Clear Sky Science · en
Essential role of NONO-HOXA1-Wnt axis in cardiomyocyte differentiation
Why tiny heart cells matter to all of us
Every heartbeat depends on billions of specialized muscle cells that form in the womb. When this process goes wrong, babies can be born with serious heart defects. This study uses human stem cells to uncover a crucial control system—built from molecules called NONO and HOXA1 and a well‑known signal called Wnt—that helps early cells commit to becoming healthy heart muscle. Understanding this hidden circuitry may eventually guide better diagnosis and treatment of congenital heart disease.
From blank slate cells to early heart builders
The researchers started with human induced pluripotent stem cells—versatile cells that can be coaxed to become almost any cell type. Using a standard recipe, they directed these cells to become cardiomyocytes, the beating cells of the heart. When the NONO gene was removed, far fewer cells successfully adopted a heart identity. Key early markers that normally appear as cells leave their primitive state and head toward the heart lineage were sharply reduced. Single‑cell RNA sequencing showed that many NONO‑deficient cells stalled halfway, stuck in an immature mesendoderm‑like state instead of progressing toward fully fledged heart muscle.
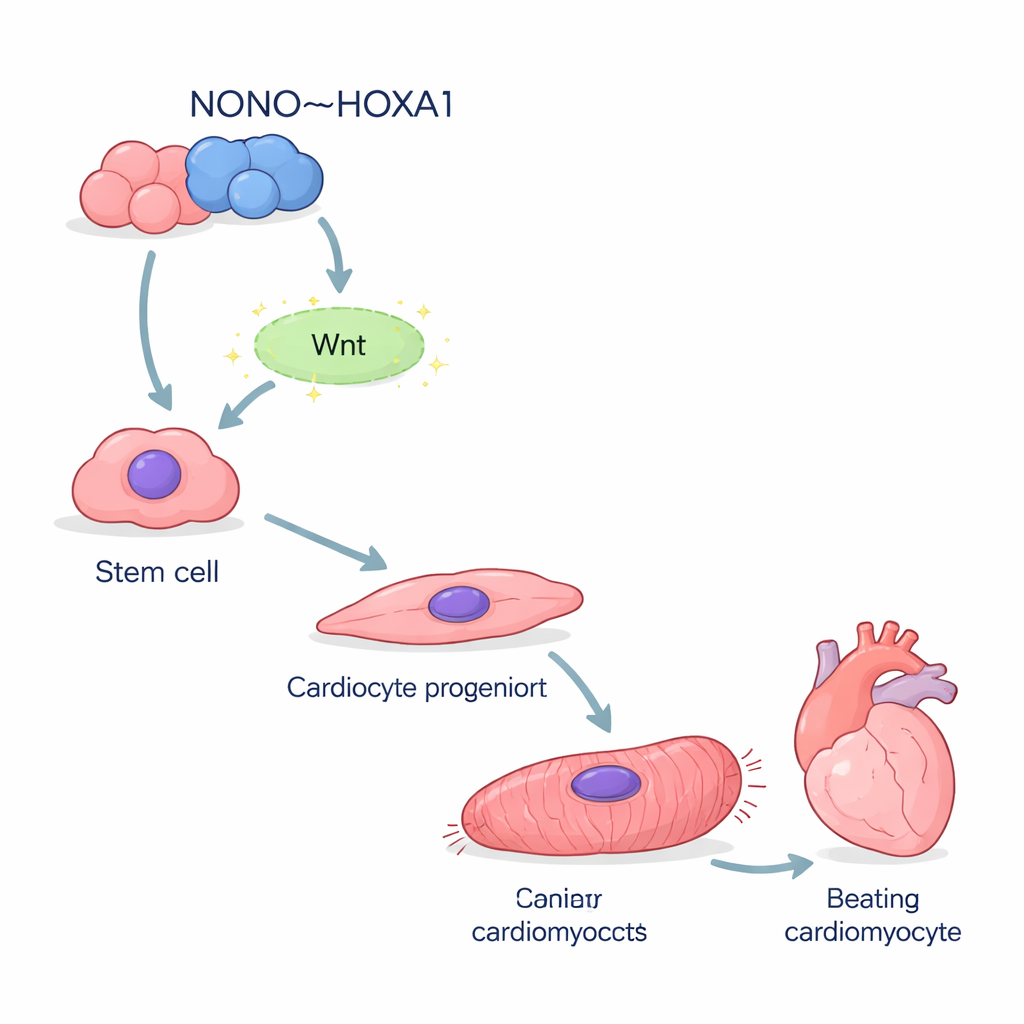
When the scaffold is missing, heart cells falter
Those cells that did become cardiomyocytes without NONO were far from healthy. They expressed much lower levels of structural proteins that form the sarcomere, the microscopic machinery that powers contraction. Under the microscope, most mutant cells showed disorganized, fragmented contractile fibers instead of the tidy stripes seen in normal cells. Electron microscopy confirmed that the usual Z‑disc “rungs” of the contractile ladder were missing or malformed. Functionally, these cells beat more slowly and irregularly. Measurements of calcium surges—electrical events that trigger each heartbeat—revealed erratic waveforms with delayed peaks and slow recovery, echoing the gene‑expression defects in calcium‑handling channels.
Rescuing development and exposing the NONO–HOXA1 partnership
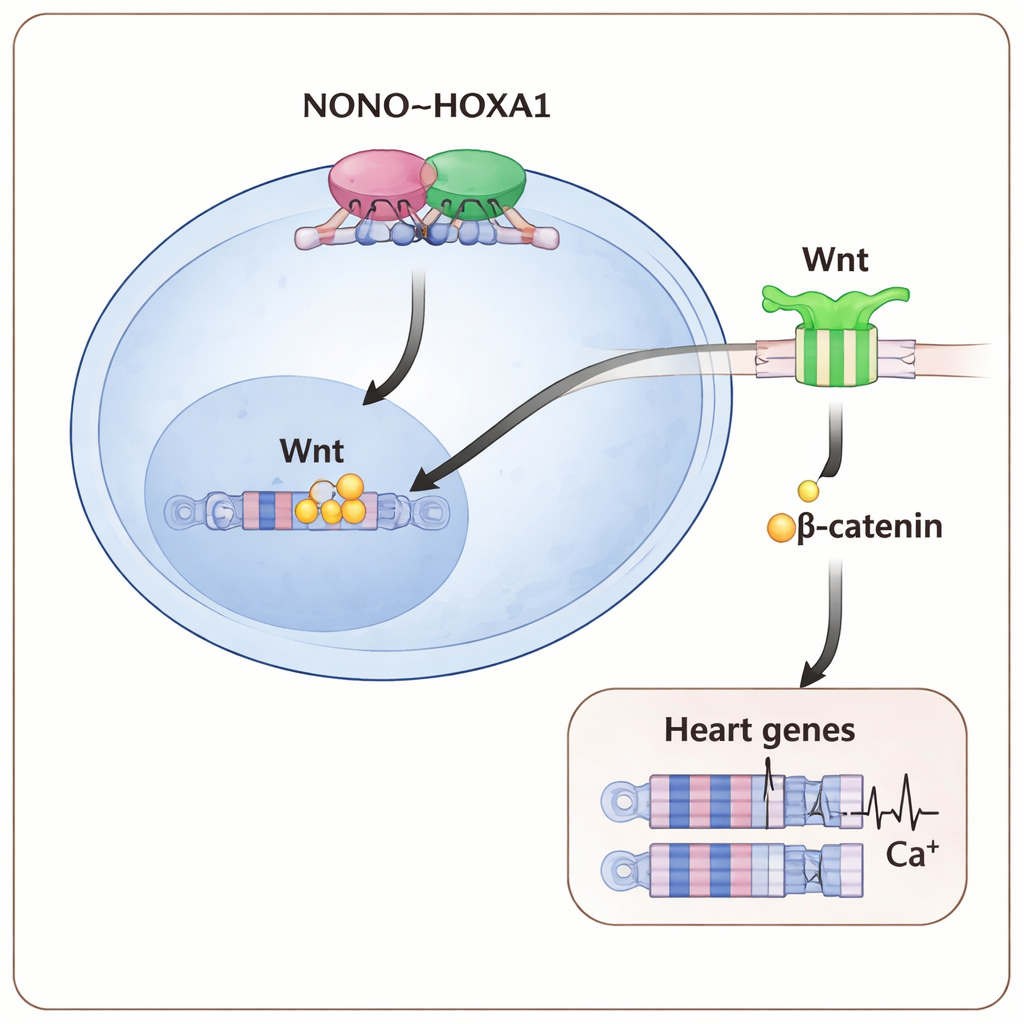
To test whether these problems truly stemmed from loss of NONO, the team re‑introduced NONO in a controlled, drug‑inducible way during the first two days of differentiation. This brief pulse of NONO largely restored the rise of early cardiac genes, increased the share of beating heart cells, and rebuilt organized sarcomeres and Z‑discs. Gene‑expression profiling confirmed that many muscle‑contraction and heart‑development genes rebounded toward normal levels. By contrast, patient‑derived mutated versions of NONO failed to rescue, supporting the idea that such variants cause disease by disrupting this early program. Protein‑interaction experiments then revealed that NONO physically binds a transcription factor called HOXA1, and that this contact stabilizes HOXA1 and helps it form the dimers needed to grip DNA.
Switching on the Wnt signal to commit to a heart fate
Mapping where these proteins land on the genome showed that NONO and HOXA1 frequently occupy the same regulatory regions, including those controlling important early heart and Wnt‑pathway genes. When NONO was missing, HOXA1’s grip on many of these sites weakened, especially at genes that drive precardiac mesoderm—the earliest heart‑forming tissue. The downstream Wnt/β‑catenin pathway, which normally helps launch mesoderm and heart development, was also blunted: less β‑catenin accumulated in the nucleus and Wnt‑responsive reporter tests fell. Strengthening Wnt signals with a higher dose of a Wnt‑activating drug during the earliest days of differentiation could partly overcome the absence of NONO, restoring more heart cells and boosting cardiac gene expression.
What this means for understanding birth defects
For non‑specialists, the central message is that a small molecular team—NONO working hand‑in‑hand with HOXA1 to fine‑tune Wnt signals—acts as an early “traffic controller” that tells young cells to become heart muscle and equips them to beat properly. When any part of this NONO–HOXA1–Wnt axis is disturbed, cells hesitate, key heart genes stay off, and the resulting cardiomyocytes are structurally and electrically fragile. These insights offer a clearer molecular explanation for why mutations in NONO are linked to congenital heart disease, and they point toward ways to manipulate this pathway in stem‑cell models or, one day, in therapies aimed at preventing or repairing heart malformations.
Citation: Feng, Z., Gao, Y., Gao, H. et al. Essential role of NONO-HOXA1-Wnt axis in cardiomyocyte differentiation. Nat Commun 17, 2013 (2026). https://doi.org/10.1038/s41467-026-68760-2
Keywords: congenital heart disease, cardiomyocyte differentiation, Wnt signaling, stem cell heart models, gene regulation