Clear Sky Science · en
Proteomic profiling of UV damage repair patches uncovers histone chaperones with central functions in chromatin repair
How Our Cells Heal Sunlight Damage
Every time our skin meets strong sunlight, invisible injuries appear in the DNA of our cells. If left unchecked, these tiny scars can build up, contributing to aging and cancer. But DNA is not naked; it is carefully wrapped around proteins called histones, forming a structure known as chromatin that helps define each cell’s identity. This study asks a deceptively simple question with big implications: when cells repair UV damage in their DNA, how do they rebuild this intricate chromatin architecture so that cell identity is preserved rather than scrambled?
Uncovering the Hidden Repair Zone
To answer this, the researchers had to peer specifically at the spots in the genome where repair was actively happening, instead of surveying the whole nucleus at once. They devised an approach called IPOND-R that chemically tags short stretches of freshly made DNA produced during repair, not during normal copying of the genome. By pulling down these tagged DNA “repair patches” along with all the proteins attached to them, and then identifying those proteins with high-resolution mass spectrometry, they created a time-resolved catalog of who shows up, and when, during the response to UV damage in human cells. This gave them an unbiased snapshot of the specialized protein neighborhood that forms directly at repair sites.
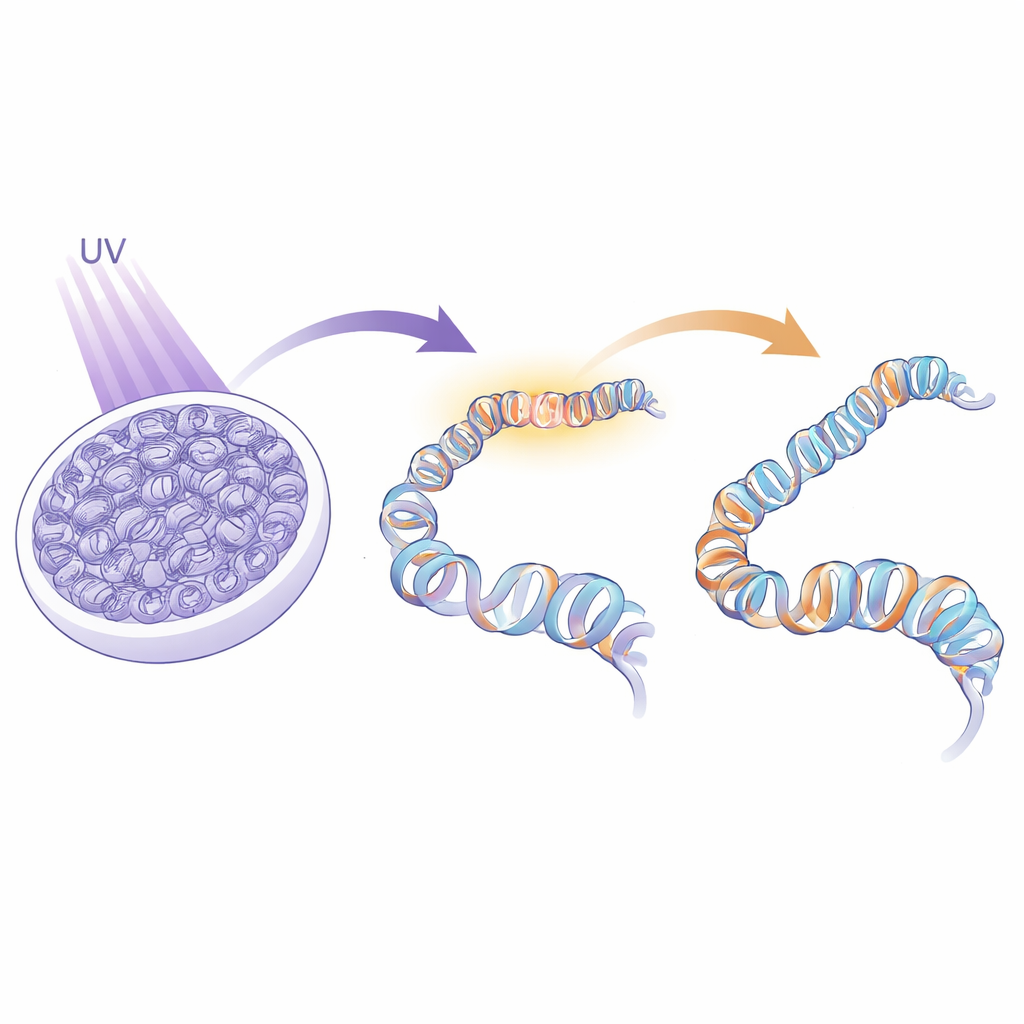
A Busy Crowd at the Repair Scaffold
The IPOND-R method revealed hundreds of proteins enriched on DNA undergoing repair shortly after UV exposure. As expected, well-known DNA repair factors that recognize and cut out UV-induced lesions were present. But the dataset went much further, capturing proteins involved in gene regulation, nuclear architecture and, crucially, chromatin organization. Many of these visitors were histone chaperones—specialized proteins that escort histones on and off DNA. Comparing early and late time points showed that most of these chaperones appear only transiently, suggesting a carefully choreographed sequence of chromatin disassembly and reassembly tightly coupled to the DNA repair process.
New Suppliers and Recyclers of Histones
Among the proteins enriched at repair patches, two histone chaperones stood out: DNAJC9 and MCM2. These factors were previously known mainly for roles near DNA replication forks, where the genome is copied, but had not been connected to UV damage repair. Here, the team shows that DNAJC9 is a central supplier of fresh histone units (specifically H3–H4 pairs) during repair. Using fluorescent tags that distinguish new histones from old ones, they found that reducing DNAJC9 levels strongly diminished the arrival of newly made histone variants at UV-damaged sites, while leaving the core DNA repair steps intact. DNAJC9 acts upstream of known histone-depositing chaperones CAF-1 and HIRA, feeding them new histones without altering their own presence at damage sites.
Balancing Old and New Building Blocks
Repairing chromatin is not simply a matter of patching in new parts. The original histones also carry chemical marks that store epigenetic information—signals that tell a cell which genes to keep on or off. The study shows that DNAJC9 also helps bring these parental histones back after they temporarily move away from damaged regions. When DNAJC9 was depleted, old histones failed to fully return, and overall histone density at repair sites dropped. The researchers then discovered that DNAJC9 works together with MCM2 to coordinate this delicate balance: both are required for efficient recycling of old histones and for proper deposition of new ones, even in cells that are not actively replicating their DNA. This points to a replication-independent role for MCM2 and establishes a functional partnership between the two chaperones at repair sites.
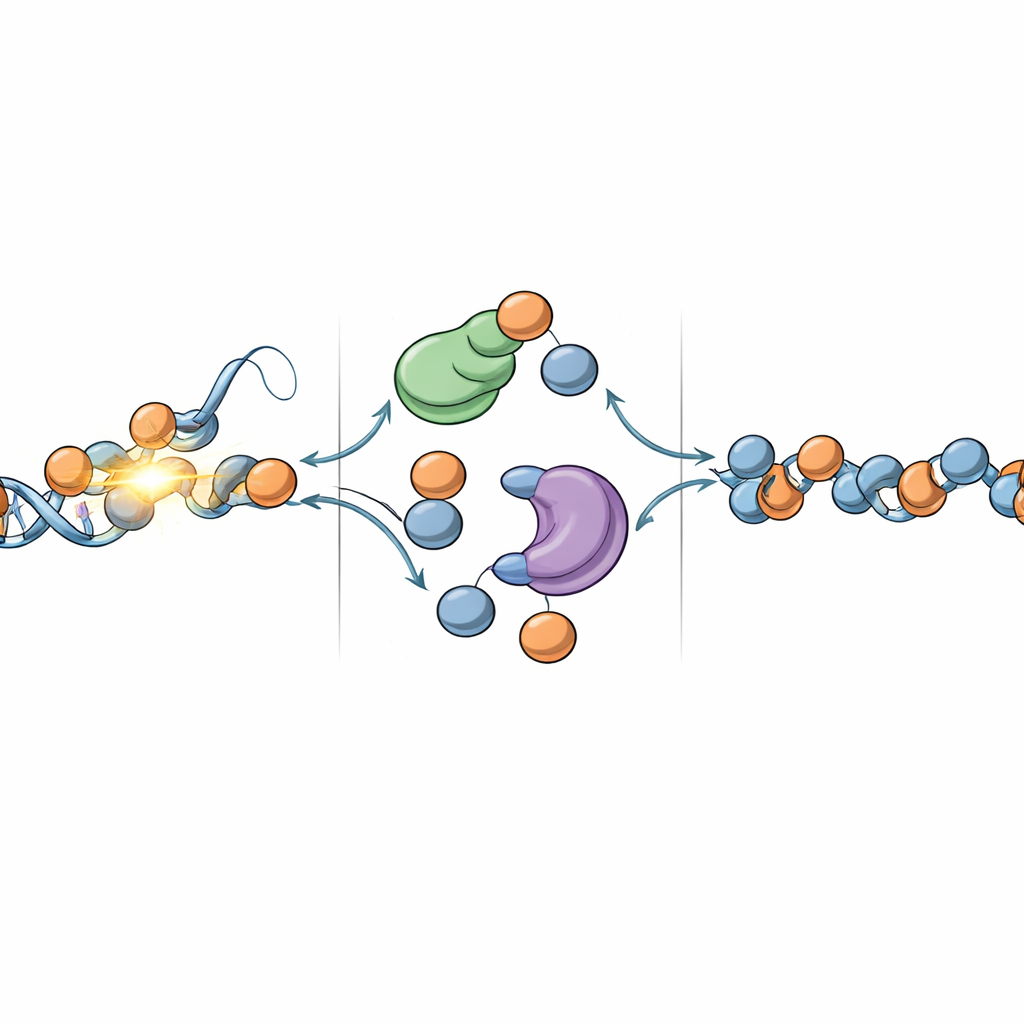
Why This Matters for Cell Identity and Disease
The work paints a picture of chromatin repair as a two-sided operation: damaged DNA is fixed while the surrounding histone landscape is rebuilt using a mix of reused and freshly supplied components. DNAJC9 and MCM2 sit at a crossroads in this process, coordinating the flow of old and new histones so that, after UV damage, cells can restore not just the genetic code but also the epigenetic patterns that define their identity. Because similar challenges arise with many types of DNA damage, this framework offers a foundation for understanding how cells maintain a stable epigenome under stress—an issue central to aging, cancer and other diseases where chromatin regulation goes awry.
Citation: Plessier, A., Chansard, A., Petit, E. et al. Proteomic profiling of UV damage repair patches uncovers histone chaperones with central functions in chromatin repair. Nat Commun 17, 2127 (2026). https://doi.org/10.1038/s41467-026-68781-x
Keywords: DNA damage repair, chromatin, histone chaperones, UV radiation, epigenome stability