Clear Sky Science · en
Granzyme B-mimetic nanozyme for nanovesicle targeted anticancer applications
Reinventing the Body’s Cancer Killers
Our immune system has specialized cells that can hunt down and kill cancer, but in many solid tumors they struggle to get in, tire quickly, or attack the wrong targets. This study describes a fully artificial, nanoscale system that imitates one of the immune system’s most powerful weapons and delivers it directly into tumor cells. The work matters because it points toward cancer treatments that behave like living immune cells, yet are made from stable, controllable materials rather than fragile human cells.
Turning a Natural Assassin into a Blueprint
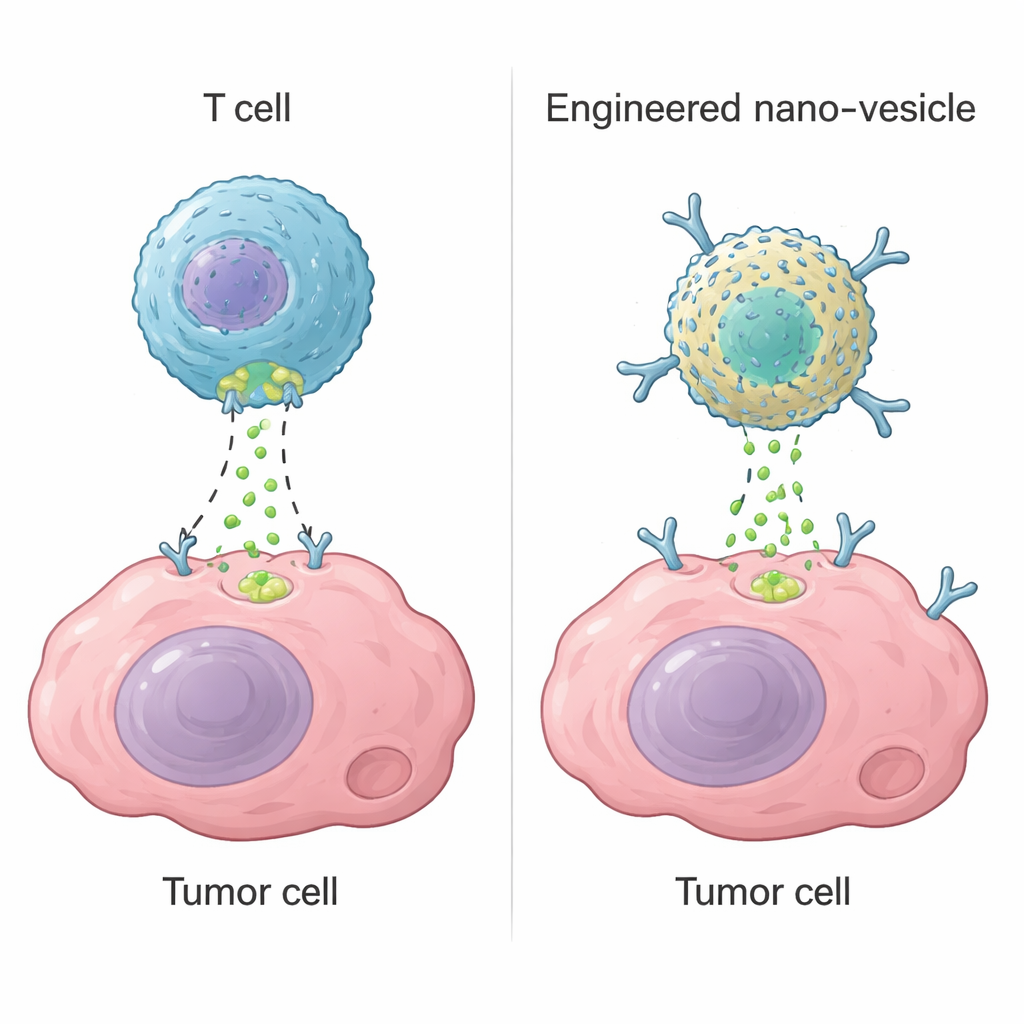
Cytotoxic T cells, a type of white blood cell, kill dangerous cells using an enzyme called granzyme B. Once they recognize a target, they punch small holes in its membrane and inject granzyme B, which switches on suicide programs inside the cell. This strategy underlies modern therapies such as CAR-T cells, which can cure some blood cancers. However, in solid tumors, CAR-T cells often fail because they cannot reach all tumor regions, become exhausted, or hit healthy tissues by mistake. The authors asked whether they could build a non-living, nanoscale device that reproduces the key function of granzyme B—flipping on the cell’s self-destruct machinery—without relying on living T cells.
Designing a Tiny Artificial Enzyme
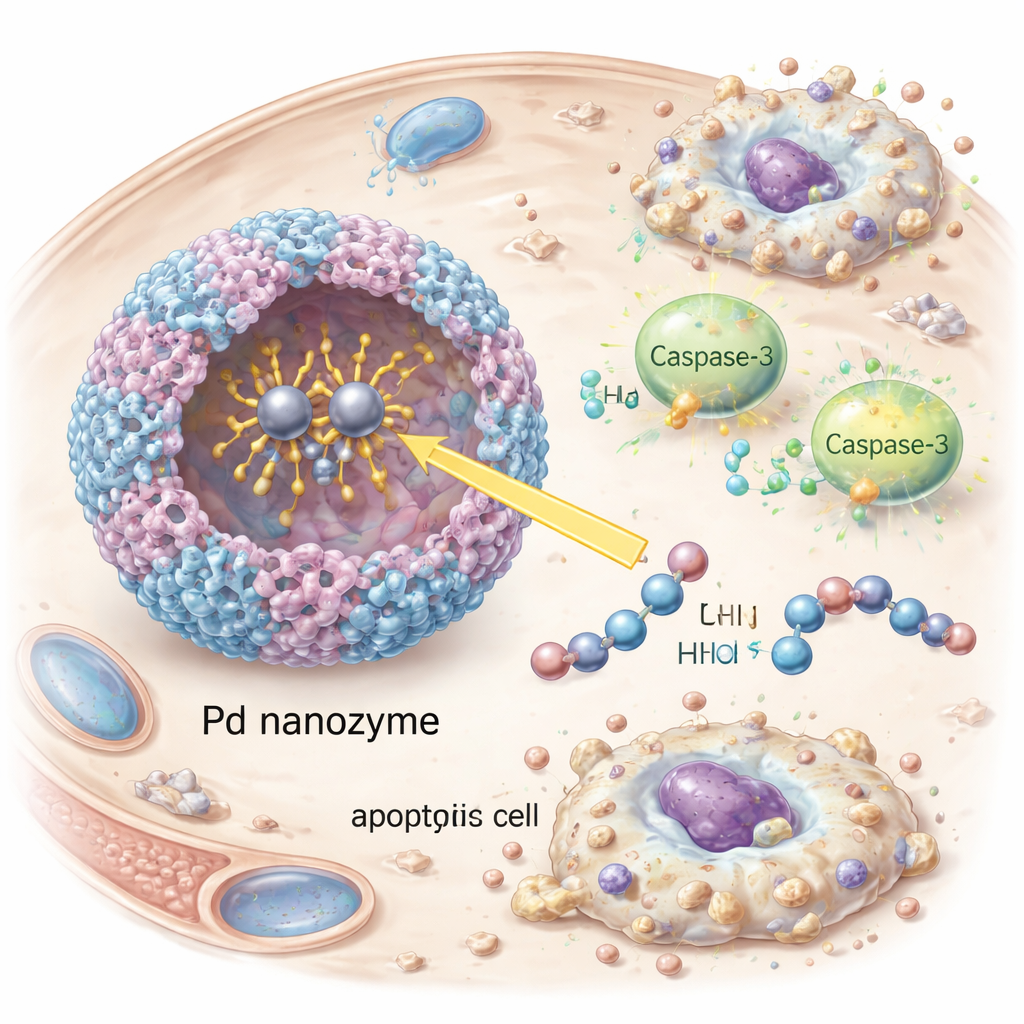
The team started from ferritin, a natural protein that forms hollow nanocages and is easy to produce in large amounts. By binding palladium ions to human ferritin, they created a “nanozyme” with enzyme-like behavior. Careful structural studies revealed that two palladium atoms nestle into a specific pocket on the protein surface, held in place by sulfur- and nitrogen-containing amino acids and water molecules. This binuclear metal center acts as an artificial cutting site that recognizes the same short sequence used by granzyme B on a crucial target known as caspase-3. In lab tests, the nanozyme efficiently clipped caspase-3 at the correct position, activating it, while ignoring closely related proteins. The artificial enzyme was slightly less efficient than natural granzyme B but notably more stable across a wide range of temperatures and acidity.
Smuggling the Nanozyme into Tumor Cells
Enzymes like granzyme B only work if they reach the inside of tumor cells. To achieve this, the researchers packaged their palladium–ferritin nanozyme inside lipid nanovesicles—small, soft bubbles made from fat-like molecules. They then coated these vesicles with fragments of antibodies that recognize HER2, a protein often overproduced on breast and bladder cancer cells. When these camouflaged vesicles encounter HER2-rich cells, they bind tightly and fuse with the cell membrane, allowing the nanozyme cargo to slip straight into the cytoplasm instead of being trapped and digested in cellular recycling compartments. Microscopy showed the vesicles lingering at the cell surface while the nanozyme diffused into the interior, setting the stage for controlled self-destruction from within.
Triggering Cancer Cell Suicide with Precision
Once inside cancer cells, the nanozyme switched on caspase-3, a central executioner of programmed cell death. The researchers observed the appearance of activated caspase-3 and its downstream target PARP, as well as clear signs of apoptosis—cells shrinking and fragmenting—in cultured HER2-positive tumor cells. Silencing caspase-3 with RNA interference dramatically reduced cell death, confirming that the effect depended on this pathway rather than on non-specific damage. In mice bearing HER2-positive tumors, the antibody-decorated vesicles circulated longer in the bloodstream, accumulated more strongly in tumors, and slowed tumor growth more effectively than vesicles lacking either the targeting antibodies or the nanozyme cargo. Importantly, treated animals showed no obvious weight loss, blood abnormalities, or tissue damage in major organs, suggesting a favorable safety profile in these early tests.
What This Could Mean for Future Cancer Care
For nonspecialists, the central message is that the researchers have built a tiny, non-living device that behaves like a key part of our immune system’s cancer-killing machinery. Instead of engineering living T cells, they engineered a stable nanoenzyme that activates the same suicide switch inside tumor cells and wrapped it in a smart delivery shell that homes to specific cancers. While much work remains before human use, this approach offers a flexible platform: in principle, the antibody coating could be swapped to aim at different tumor markers, and the artificial enzyme could be further optimized. The study demonstrates a promising route toward cell-free immunotherapy, where carefully designed nanomaterials, rather than living immune cells, are used to hunt and destroy solid tumors.
Citation: Hu, X., Liu, Q., Kang, H. et al. Granzyme B-mimetic nanozyme for nanovesicle targeted anticancer applications. Nat Commun 17, 2031 (2026). https://doi.org/10.1038/s41467-026-68773-x
Keywords: cancer nanomedicine, immunotherapy, granzyme B, nanozyme, HER2-positive tumors