Clear Sky Science · en
A stabilized tandem antigen chimera that elicits potent malaria transmission-reducing activity
Why stopping mosquito transmission matters
Malaria vaccines now in use can greatly reduce severe disease and death in young children, yet they leave a major loophole: people who do not feel sick can still carry parasites that infect mosquitoes and keep transmission going. This study introduces a next‑generation vaccine design meant not to protect just one person, but to cut the chain of infection between humans and mosquitoes. By precisely engineering how key parasite proteins are displayed to the immune system, the researchers aim to generate antibodies that make mosquitoes poor hosts for malaria, helping communities move closer to elimination.
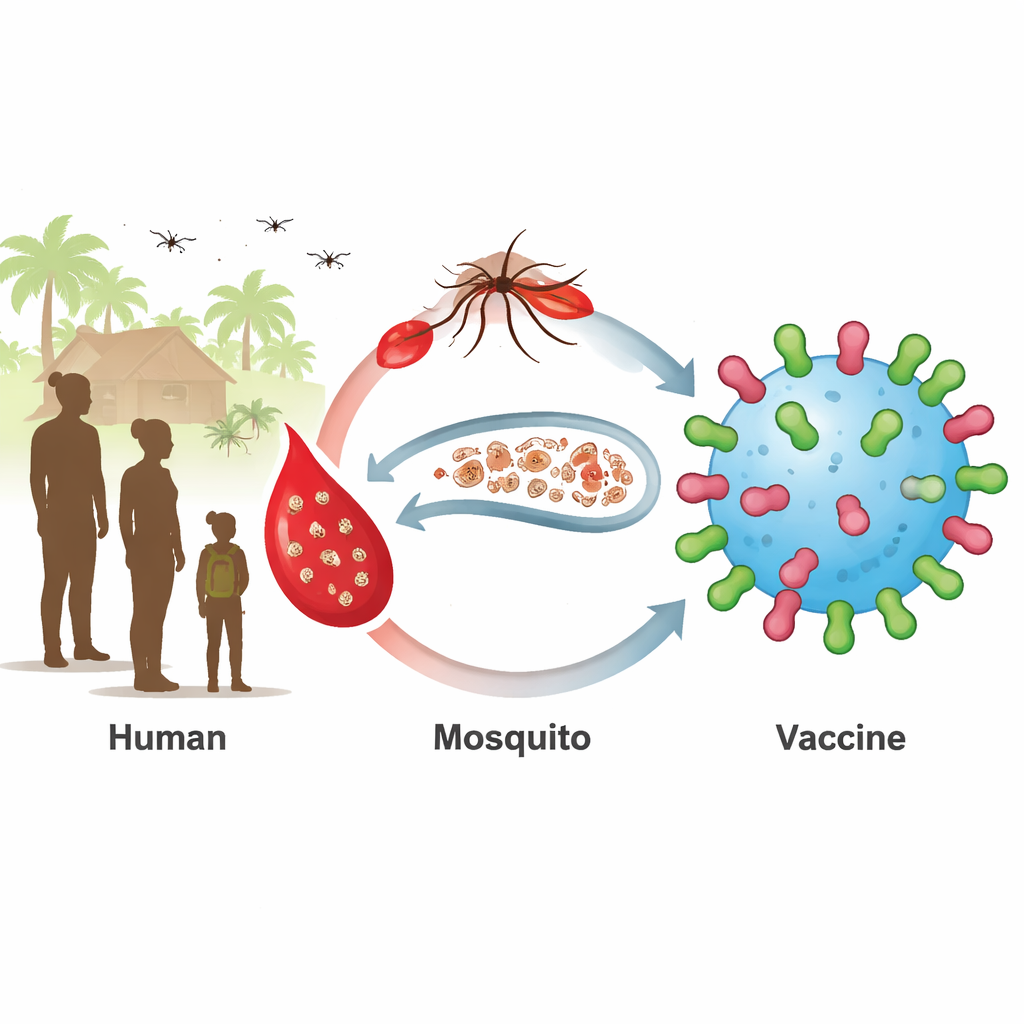
A hidden reservoir that keeps malaria alive
Current malaria vaccines focus on the early stage of infection in the liver and blood. They work best in young children, but older children and adults often harbor mature sexual‑stage parasites, called gametocytes, without symptoms. When mosquitoes bite these people, they pick up the parasites and spread them further. Two parasite surface proteins, known as Pfs230 and Pfs48/45, sit on these sexual stages and are attractive targets for so‑called transmission‑blocking vaccines. Vaccines against fragments of these proteins have entered clinical trials, but they have struggled to produce strong and lasting antibody responses in all recipients.
Building a smarter decoy for the immune system
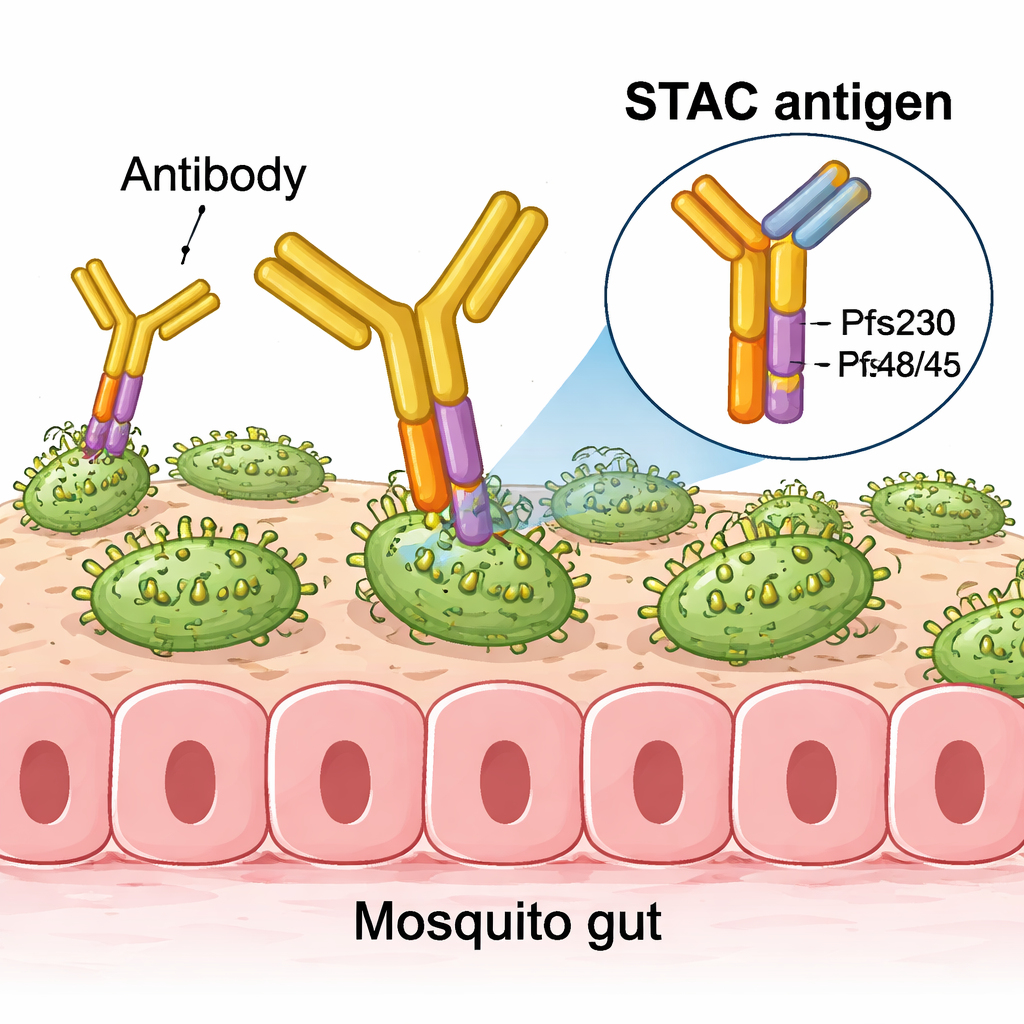
To improve on earlier designs, the authors took a structural biology approach, looking in atomic detail at how potent human antibodies bind to Pfs230 and Pfs48/45. They identified small regions, or epitopes, that are especially effective at blocking parasite development inside mosquitoes. Just as important, they mapped “non‑functional” epitopes that attract antibodies which bind but do not stop transmission—essentially wasting immune effort. Using computer modeling and protein‑engineering tools, they fused the most powerful region from Pfs230 (its first domain) to the key region from Pfs48/45 (its third domain) in a single, carefully arranged protein they call a stabilized tandem antigen chimera, or STAC.
Hiding the useless targets, keeping the useful ones
The challenge was to present these two domains together in a way that mimics how they sit on the parasite, while physically hiding the non‑functional surfaces that are normally buried in the intact protein. The team iteratively redesigned the short linker and interface between the two domains, testing each version for stability, correct folding, and whether it still bound a panel of well‑characterized antibodies. Advanced design software suggested mutations that tightened the interface and improved expression in cultured cells. Structural studies using X‑ray crystallography, small‑angle X‑ray scattering, and cryo‑electron microscopy confirmed that in the final STAC construct, the wanted epitopes are displayed with high fidelity, while the known unhelpful sites are sterically blocked.
From single molecules to powerful nanoparticles
Testing in mice showed that when STAC was attached to specialized liposomes or to self‑assembling protein nanoparticles, it triggered strong antibody responses capable of sharply reducing parasite development in standard mosquito feeding assays. At equal or lower doses, STAC often matched or outperformed vaccines that used Pfs230 and Pfs48/45 fragments separately, whether delivered alone, mixed together, or co‑displayed on the same particle. In highly sensitive tests that diluted mouse serum many fold, antibodies elicited by STAC‑decorated nanoparticles maintained transmission‑reducing activity better than those from other formulations, indicating particularly potent and focused responses.
What this could mean for malaria control
For non‑specialists, the key idea is that STAC is a custom‑built decoy that shows the immune system exactly the parts of the malaria parasite that matter most for blocking spread, while hiding distracting regions that do not help. In animal models, this design yields antibodies that make it hard for parasites to complete their life cycle in mosquitoes, even when antibody levels are relatively low. If similar results hold in humans, STAC could become a powerful partner to existing malaria vaccines: protecting vaccinated individuals from disease while also shrinking the infectious reservoir in the community. More broadly, the work showcases how detailed structural insight and protein engineering can be used to craft multi‑component vaccines that are potent, stable, and potentially cheaper to manufacture.
Citation: Ivanochko, D., Miura, K., Hailemariam, S. et al. A stabilized tandem antigen chimera that elicits potent malaria transmission-reducing activity. Nat Commun 17, 2010 (2026). https://doi.org/10.1038/s41467-026-68761-1
Keywords: malaria vaccines, transmission blocking, Pfs230, Pfs48/45, nanoparticle immunogen