Clear Sky Science · en
Homotypic membrane-powered electrochemical microfluidic analysis of extracellular vesicles for precise cancer diagnosis
Turning Cancer’s Own Armor into a Diagnostic Tool
The membranes that surround cancer cells are packed with distinctive molecular “flags.” This study shows how scientists can peel off that armor, spread it onto tiny gold chips, and then use it to fish out telltale particles from blood. The result is a highly sensitive blood test that can reveal not just whether breast cancer is present, but which subtype it is—information that is crucial for choosing the right treatment.
Why Breast Cancer Subtype Matters
Breast cancer is not a single disease. Tumors are grouped into subtypes such as estrogen receptor–positive (ER+), HER2-positive, and triple-negative, each responding differently to hormone therapy, targeted drugs, or chemotherapy. Today, doctors usually determine subtype from a tissue biopsy taken directly from the tumor. That procedure is invasive, cannot be done frequently, and may miss changes that occur as the disease evolves. A blood-based test that can track these subtype signatures over time would let physicians adjust therapy earlier and more precisely.
Tiny Messengers Floating in Blood
Cancer cells constantly shed nanoscale bubbles called extracellular vesicles (EVs) into the bloodstream. EVs carry proteins, fats, and genetic material that closely resemble those of their parent cells, making them attractive candidates for “liquid biopsies.” But there is a challenge: EVs from different breast cancer subtypes can look very similar if you focus on just one or two proteins. Existing tests either target shared markers—making it hard to distinguish subtypes—or rely on complex genetic assays that are slow and can give misleading results. The authors set out to read the fuller molecular fingerprint on EV surfaces in a simple, robust way.
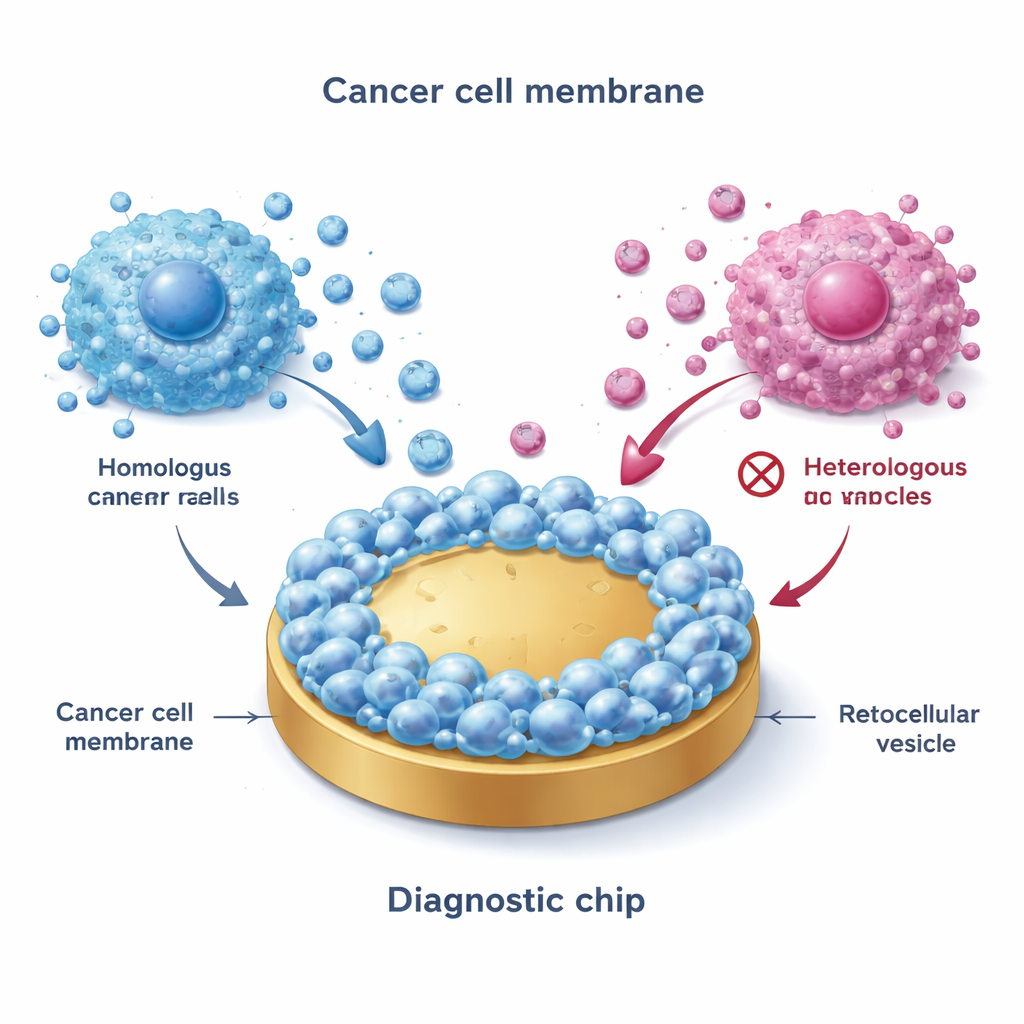
Building a Biomimetic Fishing Net
The team began by harvesting membranes from cultured breast cancer cells representing major subtypes: ER+ (such as MCF-7), HER2-positive, and triple-negative. Using a freeze–thaw process, they broke cells open, isolated the outer membranes, and then fused these membranes onto flat gold surfaces. This created a “biomimetic interface” that closely mimics the exterior of the original cancer cells. Remarkably, when EVs were flowed over these coated surfaces, only vesicles from matching, or closely related, cancer subtypes stuck strongly—a phenomenon called homotypic binding. Advanced imaging and electrical measurements confirmed that the membrane-coated surfaces were rougher, more water-loving, and far less prone to nonspecific sticking than bare gold, making them well suited for clean measurements in complex fluids like blood.
Converting Vesicle Catching into an Electrical Signal
To turn binding into a readable signal, the researchers targeted a plentiful protein called CD47 on the EV surface. They attached CD47 antibodies to silver nanoparticles using a short DNA bridge, creating tiny electroactive tags. After the membrane-coated surface captured its preferred EVs, these tags latched onto CD47 on the bound vesicles. When exposed to an acid solution and then measured electrochemically, the silver produced a sharp, easily quantifiable current. The strength of this signal rose in a straight-line fashion over a wide range of EV concentrations, down to just a few hundred vesicles per milliliter—far more sensitive than common methods such as ELISA kits or nanoparticle tracking. Each type of cancer membrane produced a strong signal only with its matching EVs, even in the presence of vesicles from other subtypes, platelets, or unrelated cells.
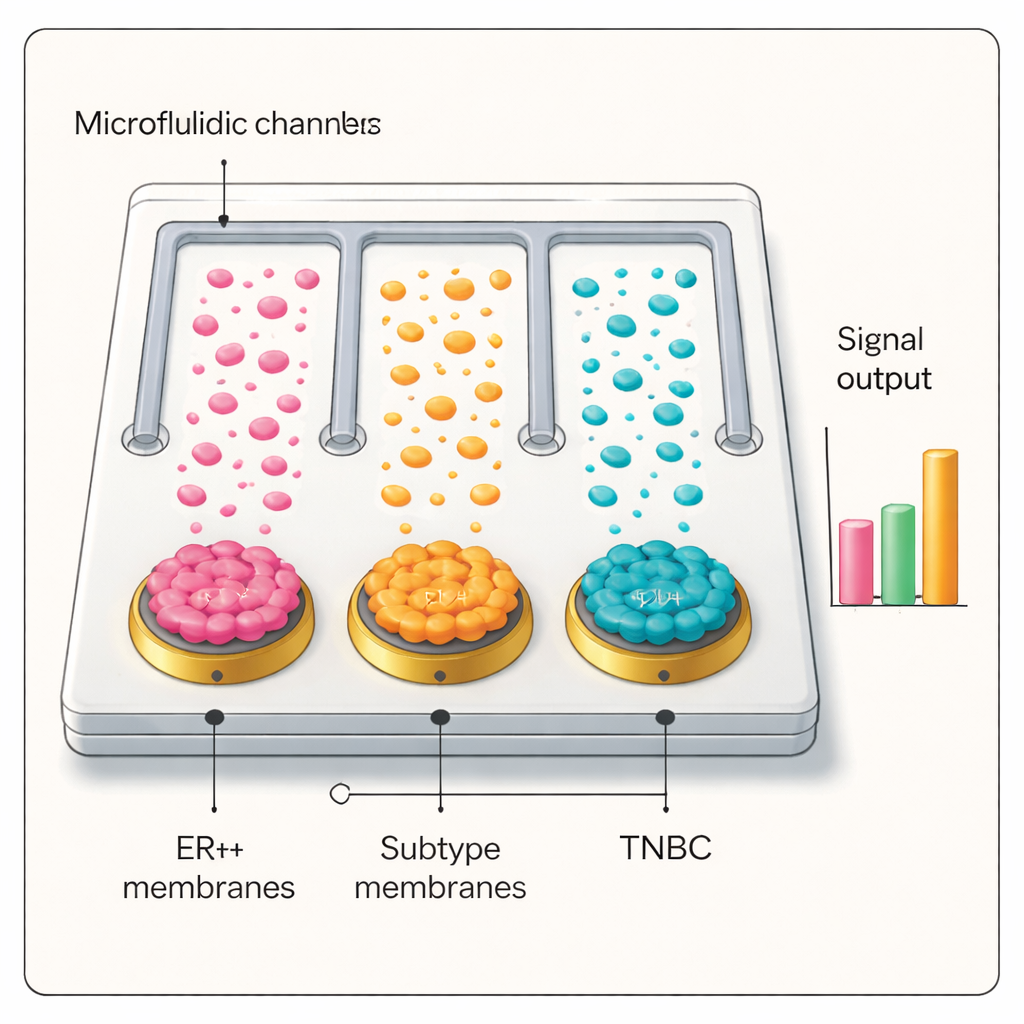
From Lab Bench to Patient Blood Samples
Armed with this selective and sensitive interface, the team built a microfluidic chip containing parallel channels, each coated with a different breast cancer cell membrane, plus an uncoated control. When plasma from patients was run through the chip and probed with the silver–antibody tags, only the channel whose membrane matched the patient’s tumor subtype produced a high electrical signal. In blinded tests with dozens of people—including ER+ and triple‑negative breast cancer patients, individuals with benign breast disease, lung cancer patients, and healthy volunteers—the platform accurately distinguished cancer from non‑cancer and correctly identified breast cancer subtypes. The diagnostic performance matched or exceeded standard pathology in many cases and showed promise for staging disease and tracking its progression.
What This Could Mean for Future Cancer Care
In simple terms, the researchers have turned cancer’s own outer shell into a highly selective bait for the tiny particles that tumors release into blood. By reading the electrical signature of which channel “lights up,” doctors could one day determine not only whether cancer is present but which kind it is, using only a blood sample. While the system still needs further refinement—such as adding more markers and more membrane types, and integrating smart data analysis—it points toward a future in which routine, minimally invasive blood tests help guide personalized cancer treatment and monitor how tumors change over time.
Citation: Zou, Z., Jin, X., Yu, X. et al. Homotypic membrane-powered electrochemical microfluidic analysis of extracellular vesicles for precise cancer diagnosis. Nat Commun 17, 1953 (2026). https://doi.org/10.1038/s41467-026-68770-0
Keywords: breast cancer subtypes, liquid biopsy, extracellular vesicles, microfluidic diagnostics, cell membrane sensors