Clear Sky Science · en
Collagen binding adhesin restricts Staphylococcus aureus skin infection
Why a skin germ’s “Velcro” matters
Staphylococcus aureus is the main cause of painful skin boils and abscesses, and drug‑resistant strains like MRSA make these infections hard to treat. This study looks at an unexpected twist: a bacterial surface protein called Cna that acts like Velcro for collagen, the body’s main structural protein in skin and wounds. Instead of making infections worse, the authors find that this collagen‑grabbing protein actually tames skin disease in mice by shaping how immune cells attack the bacteria.
A sticky protein with a surprising job
Collagen is the most abundant protein in our bodies and is essential for holding skin together and healing wounds. Some S. aureus strains carry Cna, a protein that anchors in the bacterial cell wall and binds tightly to collagen and to a collagen‑like tail on a blood protein called C1q. C1q normally helps mark microbes for destruction by the immune system. Earlier work had linked Cna to serious infections in joints and eyes, but its role in skin disease was unclear. The researchers used a mouse skin abscess model and genetically engineered bacteria that either made Cna, lacked it, or had it restored from a plasmid, to ask how this single adhesin changes the course of infection.
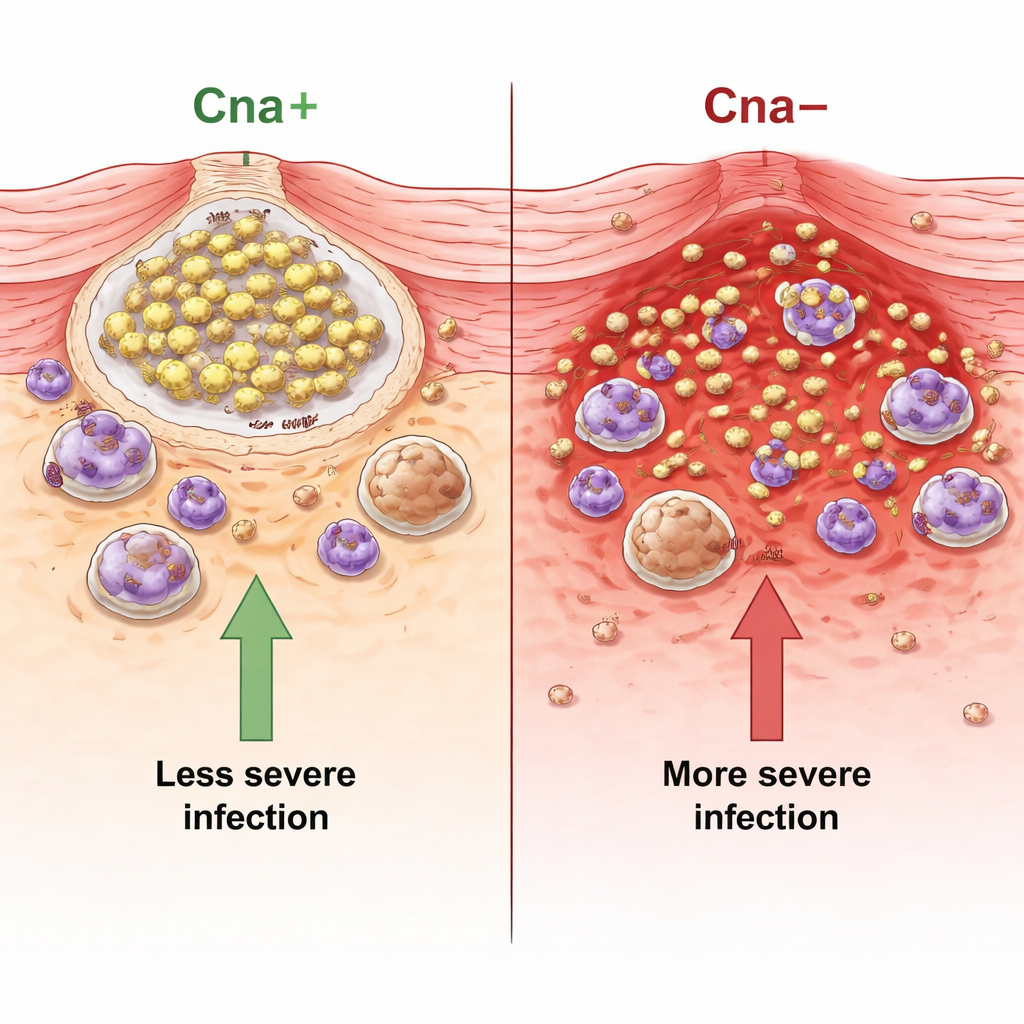
When bacteria can’t grab collagen, infections get worse
In mice injected with Cna‑positive S. aureus, skin lesions stayed smaller, animals lost less weight, and far fewer bacteria were recovered from the abscesses than in mice infected with Cna‑negative mutants. Restoring Cna reversed these effects, showing the protein itself was responsible. The team also tested USA300, a common MRSA strain that naturally lacks Cna and is notorious in human skin infections. For this strain too, adding Cna reduced lesion size and bacterial counts. Surveys of patient isolates from skin infections, nasal carriers, and pneumonia revealed that most circulating S. aureus strains do not carry the cna gene at all, suggesting that losing this protein may actually favor more aggressive, spreading disease.
How collagen shields bacteria and calms inflammation
Tissue sections from mouse abscesses told a clear story. Infections with Cna‑positive bacteria showed compact bacterial clusters wrapped in collagen, with immune cells such as neutrophils and macrophages gathered at the edges. In contrast, Cna‑negative infections were ragged and infiltrated, packed with dead immune cells and intense inflammation. Chemical analysis of the tissue showed that Cna‑negative infections triggered higher levels of inflammatory signals like IL‑6, TNF‑α, and IL‑1β, and of enzymes called matrix metalloproteases (MMP‑9 and MMP‑12) and the lipid mediator leukotriene B4, all of which amplify immune cell influx and tissue damage. Flow cytometry and imaging confirmed that neutrophils in Cna‑negative abscesses were far more likely to be dead or dying, while live immune cells were relatively scarce.
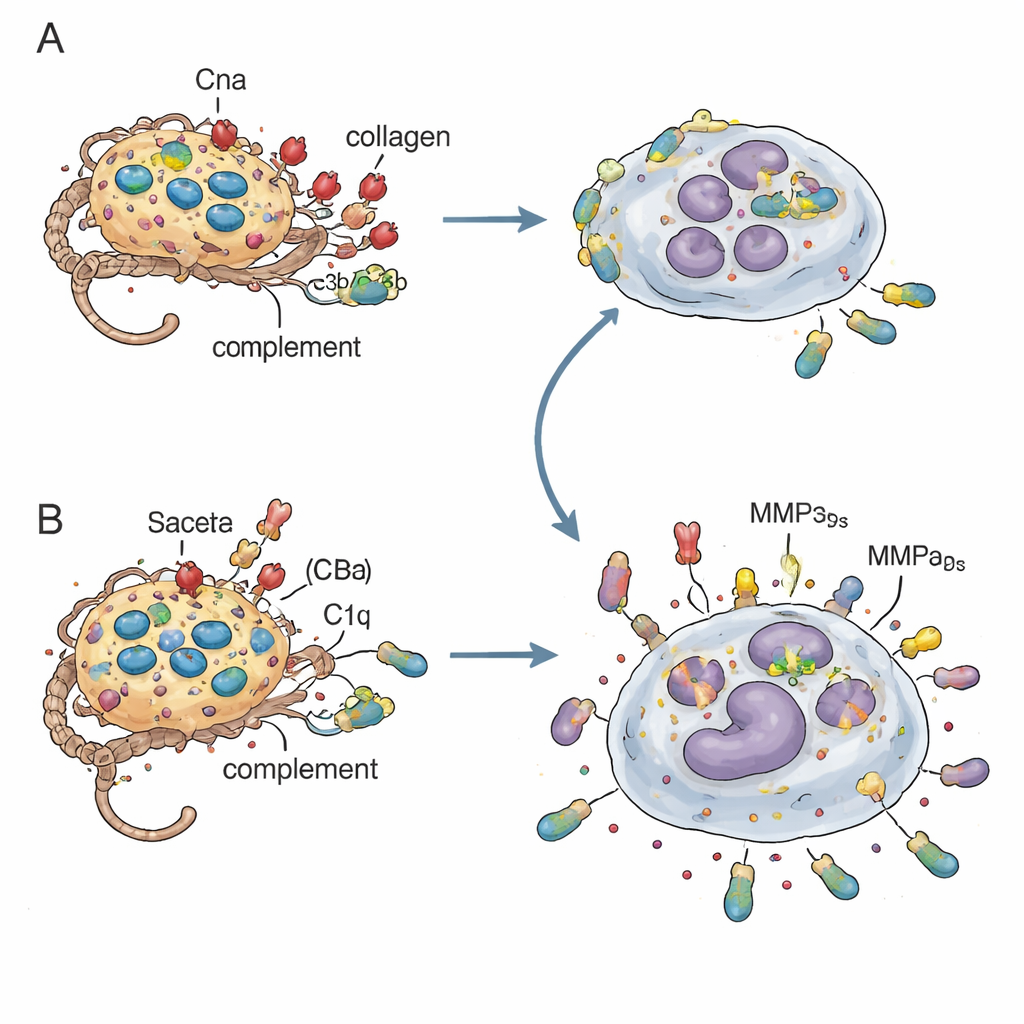
Hijacking complement to avoid a deadly embrace
The key mechanistic link was C1q, a starter molecule of the complement system that coats bacteria so neutrophils can engulf them. In test‑tube experiments, bacteria lacking Cna accumulated more complement components (C4b and C3b) on their surface and were taken up more avidly by human neutrophils. But this overzealous uptake backfired: neutrophils stuffed with Cna‑negative bacteria tended to lyse, spilling live bacteria and inflammatory molecules back into the tissue. When the researchers used serum depleted of C1q, or mice genetically lacking C1q, the differences between Cna‑positive and Cna‑negative infections shrank, showing that Cna’s binding to the collagen‑like tail of C1q blunts complement tagging and tempers neutrophil overreaction. At the same time, collagen bound around Cna‑positive bacteria appears to create a physical barrier, slowing direct contact with neutrophils and allowing more controlled killing.
Balancing defense and damage in skin infections
To a non‑specialist, the main message is that not all bacterial “stickiness” makes infections worse. In this case, S. aureus strains that can cling to collagen and C1q provoke a more measured immune response: fewer neutrophils die explosively, inflammation is lower, and skin damage is reduced, even though the bacteria are still present. Strains that lack Cna, including many important MRSA clones, are more easily flagged by complement, drive a storm of neutrophil activity, and end up causing larger, more destructive abscesses. Understanding this fine balance between bacterial hiding and immune overreaction could inform new strategies to treat skin infections—either by targeting how bacteria interact with collagen and C1q, or by modulating the inflammatory enzymes and lipids that turn a contained abscess into a damaging lesion.
Citation: Bhattacharya, M., Spencer, B.L., Kwiecinski, J.M. et al. Collagen binding adhesin restricts Staphylococcus aureus skin infection. Nat Commun 17, 1980 (2026). https://doi.org/10.1038/s41467-026-68788-4
Keywords: Staphylococcus aureus, skin abscess, collagen, neutrophils, complement system