Clear Sky Science · en
Structural basis and pathological implications of the dimeric OS9-SEL1L-HRD1 ERAD Core Complex
Cellular Clean-Up Crew Under the Microscope
Inside each of our cells, a busy factory turns genetic instructions into working proteins. Like any factory, mistakes happen. When proteins misfold, they can clog up the system and contribute to disease. This study zooms in on one of the cell’s main quality-control machines—the SEL1L‑HRD1 complex—to reveal its detailed 3D structure and show how tiny genetic changes can break this machinery and potentially lead to human illness.
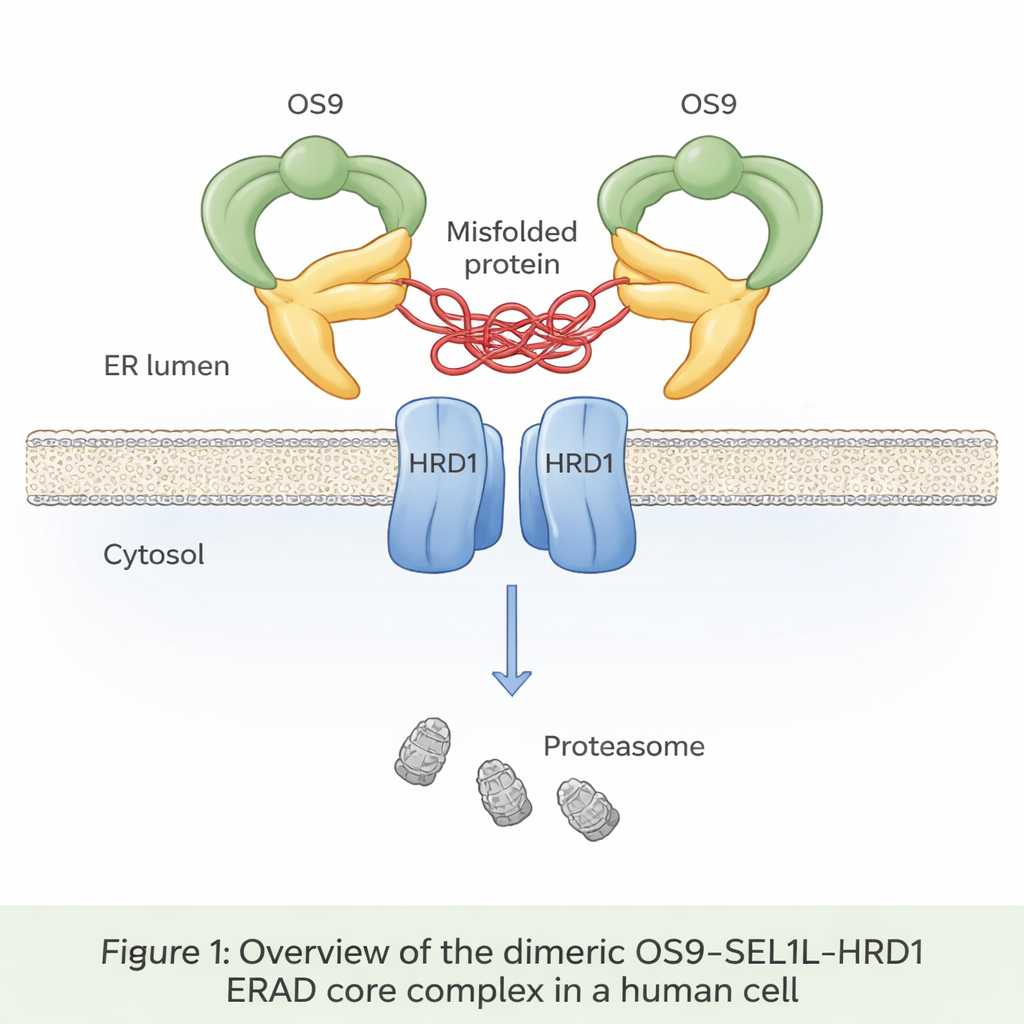
A Hidden Conveyor Belt in the Cell
Up to a third of all newly made proteins enter a compartment called the endoplasmic reticulum, or ER, where they are folded and checked. Misfolded proteins are normally recognized, pulled back out of the ER, and destroyed in a process known as ER‑associated degradation (ERAD). At the heart of one major ERAD pathway sits a trio of proteins: OS9, SEL1L, and HRD1. OS9 acts as a sensor for faulty, sugar‑tagged proteins; SEL1L serves as a scaffold; and HRD1 tags doomed proteins with small ubiquitin flags that mark them for destruction by the cell’s waste‑disposal units, the proteasomes. Until now, however, no one had seen in atomic detail how these three pieces fit together in human cells.
Revealing the Shape of the Core Machine
The authors used cryo‑electron microscopy, a technique that images flash‑frozen molecules at near‑atomic resolution, to visualize the OS9‑SEL1L‑HRD1 complex purified from human cells. They discovered that it forms a dimer—essentially two identical copies joined together—rather than staying as single units. On the ER lumen side (the inside of the ER), two OS9 and two SEL1L molecules assemble into a crab‑claw‑like ring with a central opening that appears designed to grip misfolded proteins. Within the membrane itself, two HRD1 molecules pair up to form a shared channel. This arrangement positions the “claw” directly above the HRD1 doorway, creating a continuous path for misfolded proteins to move from the ER lumen, through the membrane, and toward degradation in the cytosol.
How Tiny Changes Break a Big System
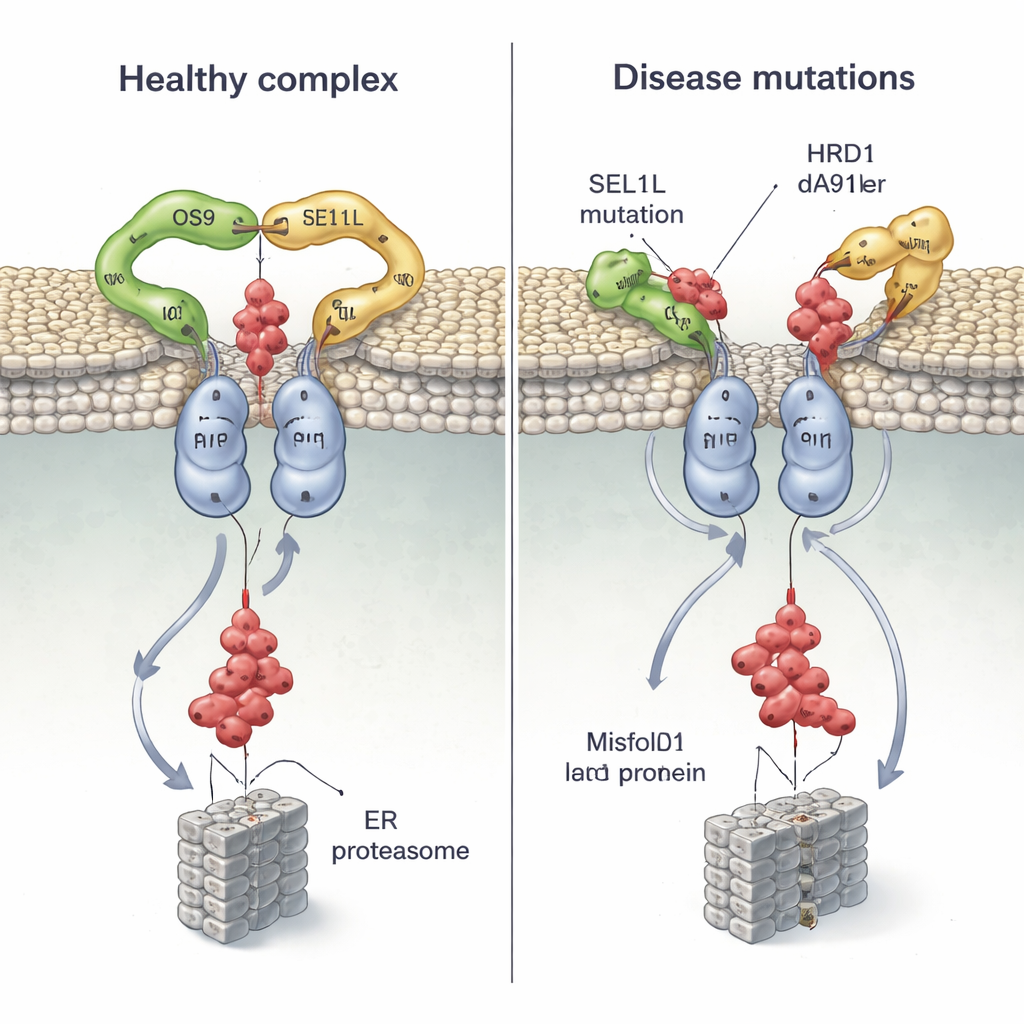
Because mutations in SEL1L and HRD1 have been found in patients with severe neurodevelopmental and other disorders, the team mapped several disease‑linked variants onto their structure and tested how they behave in cells. Two SEL1L mutations, G585D and S658P, sit right at the contact points with OS9 and HRD1, respectively. In cell experiments, G585D almost abolished SEL1L’s ability to bind OS9, while S658P greatly weakened its grip on HRD1; combining both mutations essentially broke the core complex, yet left other partners untouched. As a result, the cells struggled to tag and clear a misfolded hormone precursor, allowing faulty proteins to linger.
A Disease Mutation in the Membrane Channel
The structure also shows that HRD1’s transmembrane segment 3 is the key interface where two HRD1 molecules meet to form the channel. The researchers engineered cysteine “handles” at specific positions and used chemical crosslinking to confirm that these regions come into close contact in living cells, proving that HRD1 truly dimerizes in vivo. When they disrupted a single, highly conserved residue (T93) at this interface, the dimer fell apart and ERAD activity collapsed, even though the complex still assembled with OS9 and SEL1L. They then examined a newly discovered patient variant, HRD1 A91D, found in a child with heart and lung problems. This change, also in the dimer interface, cut HRD1 dimerization roughly in half and severely impaired the disposal of misfolded proteins, again without disturbing the overall binding of partners.
A New View of Protein Quality Control and Disease
By combining structural biology with cell‑based tests, this work shows that the OS9‑SEL1L‑HRD1 complex operates as a paired, dimeric machine: a claw‑like catcher linked to a shared channel that shuttles faulty proteins out of the ER. Mutations that loosen the claw’s grip or destabilize the HRD1 pair do not simply tweak efficiency—they can effectively jam the system, allowing damaged proteins to build up and contribute to human disease. For non‑specialists, the key message is that even single‑letter changes in our DNA can subtly warp the shape of essential cellular machines, with far‑reaching consequences for brain development, immunity, and organ function.
Citation: Lin, L.L., Maldosevic, E., Zhou, L.E. et al. Structural basis and pathological implications of the dimeric OS9-SEL1L-HRD1 ERAD Core Complex. Nat Commun 17, 2064 (2026). https://doi.org/10.1038/s41467-026-68777-7
Keywords: protein quality control, endoplasmic reticulum, ERAD, SEL1L-HRD1 complex, protein misfolding