Clear Sky Science · en
Stereoselective cyano translocation reaction enabled by photoenzymatic catalysis
Moving Tiny Parts for Big Impact
Chemists often want to tweak a molecule the way an engineer might reposition a gear in a machine—moving a small piece without rebuilding everything from scratch. This paper shows how scientists can gently slide a cyano group, a tiny but powerful chemical handle, from one spot on a molecule to another using light and specialized enzymes. The result is a more precise and sustainable way to build the kinds of molecules used in medicines and advanced materials.
Why Shifting a Small Group Matters
The behavior of an organic molecule is largely dictated by its functional groups—the small clusters of atoms that act like control switches. Moving such a group even a short distance along a carbon chain can dramatically change how the molecule behaves in the body or in a material. Chemists know how to make these migrations happen, especially using radical reactions, but they usually struggle to control the “handedness” of the product. Like left and right hands, many molecules come in mirror-image forms, and often only one of them is useful or safe. Until now, getting the preferred hand during these group-shifting reactions has been very difficult.
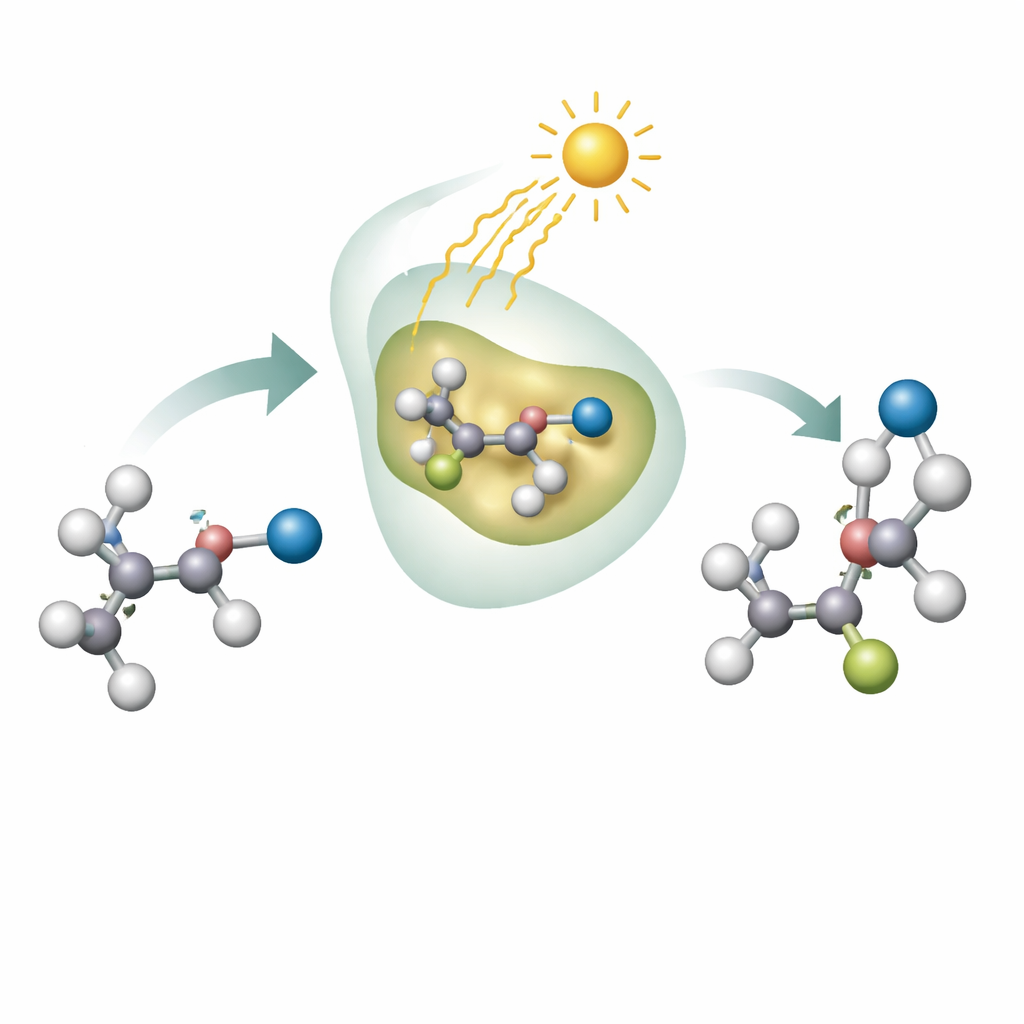
Harnessing Enzymes and Light Together
The authors combine the strengths of enzymes and light to solve this problem. They focus on moving a cyano group (the CN unit) along a carbon chain in molecules called alkyl nitriles, which are important building blocks that can be turned into many other useful functions. The team uses flavin-dependent enzymes, a common class of proteins that naturally handle redox chemistry in living cells. When the flavin cofactor inside these enzymes absorbs blue light, it enters an excited state powerful enough to snip off an iodine atom from a starting molecule, creating a highly reactive radical. Inside the snug pocket of the enzyme, this radical reaches over to the cyano group, sets off a rearrangement that relocates it, and then is carefully “switched off” by transfer of a hydrogen atom from the flavin.
Making One Mirror Image on Demand
A key achievement of this work is that the enzymes not only move the cyano group, they do so with excellent control over handedness. By screening natural enzymes and then refining them, the researchers identify systems that give one mirror image of the product with very high purity, and others that favor the opposite image. They show that a wide range of starting molecules, with different aromatic rings and side chains, can undergo this light-driven cyano shift while preserving strong preference for a single hand. Electronic fine-tuning of the starting materials—adding groups that donate or withdraw electrons—further affects how cleanly the preferred hand is formed, revealing how delicate the underlying balance of reactivity is.
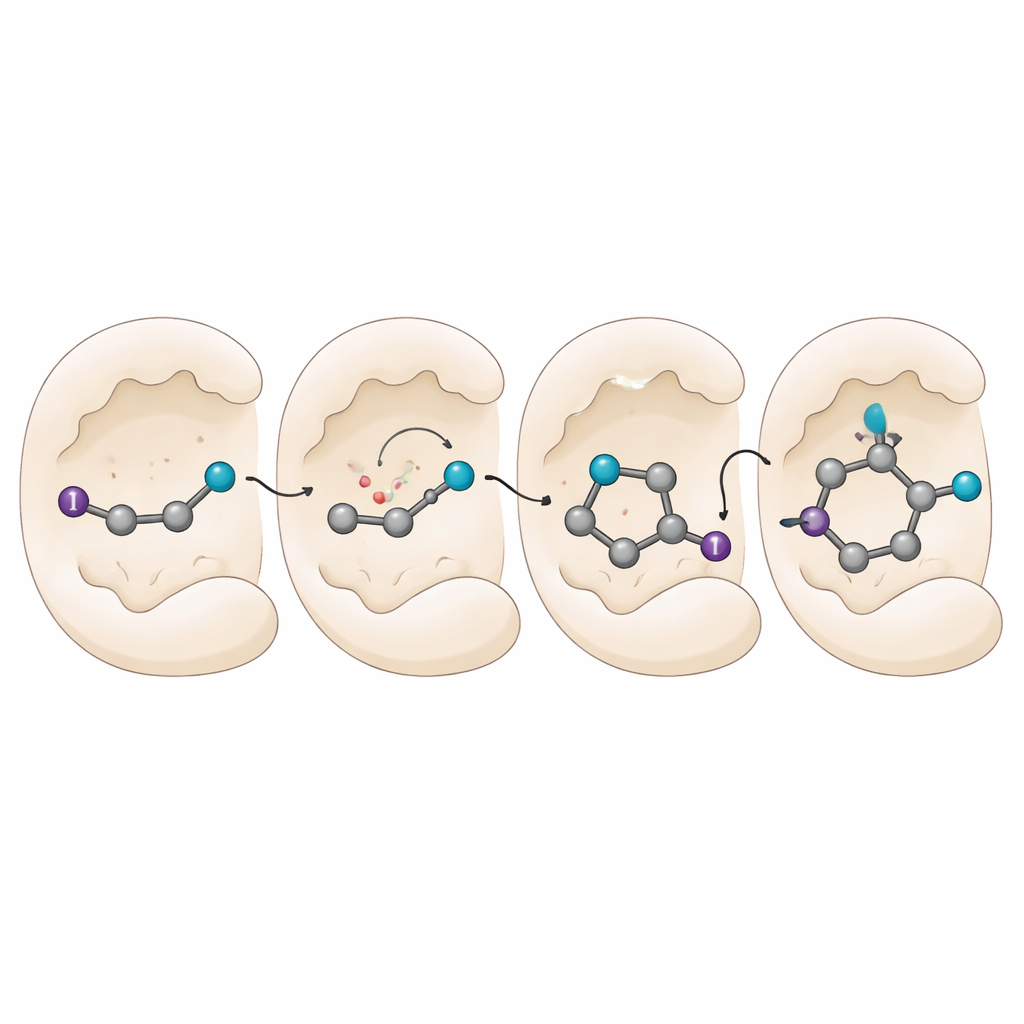
Peeking Inside the Molecular Machine
To understand how the enzymes impose such precise control, the team performs mechanistic experiments and computer simulations. Radical-trapping tests confirm that the process truly goes through radical intermediates, yet much of the chemistry is shielded inside the enzyme, where outside traps struggle to interfere. Optical measurements reveal that the enzyme and substrate form a special light-absorbing complex that helps initiate the reaction. Simulations of the enzyme–radical complex show that the cyano group is anchored by specific amino acids, while the rest of the molecule can rotate into favored poses. Subtle stacking interactions between the aromatic ring of the substrate and certain amino acids tilt the balance toward one face of the radical when the final hydrogen atom is delivered, locking in which mirror image is produced.
A New Tool for Building Better Molecules
In the end, this study introduces a new way to rearrange molecules with remarkable finesse. By using photoactivated enzymes to guide a radical-based cyano shift, the authors demonstrate that it is possible to move a functional group while also deciding exactly which mirror form the product will take. For drug discovery and materials science, this offers a flexible and greener route to fine-tuned molecular structures, expanding the toolkit that chemists can use to design safer medicines and smarter materials.
Citation: Duan, X., Xu, J., Bai, R. et al. Stereoselective cyano translocation reaction enabled by photoenzymatic catalysis. Nat Commun 17, 2133 (2026). https://doi.org/10.1038/s41467-026-68776-8
Keywords: photoenzymatic catalysis, functional group migration, cyano translocation, enzyme stereocontrol, alkyl nitriles