Clear Sky Science · en
A Glial Hub-and-Spoke Circuitry in C. elegans orchestrates bidirectional thermosensation
How tiny worms can teach us about feeling hot and cold
All animals, from humans to microscopic worms, must sense temperature to survive. We move away from painful heat, seek shelter from the cold, and gravitate toward comfortable climates. This study uses the tiny nematode worm Caenorhabditis elegans to reveal a surprising player in temperature sensing: not just neurons, but their support cells—glia—act as key hubs that both detect temperature and decide how the nervous system should respond.
A new role for the brain’s support cells
Glial cells are usually described as the nervous system’s caretakers, feeding neurons and keeping them healthy. In recent years, scientists have begun to suspect that glia do more than housekeeping. In this work, the authors show that a specific glial cell type in the worm’s head, called the AMsh glia, does something far more active: it directly senses both warming and cooling and then adjusts nearby neurons that control temperature-related behavior. Instead of acting as passive bystanders, these glia sit at the very front of the sensory system, interpreting environmental temperature and shaping what the animal does next.
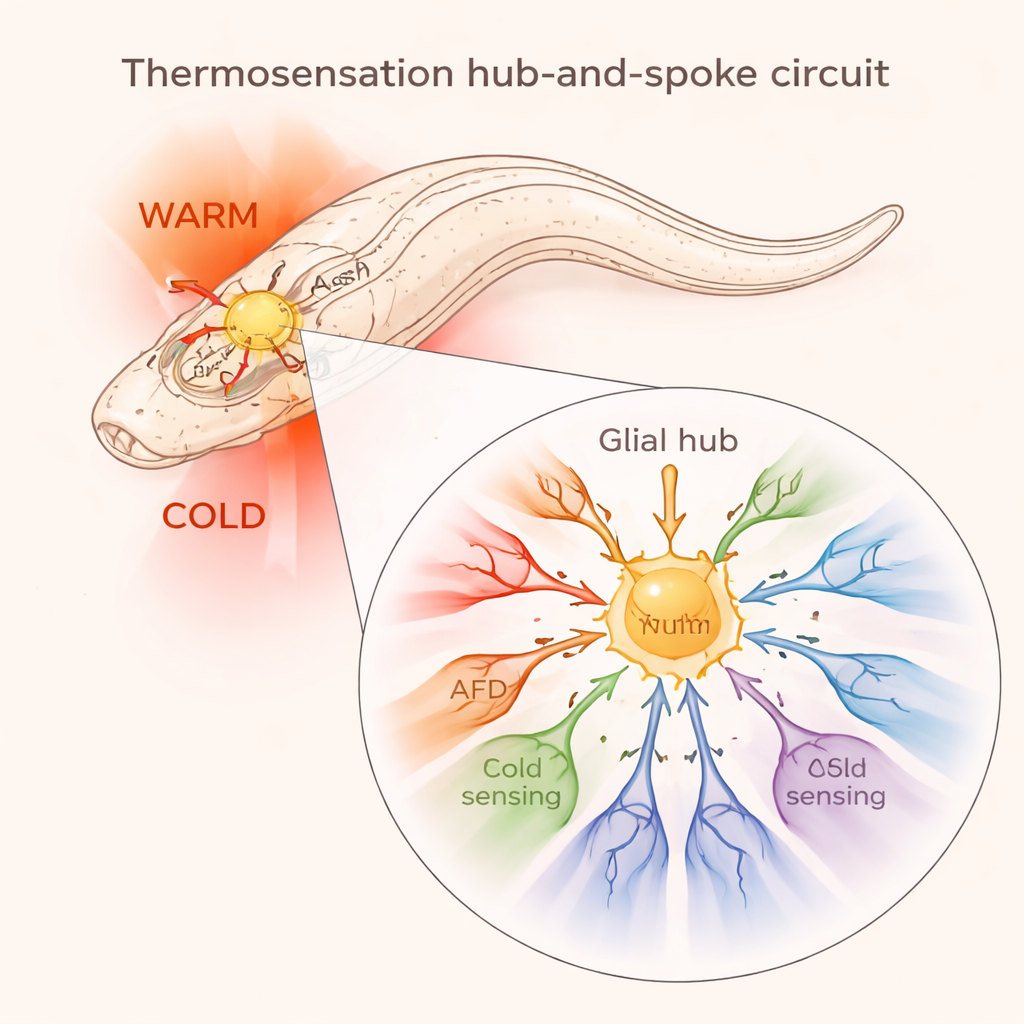
One cell that feels both heat and cold
The AMsh glial cells wrap around many temperature-sensitive neurons in the worm’s nose. Using fluorescent calcium indicators as a readout of activity, the researchers found that AMsh glia respond strongly when the environment either warms up or cools down. These signals still appeared when normal communication from neighboring neurons was blocked, and even when the glia were isolated and grown alone in a dish. That means the glia themselves can detect temperature changes without needing a neuron to tell them what is happening.
Two temperature “dials” inside a single glial hub
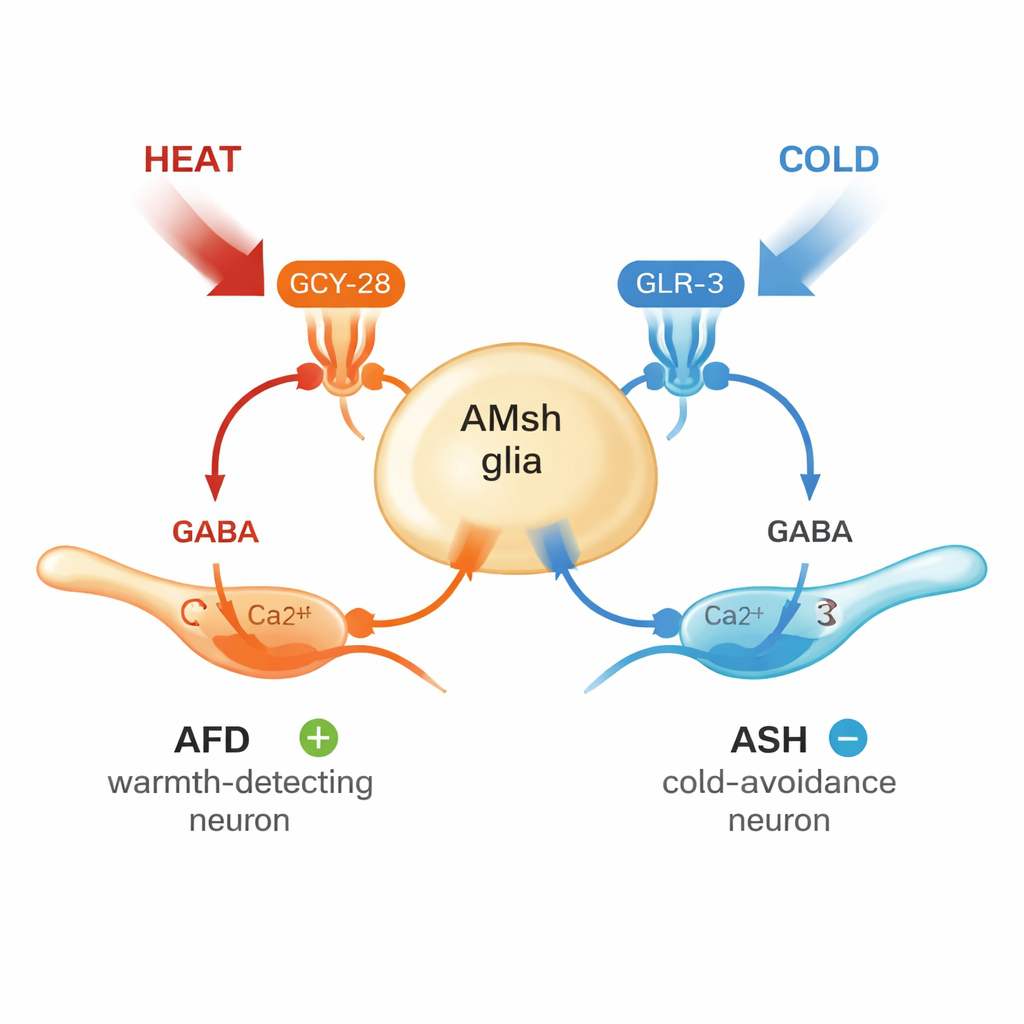
How can one type of glial cell sense both heat and cold? The team discovered that AMsh glia carry two distinct molecular sensors. For warmth, they rely on a protein called GCY-28, a guanylyl cyclase that boosts levels of a messenger molecule (cGMP) and opens ion channels, letting calcium flood into the cell. When GCY-28 was removed, the glia no longer responded to heat, and adding GCY-28 back into these glia restored the response—even when the protein was tested in human cells in a dish. For cold, the glia use a different protein, GLR-3, a glutamate receptor that, in this context, acts as a cold sensor. Loss of GLR-3 greatly weakened glial responses to cooling, and further experiments showed that these cold signals travel through internal calcium stores inside the cell. Together, GCY-28 and GLR-3 allow AMsh glia to act as a dual thermometer, reading both ends of the temperature scale.
Glia as traffic controllers of warm and cold behavior
Sensing temperature is only useful if it leads to the right behavior. The authors temporarily silenced AMsh glia using a chemogenetic switch that turns them off when worms encounter the molecule histamine. Worms with inactive glia were more eager to flee cold spots but less able to avoid heat and survive extreme high temperatures. They also showed altered preferences when navigating temperature gradients, changing where they chose to settle along warm–cold ramps. Looking more closely at the circuit, the team found that AMsh glia form a “hub-and-spoke” design: from this central glial hub, signals radiate out to different neurons. Warmth makes the glia release the chemical messenger GABA in a way that excites a warmth-detecting neuron called AFD, sharpening its response to heat. Cooling, by contrast, leads glia to release GABA onto a cold-avoidance neuron called ASH through a different type of receptor, damping down its activity and preventing overreactions to cold.
Why this matters beyond worms
By uncovering a single glial cell that can feel both hot and cold and then selectively boost or brake the neurons that drive behavior, this study challenges the traditional view that only neurons are true “sensors.” Instead, glia emerge as central decision-makers that weigh competing temperature cues and fine-tune the animal’s choices. Similar temperature-sensitive molecules exist in mammalian glia and skin cells, suggesting that comparable hub-and-spoke logic might help shape our own responses to climate and thermal stress. In that sense, the worm’s tiny nervous system offers a blueprint for how support cells throughout the animal kingdom may quietly govern when we seek shade, shiver, or simply stay put.
Citation: Zhu, L., Li, R., Qian, M. et al. A Glial Hub-and-Spoke Circuitry in C. elegans orchestrates bidirectional thermosensation. Nat Commun 17, 1899 (2026). https://doi.org/10.1038/s41467-026-68766-w
Keywords: thermosensation, glial cells, C. elegans, temperature preference, neural circuits