Clear Sky Science · en
Reactive oxygen species-activated bioorthogonal chemistry in living systems enabled by boronate-caged dihydrotetrazines
Turning Cell Stress into a Precise Switch
Many diseases, from cancer to heart trouble, are marked by cells under oxidative stress, producing high levels of reactive oxygen species such as hydrogen peroxide. This study shows how chemists can harness that very stress as a built‑in switch to turn powerful therapies on only where they are needed. By designing drug components that react only in these stressed environments, the researchers aim to kill tumor cells or destroy harmful proteins while sparing healthy tissues.
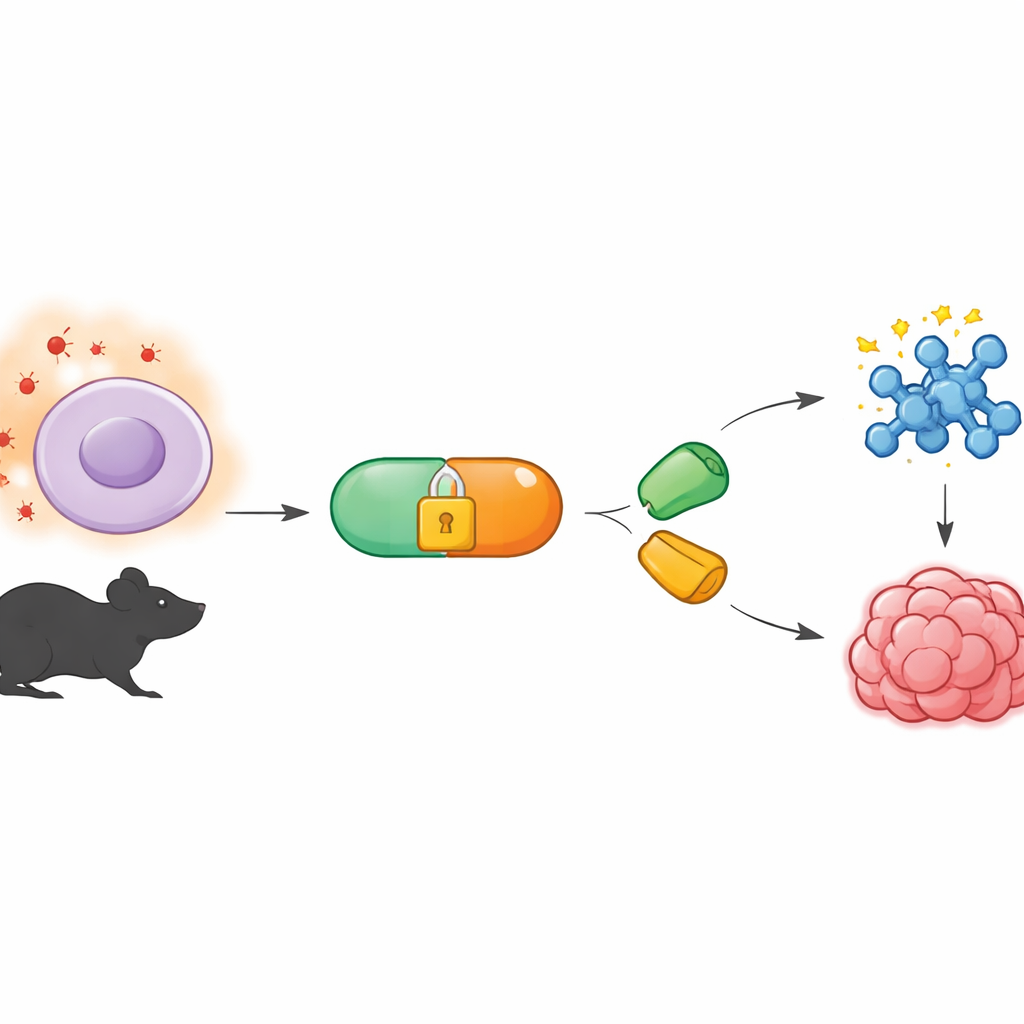
A Chemical Tool That Works Quietly in the Background
The work builds on a field called bioorthogonal chemistry, which develops reactions that can run inside living organisms without disturbing normal biology. A popular version uses special pairs of molecules that snap together like puzzle pieces, even in the crowded interior of a cell. One partner, called a tetrazine, reacts very quickly with a matching “ring” partner, known as a dienophile. Until now, many of these reactions have been controlled by shining light or adding enzymes. The authors instead wanted a system that listens to the body’s own chemical signals—specifically, the unusually high levels of reactive oxygen species found in tumors and other diseased tissues.
Building a ROS-Sensitive Chemical Trigger
To do this, the team created tetrazine precursors they call BTz, which are chemically “caged” so that they remain quiet in normal tissues. The cage is built from a boronate group linked to a reduced form of tetrazine. When hydrogen peroxide is scarce, BTz is stable in water and in cell culture for many hours. But when hydrogen peroxide is abundant—at levels similar to those measured in tumors—the boronate group is oxidized and falls apart through a self‑destruct sequence, revealing the active tetrazine. In test‑tube experiments, raising hydrogen peroxide concentrations stepwise led to a matching increase in the amount of active tetrazine generated, confirming that the reaction is both selective and strongly dose‑dependent.
Using Cell Stress to Destroy a Cancer-Driving Protein
The researchers first used their switch to control a modern drug strategy called targeted protein degradation. They attached BTz to thalidomide, a small molecule that can recruit an enzyme complex responsible for labeling proteins for disposal. In cancer cells, this BTz–thalidomide remained dormant until hydrogen peroxide levels were raised, at which point the cage opened to form an active tetrazine. This tetrazine then snapped together with a matching partner bound to a BRD4‑binding drug, assembling a larger “chimera” that drags the cancer‑related protein BRD4 to the cell’s waste machinery. In lung and cervical cancer cell lines, BRD4 levels dropped sharply only when all parts were present and hydrogen peroxide was available; blocking reactive oxygen species prevented degradation, underscoring that the process is truly stress‑activated.
Delivering a Toxic Drug Only Where It Is Needed
Next, the team turned the same chemistry into a precise delivery system for doxorubicin, a potent but heart‑damaging chemotherapy. They linked doxorubicin to a strained ring partner so that it was locked in an inactive prodrug form. When this prodrug encountered BTz in a hydrogen‑peroxide‑rich environment, the newly formed tetrazine reacted with the ring in a “click‑to‑release” process, snapping the pieces together and ejecting free doxorubicin. In colon cancer cells grown in dishes, this two‑step system killed cells almost as effectively as the free drug, but only when hydrogen peroxide was present; without it, cells remained largely unharmed. A simpler hydrogen‑peroxide‑sensitive prodrug of doxorubicin was less effective and less selective in these tests, highlighting the advantage of separating sensing and release into two coordinated steps.
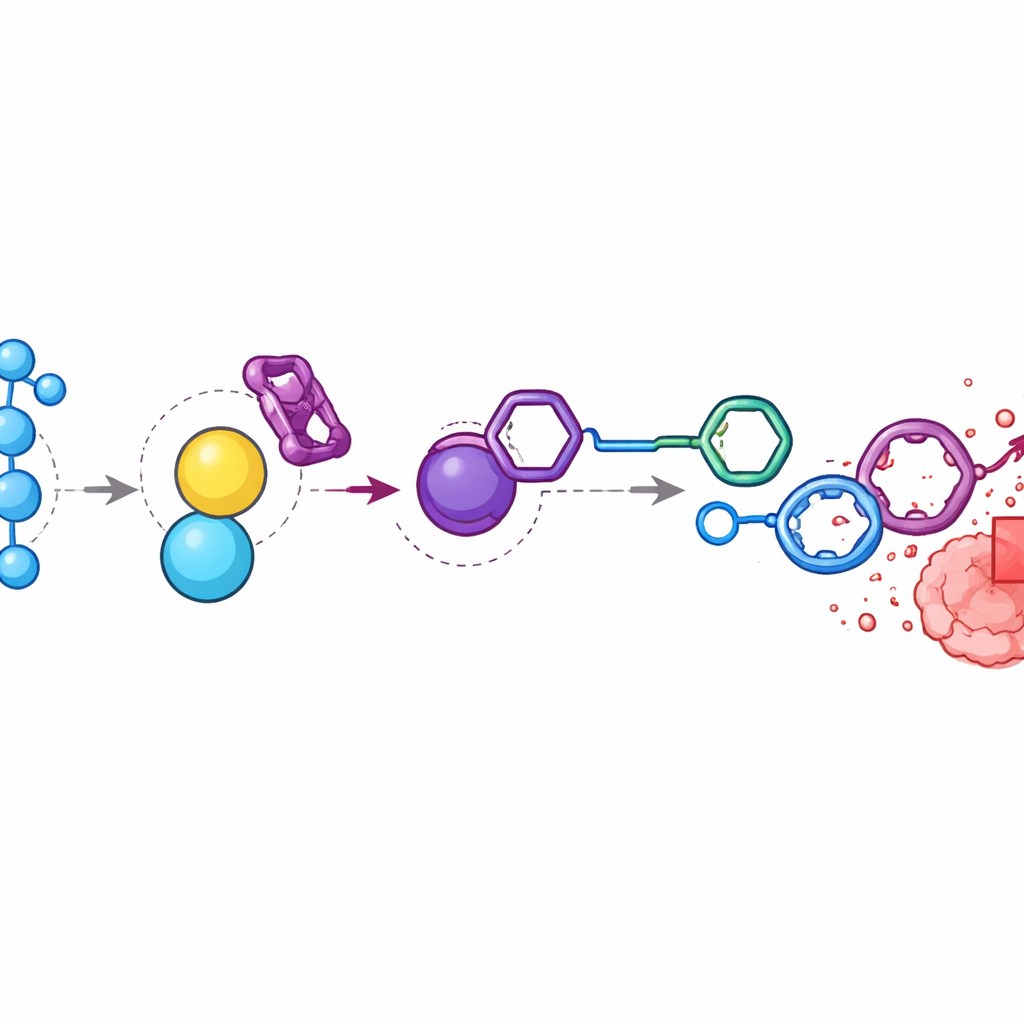
Focusing Chemotherapy Inside Tumors
In mice bearing colon tumors, the researchers found that hydrogen peroxide levels in tumors were nearly nine times higher than in surrounding muscle. They injected the doxorubicin prodrug and BTz in a timed sequence so that both accumulated near the tumor. Chemical analysis showed that the components largely disappeared from the bloodstream and organs but generated high levels of free doxorubicin inside the tumors. Compared with animals given standard doxorubicin, those treated with the activated system showed similar or better tumor shrinkage but maintained stable body weight and avoided the severe toxicity seen at higher drug doses. When the team broke the hydrogen peroxide signal by injecting catalase, an enzyme that removes hydrogen peroxide, drug release and tumor‑selective accumulation were greatly reduced, confirming that oxidative stress at the tumor site is the key driver.
From Stress Signal to Precision Therapy
Taken together, this study demonstrates a versatile chemical platform that turns a hallmark of disease—excess reactive oxygen—into a precise control knob for powerful reactions inside living systems. By keeping the reactive tetrazine handle locked until it encounters hydrogen peroxide, the approach allows researchers to build complex therapies inside the body only where they are needed, whether to dismantle a cancer‑promoting protein or to unleash a toxic drug inside a tumor. Because oxidative stress also plays roles in aging, inflammation, diabetes and neurodegenerative disorders, similar stress‑responsive reactions could ultimately be adapted for many conditions where precision and safety are paramount.
Citation: Ming, D., Zhang, J., Mu, B. et al. Reactive oxygen species-activated bioorthogonal chemistry in living systems enabled by boronate-caged dihydrotetrazines. Nat Commun 17, 2568 (2026). https://doi.org/10.1038/s41467-026-68771-z
Keywords: bioorthogonal chemistry, reactive oxygen species, tetrazine ligation, targeted drug delivery, PROTAC