, called a mixed matrix membrane, that combines the flexibility of a plastic with the precision of a crystalline sieve. The crystalline component is a covalent organic framework (COF), a solid made from organic building blocks that link together to form highly ordered, nanoscale pores. These pores can be designed to favor some gas molecules over others. The plastic component, a polymer known as polyether sulfone, provides mechanical strength, chemical resistance, and easy processing into large sheets.
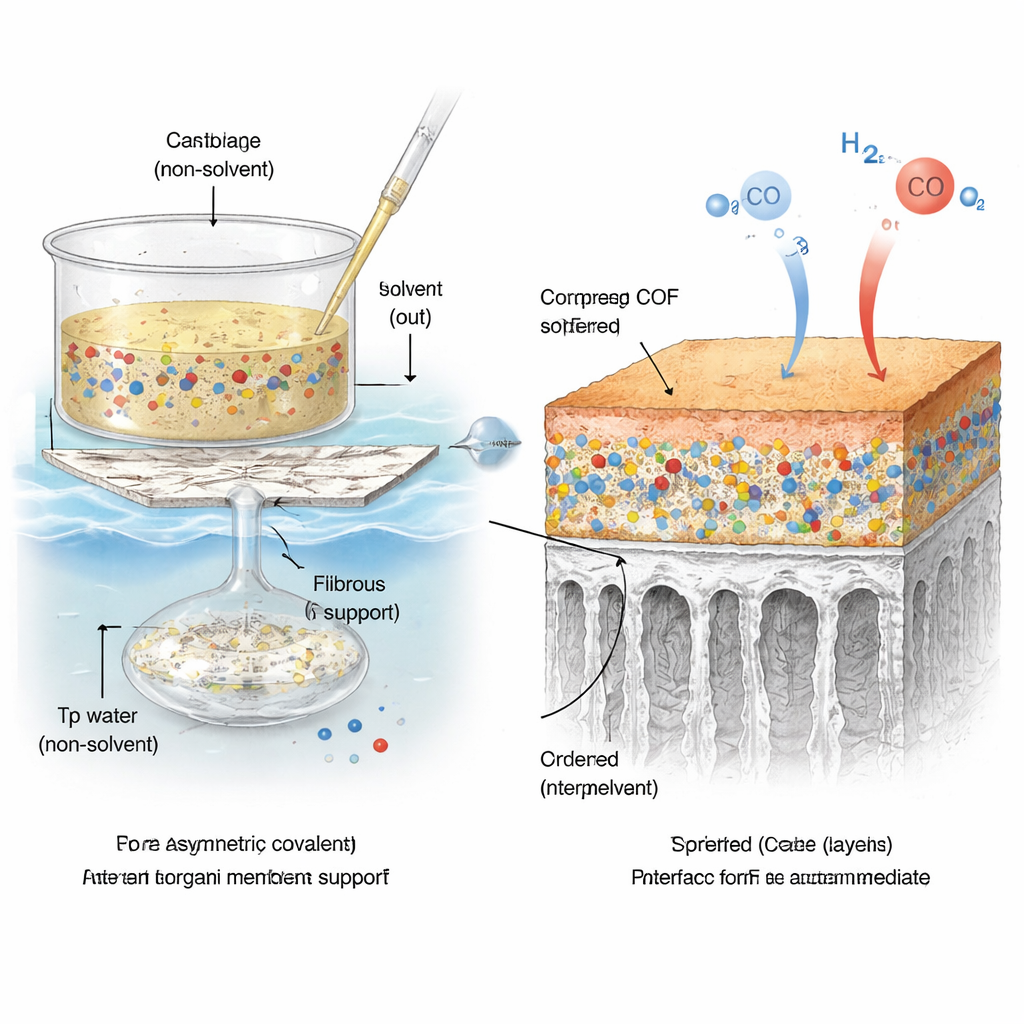
A Two‑Stage Formation Trick
To merge these very different materials without leaving defects, the team used a manufacturing method called non‑solvent induced phase separation. First, they dissolved both the polymer and one COF building block (called Tp) in a liquid and spread this mixture onto a porous glass fiber support. When this coated support was dipped into water, solvent and water exchanged rapidly, causing the polymer to solidify into an asymmetric structure with a dense “skin” on top and finger‑like pores underneath. At the same time, a second COF building block (Pa‑1), dissolved in the water bath, diffused into the forming film and reacted with Tp right at the polymer surface and within the pores.
A Layered Micro‑Architecture
This carefully timed process produced a multi‑layered architecture. On the very top sits an exceptionally thin COF film only 15–30 nanometers thick—thousands of times thinner than a human hair. Beneath it, the polymer forms a foam‑like region and long channels that connect down to the glass fiber mat. Tiny COF nanocrystals, just 4–8 nanometers across, are dispersed along the internal pore walls. High‑resolution microscopy and spectroscopy show that the polymer chains wrap tightly around these nanocrystals, forming a nearly seamless interface with no obvious gaps where gas could leak through uncontrolled. Hydrogen bonding and other weak interactions help “glue” the components together, while the glass fiber provides overall mechanical support.
Fast Hydrogen, Slowed Carbon Dioxide
When hydrogen and carbon dioxide are passed across this membrane, several separation effects work together. In the porous polymer regions, gas moves mainly by collisions with the pore walls, which naturally favor smaller, lighter molecules like hydrogen. Inside the COF domains, computer simulations and gas tests show that carbon dioxide is strongly attracted and becomes temporarily trapped, while hydrogen feels only a weak attraction and can pass more freely. As carbon dioxide fills parts of the COF pores, the effective gaps between the stacked COF layers narrow, acting as a molecular sieve that further slows the bulkier carbon dioxide while letting hydrogen slip through.
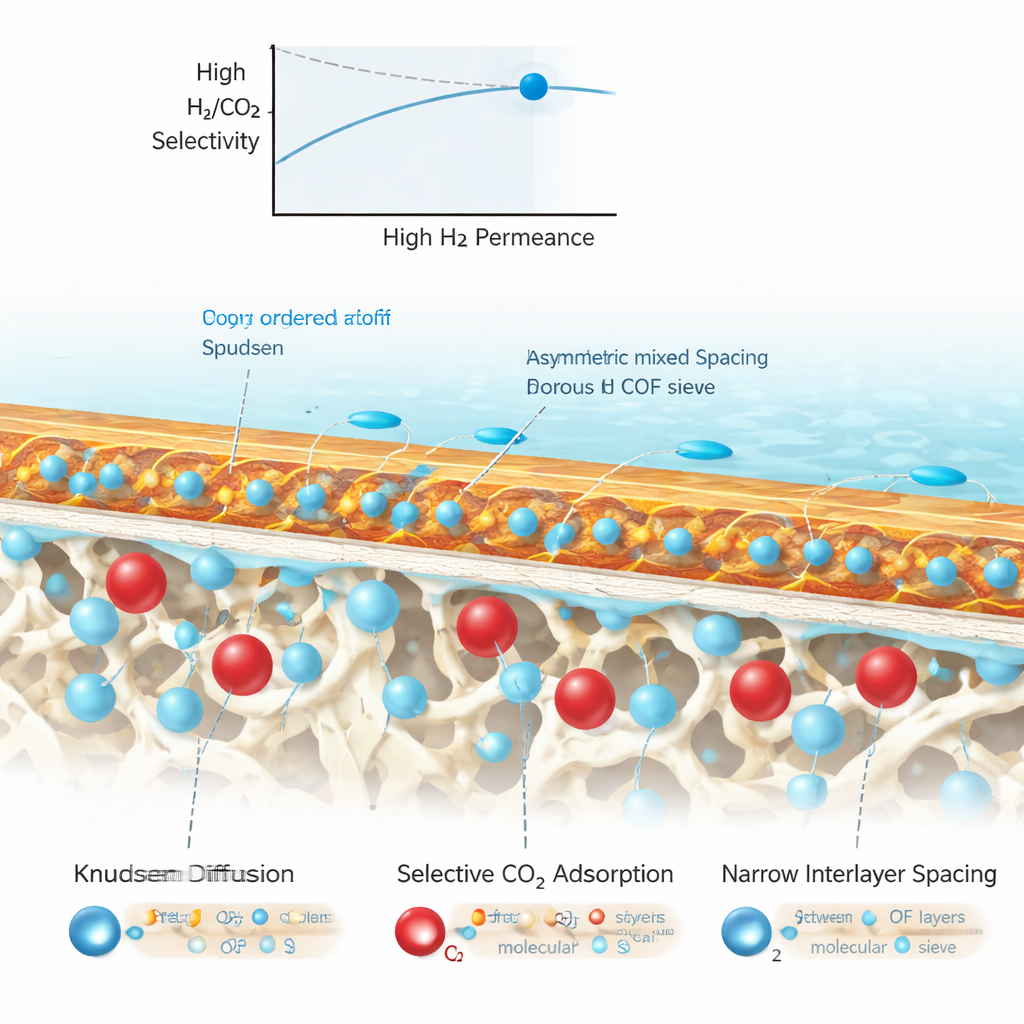
Performance That Breaks Past Old Limits
These combined effects yield a hydrogen flux that remains very high while carbon dioxide passage is strongly suppressed. At room temperature, the membrane reaches a hydrogen permeance of about 2700 GPU and a hydrogen‑to‑carbon‑dioxide selectivity near 89—figures that surpass a widely used benchmark known as the Robeson upper bound for traditional polymer membranes. The membrane also keeps working well at elevated temperatures and shows stable operation over many hours, even after mechanical handling and damage tests. This demonstrates that the unusual layered structure is not only effective but also durable and scalable.
What This Means for Clean Energy
In everyday terms, the team has built a gas filter that lets hydrogen shoot through quickly while holding back most of the carbon dioxide, all in a sheet that is thin, strong, and manufacturable over centimeter‑scale areas. By combining a plastic framework with a crystalline sieve grown directly inside it, they overcome long‑standing trade‑offs between speed and accuracy in gas separation. If adapted to industrial modules, such membranes could help make hydrogen production and carbon capture more energy‑efficient, supporting cleaner fuels and reduced emissions.
Citation: Qi, LH., Wang, Z., Zhang, TH. et al. Asymmetrical covalent organic framework mixed matrix membranes for highly efficient gas separation. Nat Commun 17, 1947 (2026). https://doi.org/10.1038/s41467-026-68790-w
Keywords: hydrogen separation, gas membranes, covalent organic frameworks, carbon capture, mixed matrix materials