Clear Sky Science · en
Endothelial PDLIM5 promotes tip cell filopodia formation and tumor angiogenesis by regulating ACTN1/ACTN4-dependent actin bundling
How Cancer Co-Opts Blood Vessels
Cancer cannot grow beyond a tiny cluster without persuading nearby blood vessels to bring in oxygen and nutrients. This study uncovers how a little-known protein in the inner lining of blood vessels helps tumors sprout new, abnormal branches. Understanding this process could open the door to smarter treatments that not only starve tumors but also make immunotherapy work better.
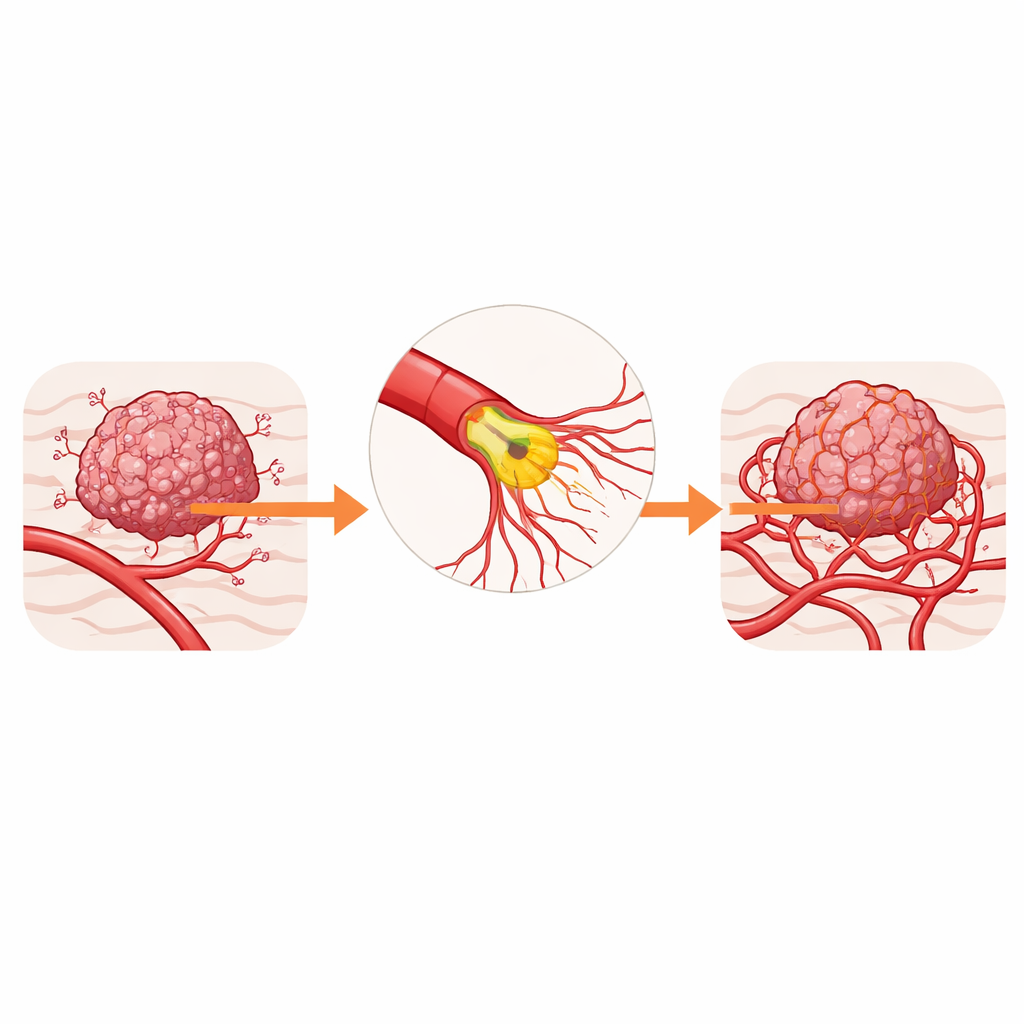
The Hidden Architects of Tumor Blood Supply
Blood vessels that feed tumors are built from specialized cells called endothelial cells, which line the vessel interior. At the growing front of each new vessel branch sits a “tip cell” that sends out long, finger-like projections known as filopodia. These projections sense chemical cues from the tumor and steer new sprouts toward areas that are starved of oxygen. In many cancers, this sprouting goes into overdrive, creating leaky, twisted vessels that worsen low-oxygen conditions and make it harder for drugs and immune cells to reach the tumor.
A Protein That Supercharges Sprouting
By examining individual cells from human lung tumors, the researchers found that tumor-associated endothelial cells strongly activate a set of genes that control the internal scaffolding of the cell, especially a protein called PDLIM5. PDLIM5 was more abundant in tumor vessels than in normal vessels, and patients whose tumor vessels expressed more PDLIM5 tended to have shorter survival. In mouse models of several cancers, boosting PDLIM5 in the vessel lining made tumors grow faster and develop denser vessel networks, suggesting that this protein acts as a powerful driver of tumor blood-vessel growth.
How PDLIM5 Builds Cellular “Feelers”
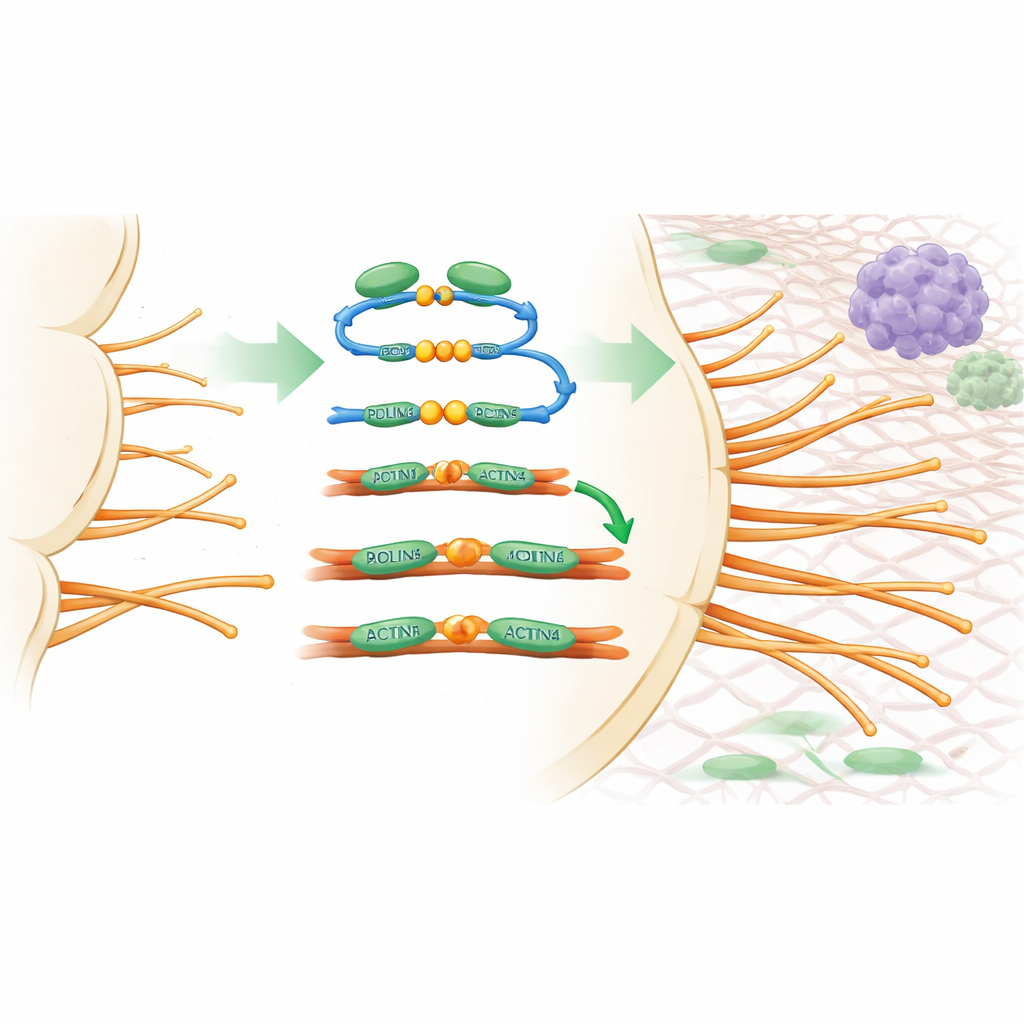
To see how PDLIM5 affects vessel behavior, the team turned to experimental systems where new sprouts can be watched in detail, such as growing blood vessels in the mouse retina and three-dimensional cultures of endothelial cells. When PDLIM5 was removed specifically from endothelial cells, sprouts advanced more slowly, had fewer tip cells, and produced many fewer filopodia. At the microscopic level, PDLIM5 was found to link two key actin-bundling proteins, ACTN1 and ACTN4, to long strands of actin that form the cell’s internal framework. This bridging action helps pack actin strands into stiff bundles that support filopodia. Mutating two critical points on PDLIM5 (named S593 and F596) broke this connection, weakened actin bundling, and prevented PDLIM5 from restoring normal sprouting, proving that this molecular handshake is essential for building the tip cell’s “feelers.”
Rewiring Tumor Vessels to Help the Immune System
Surprisingly, blocking PDLIM5 in the vessel lining did more than just slow tumor growth. Tumor blood vessels became more regular in shape, less leaky, and better covered by stabilizing support cells. As a result, oxygen delivery improved and areas of severe hypoxia shrank. These more “normalized” vessels allowed greater entry of immune T cells into the tumor. The loss of PDLIM5 also changed how adhesion molecules on endothelial cells were organized, making it easier for T cells to stick to vessel walls and squeeze through into the tumor tissue. In mouse tumors that normally resist immune checkpoint drugs, deleting PDLIM5 made treatment with an anti–PD-L1 antibody far more effective, shrinking tumors while increasing both helper and killer T cells inside them.
Why This Matters for Future Cancer Therapy
Taken together, the findings show that PDLIM5 acts as a central organizer of the tip cell’s actin scaffold, enabling robust filopodia formation and rapid vessel sprouting toward tumors. Removing or disabling PDLIM5 in endothelial cells slows the expansion of the tumor’s blood supply, straightens and stabilizes remaining vessels, relieves oxygen starvation, and welcomes immune cells into the tumor. For patients, this suggests that drugs designed to disrupt the PDLIM5–ACTN1/ACTN4 partnership could both starve cancers of new vessels and transform chaotic tumor vasculature into a network that better supports chemotherapy and immunotherapy.
Citation: Xu, Z., Shi, Y., Yang, Y. et al. Endothelial PDLIM5 promotes tip cell filopodia formation and tumor angiogenesis by regulating ACTN1/ACTN4-dependent actin bundling. Nat Commun 17, 2103 (2026). https://doi.org/10.1038/s41467-026-68765-x
Keywords: tumor angiogenesis, endothelial tip cells, PDLIM5, actin cytoskeleton, cancer immunotherapy