Clear Sky Science · en
Silencing lipid catabolism determines longevity in response to fasting
Why turning fat burning off can be a good thing
Many people think fasting is healthy because it forces the body to burn fat for fuel. This study, done in the tiny roundworm Caenorhabditis elegans, turns that idea on its head. The researchers find that the key to living longer after fasting is not how strongly cells switch fat burning on, but how precisely they switch it off once food returns. Understanding this on–off control in a simple animal could eventually shape how we design fasting and diet strategies in humans.
A simple animal with flexible metabolism
The authors use C. elegans, a microscopic worm whose metabolism and aging pathways share many features with our own. They subjected young adult worms to a 24-hour fast followed by a return to food. During fasting, worms quickly drew down their main fat stores—triglycerides packed into intestinal droplets—while preserving essential cholesterol. When food was restored, fat droplets and free fatty acid profiles rebounded to pre-fasting levels, and worms maintained normal energy (ATP) levels in their muscles. Despite this energetic stability, a single fast-and-refeed cycle extended median lifespan by about 40% and kept the animals more vigorous at older ages.
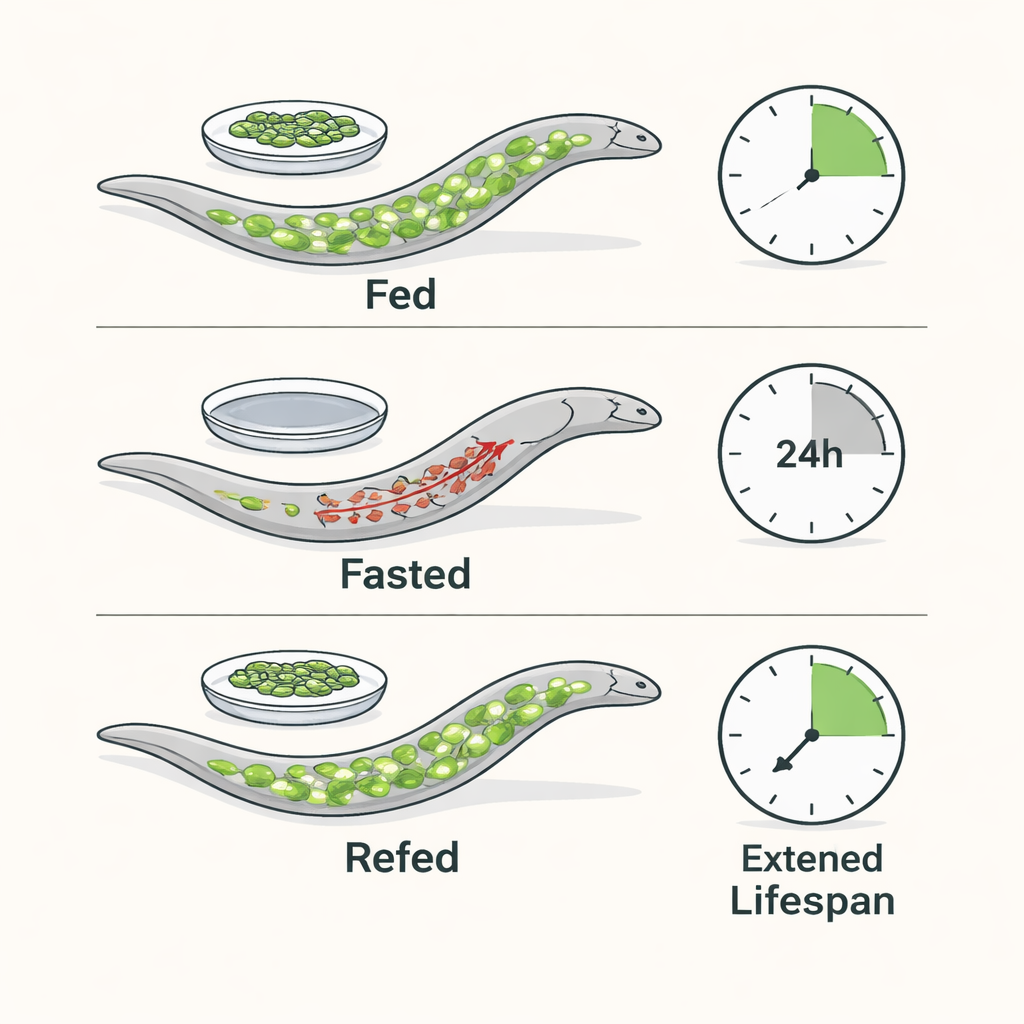
Fat burning is not required for fasting’s longevity boost
Because fasting normally pushes cells to break down fats, the team asked whether this fat burning is actually necessary for longer life. They focused on NHR-49, a nuclear hormone receptor that switches on genes for mitochondrial β-oxidation, the main fat-burning pathway. Worms lacking NHR-49 couldn’t properly ramp up these genes during fasting and showed larger drops in ATP. Yet, surprisingly, they still gained a strong lifespan extension from fasting, similar to normal worms. The same was true when the researchers blocked upstream steps in fat breakdown with genetic tools. Together, these experiments show that robust activation of lipid catabolism during the fast itself is largely dispensable for the longevity benefit.
A timing switch that shuts fat burning down
If turning fat burning on is not essential, turning it off might be. The study reveals that NHR-49 is controlled by a ligand-independent mechanism—rather than being flipped by a classical hormone-like small molecule, its activity is tuned by chemical tags added to the protein itself. During recovery from fasting, NHR-49 receives a specific pattern of phosphate groups at neighboring serine residues in a flexible “hinge” region. These negatively charged tags weaken NHR-49’s grip on DNA and promote its removal from fat-burning genes, effectively silencing them when food returns. Without this timed shutoff, cells risk staying stuck in a high-fat-burning mode that can deplete reserves and disturb long-term balance.
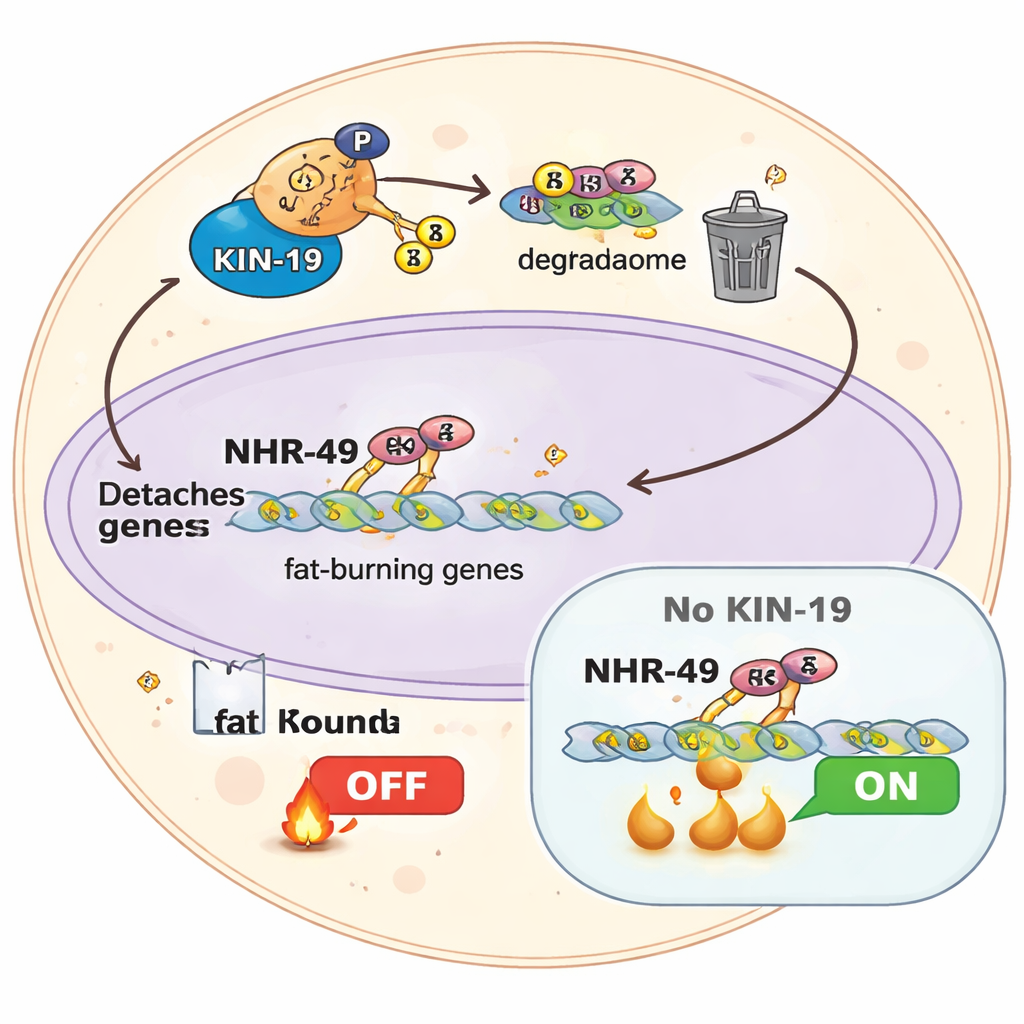
KIN-19: the enzyme that ends the fast
The authors identify a casein kinase, KIN-19, as a central player in this shutoff system. Biochemical assays show that once NHR-49 is “primed” by an initial phosphorylation, KIN-19 efficiently adds further phosphates nearby. Worms in which KIN-19 is knocked down behave as if they are permanently fasted: they are smaller, have low fat stores, altered mitochondrial shape, and reduced oxygen consumption, even when food is plentiful. Their gene expression profiles resemble a chronic fasting state, and their energy levels fail to recover properly after refeeding. Crucially, when KIN-19 is impaired, a 24-hour fast no longer extends lifespan; the longevity gain shrinks to a minimal effect. This indicates that switching off NHR-49-driven fat burning via KIN-19 is essential for fasting to promote healthy aging.
What this means for fasting and healthy aging
To a lay reader, the central message is that the body’s ability to change gears may matter more than how hard it pushes any single gear. In worms, fasting is beneficial not because they burn as much fat as possible, but because they can smoothly transition back to a storage and repair mode when food returns. The enzyme KIN-19 helps end the fat-burning phase by tagging and clearing NHR-49, preventing a damaging, never-ending “emergency” state. While humans are far more complex, this work suggests that successful fasting strategies may depend on carefully timed periods of both activation and rest in metabolism, rather than on continuous, aggressive fat loss alone.
Citation: Tatge, L., Kim, J., Solano Fonseca, R. et al. Silencing lipid catabolism determines longevity in response to fasting. Nat Commun 17, 1919 (2026). https://doi.org/10.1038/s41467-026-68764-y
Keywords: fasting and longevity, lipid metabolism, nuclear hormone receptors, metabolic flexibility, C. elegans aging