Clear Sky Science · en
Spinal cord stimulation therapy for gait impairment in Parkinson’s disease: a double-blinded, randomised feasibility trial with an open extension
When Walking Becomes a Daily Struggle
Many people with Parkinson’s disease find that walking slowly turns from an automatic act into a constant challenge. Their feet may suddenly feel “glued” to the floor, making everyday tasks risky and exhausting. This study asked a straightforward but important question: can a small implanted device that sends gentle electrical pulses to the spinal cord safely help these patients walk better, and is it worth testing in larger trials?
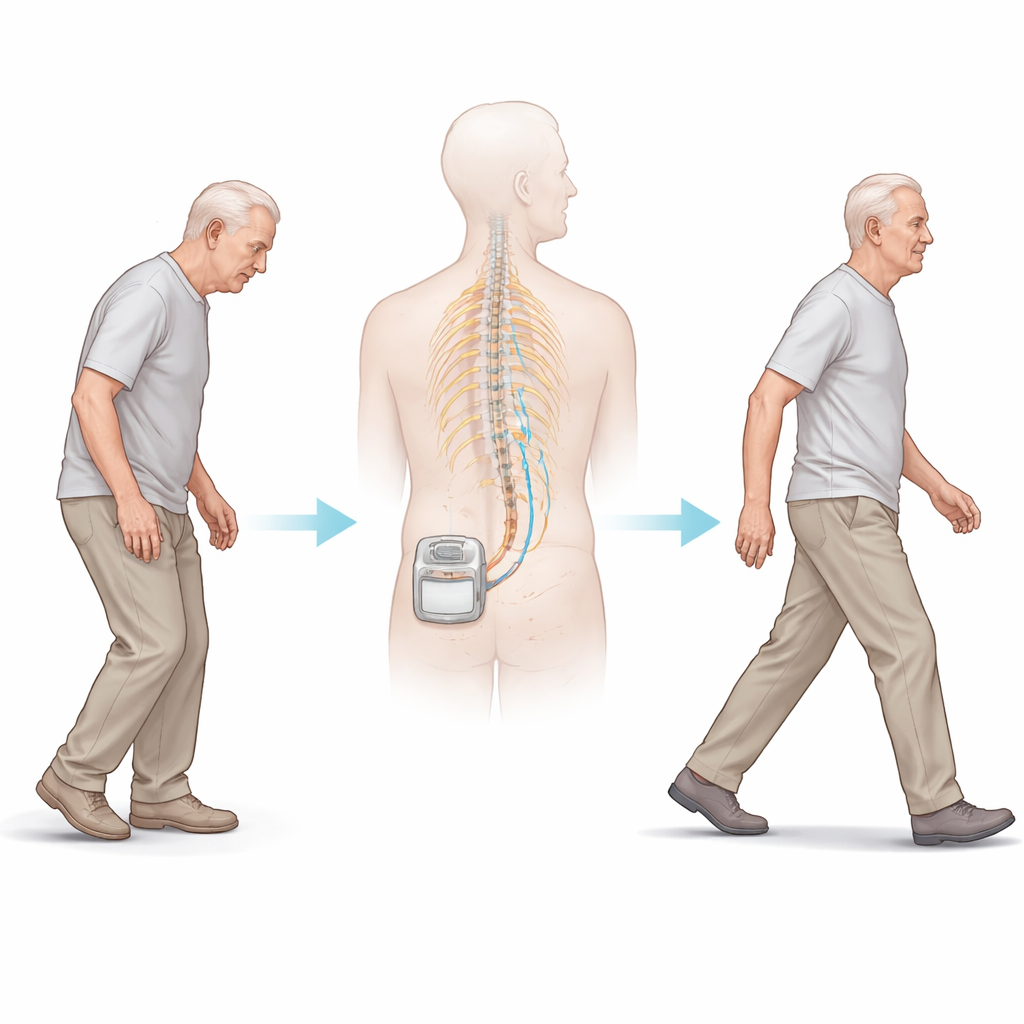
A New Use for a Pain Therapy
Spinal cord stimulation is already used to ease severe, long‑lasting pain. Surgeons place a thin lead along the spine and connect it to a battery-powered pulse generator under the skin. The device delivers tiny electrical bursts to nerve pathways. Over the past decade, doctors have reported that some people with Parkinson’s who received this treatment for back pain also seemed to walk more easily. That observation inspired the STEP-PD trial, which tested a specific “burst” pattern of stimulation designed not to cause tingling sensations, making it possible to fairly compare real stimulation with a sham (device off) condition.
How the Trial Was Set Up
The researchers recruited people with Parkinson’s disease who had troublesome freezing of gait despite well-tuned medication. After surgery to implant the spinal cord stimulator near the middle back, twelve participants entered a six‑month double‑blind phase: half were randomly assigned to receive burst stimulation, and half to have their device left off. Neither the patients nor the examining doctors knew who was in which group. All participants then continued for another six months in an open phase where everyone received active stimulation. Along the way, the team measured balance and walking using standard movement scores, simple walking tests, home sensors, and questionnaires about mobility and quality of life. Brain scans using two types of PET imaging tracked changes in brain activity and in a chemical signal system related to attention and movement.
Safety First, Signals Second
The trial showed that this kind of spinal cord stimulation was largely safe and acceptable. Most problems were linked to the surgery itself, such as temporary pain, swelling, or the need to reposition a lead, and these issues were managed without lasting harm. Importantly, the study’s main clinical test—an overall score of balance and walking problems—did not improve significantly after six months of burst stimulation compared with the sham group. Yet when the researchers looked more closely, they found encouraging signs: stiffness and slowness in the legs improved during stimulation, and a detailed “Lower Body and Gait” score dropped over time, especially after a full year of treatment. When the team compared these patients to a similar group from a large Parkinson’s database, those without stimulation tended to worsen over a year, while those with stimulation tended to hold steady or improve slightly.
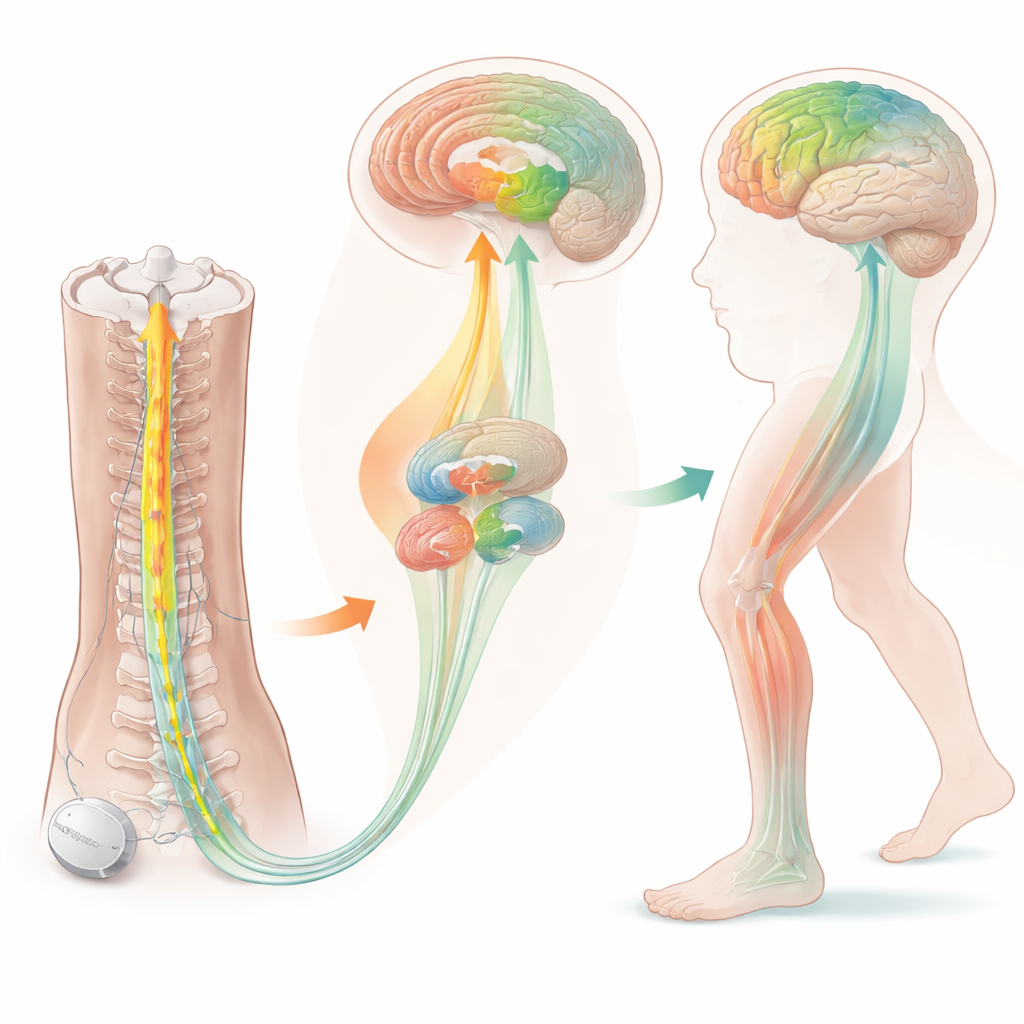
What the Brain Scans Revealed
The brain imaging added a window into how stimulation might be working. Before treatment, people with freezing of gait showed unusual activity in brain regions involved in attention and movement control, including the right frontal area and a hub called the anterior insula. After months of stimulation, glucose use in the thalamus—a relay station that helps coordinate movement—decreased, and signals from certain cholinergic (acetylcholine-using) nerve endings in motor and attention regions also went down. Rather than indicating damage, these shifts likely reflect a normalization of overactive circuits that the brain had been using to compensate for gait problems. These biological changes aligned with the gradual easing of leg rigidity and slowness seen in clinical testing.
Why Larger, Smarter Trials Are Needed
From a layperson’s perspective, the message is mixed but hopeful. This small, carefully controlled trial found that burst spinal cord stimulation did not deliver a clear, short‑term fix for walking difficulties in Parkinson’s disease, so it cannot yet be recommended as a proven gait treatment. However, the therapy appeared safe, and over time it seemed to ease leg stiffness and slowness, with brain scans pointing to meaningful shifts in movement networks. The study also highlighted important design lessons—who to enroll, which scores are most sensitive, how long to treat—that will guide larger, more definitive trials. In other words, while spinal cord stimulation is not yet a walking cure, it may become part of a future toolbox to help people with Parkinson’s stay on their feet longer and more safely.
Citation: Terkelsen, M.H., Hvingelby, V.S., Johnsen, E.L. et al. Spinal cord stimulation therapy for gait impairment in Parkinson’s disease: a double-blinded, randomised feasibility trial with an open extension. Nat Commun 17, 2168 (2026). https://doi.org/10.1038/s41467-026-68782-w
Keywords: Parkinson’s disease, gait impairment, spinal cord stimulation, deep brain networks, neuromodulation