Clear Sky Science · en
Asymmetric Pt1C3-Pt1O1C3 catalytic pairs for efficient transfer hydrogenation of azobenzene
Clever Catalysts for Cleaner Chemistry
Chemists are always searching for ways to make important chemicals using less energy, fewer materials, and cleaner methods. This study introduces a new kind of ultra-efficient catalyst made from individual platinum atoms arranged in carefully spaced pairs on a sheet of carbon. These tiny structures dramatically speed up a key reaction that turns an industrial dye-like compound, azobenzene, into a more useful product, while pointing the way toward greener manufacturing of fine chemicals.
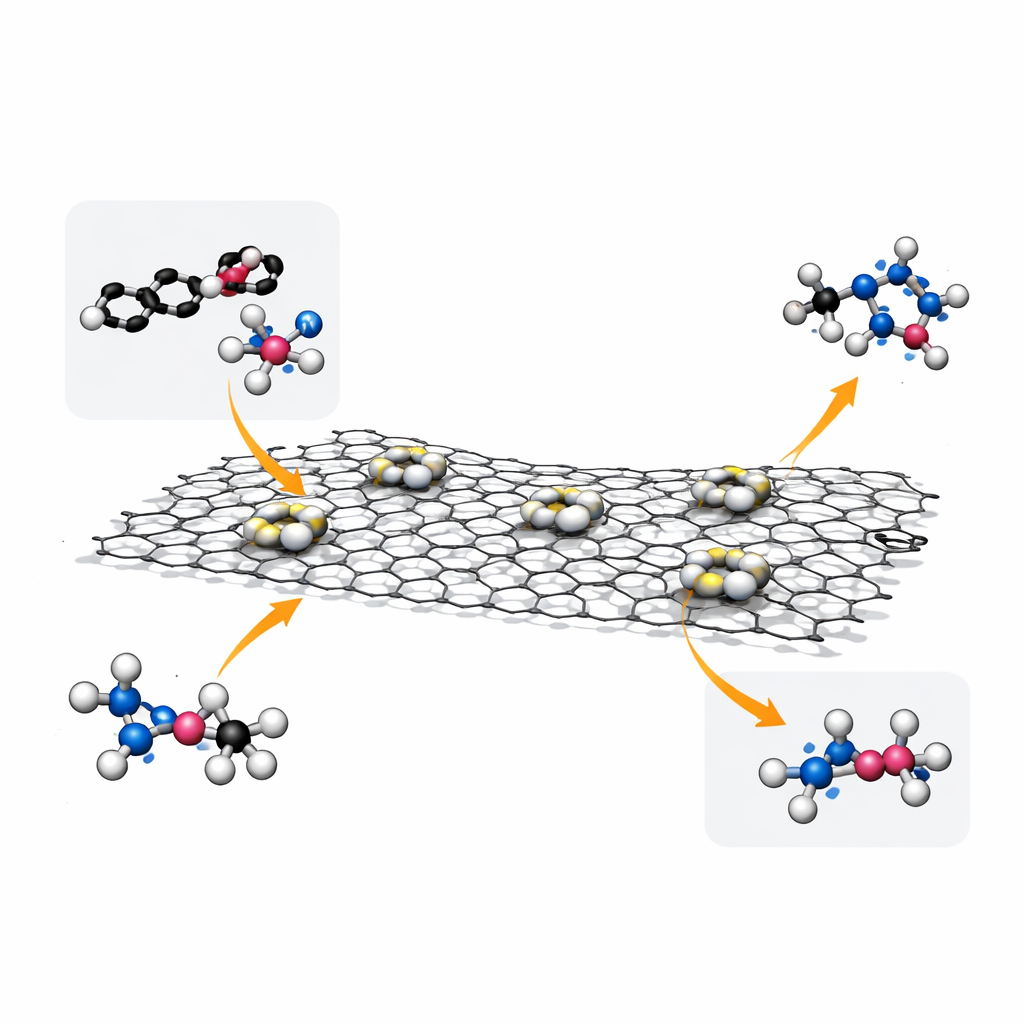
Why Tiny Metal Pairs Matter
Catalysts are substances that make chemical reactions faster without being used up, and they are central to everything from fuel cells to making medicines. In recent years, researchers have learned how to spread precious metals such as platinum as single atoms on supports, squeezing the most performance out of every costly atom. But many real-world reactions are too complex for a lone atom to handle efficiently; they work best when two nearby sites can cooperate. The team behind this work focuses on such “catalytic pairs”: two metal atoms close enough to share the workload but arranged with atomic precision so that they function better than larger particles or isolated atoms.
Designing an Atomic Double-Act
In this study, the researchers built pairs of platinum atoms anchored on reduced graphene oxide, a thin, conductive carbon sheet. Each pair is asymmetric: one platinum atom is bound to three carbon atoms, while its partner is bound to three carbons and one oxygen atom. This subtle difference changes how each platinum atom interacts with the reacting molecules. Using specialized synthesis methods, the team controlled how densely these pairs sit on the surface and, crucially, how far apart neighboring pairs are. High-resolution electron microscopy and advanced spectroscopic techniques confirmed that the platinum atoms are individually dispersed, form true pairs rather than clumps, and maintain a stable chemical state during operation.
Finding the Sweet Spot for Speed
The catalyst was tested on the transfer hydrogenation of azobenzene, a reaction in which hydrogen is supplied indirectly by a solid reagent called ammonia–borane in the presence of water. By keeping the total amount of platinum constant but changing how much was loaded onto the carbon support, the researchers altered the distance between neighboring platinum pairs. They discovered that catalytic activity did not simply rise with more metal: performance peaked when the average gap between pairs was about 5.3 angstroms (roughly half a billionth of a meter). At this spacing, the catalyst reached an exceptionally high turnover frequency—over an order of magnitude better than comparable platinum nanoparticles or single-atom systems—and remained stable over many reaction cycles. It also worked well for a variety of azobenzene derivatives, showing that the design is broadly useful rather than tailored to a single molecule.
How Shape and Spacing Control the Reaction
To understand why this precise arrangement works so well, the team used quantum mechanical simulations to model how electrons and atoms move during the reaction. The asymmetric platinum pair, together with the optimal spacing between neighboring pairs, tunes the electronic structure of the metal atoms in such a way that both azobenzene and ammonia–borane can sit comfortably on the surface at the same time without sticking too strongly. The calculations reveal a stepwise path in which hydrogen is shuttled from ammonia–borane, through the platinum pair and nearby carbon and oxygen atoms, and onto the nitrogen–nitrogen bond in azobenzene. If both platinum atoms are of the same type, or if they are arranged too close together or too far apart, hydrogen either clings too tightly, cannot move efficiently, or the reactants fail to adsorb properly, all of which slow the reaction.
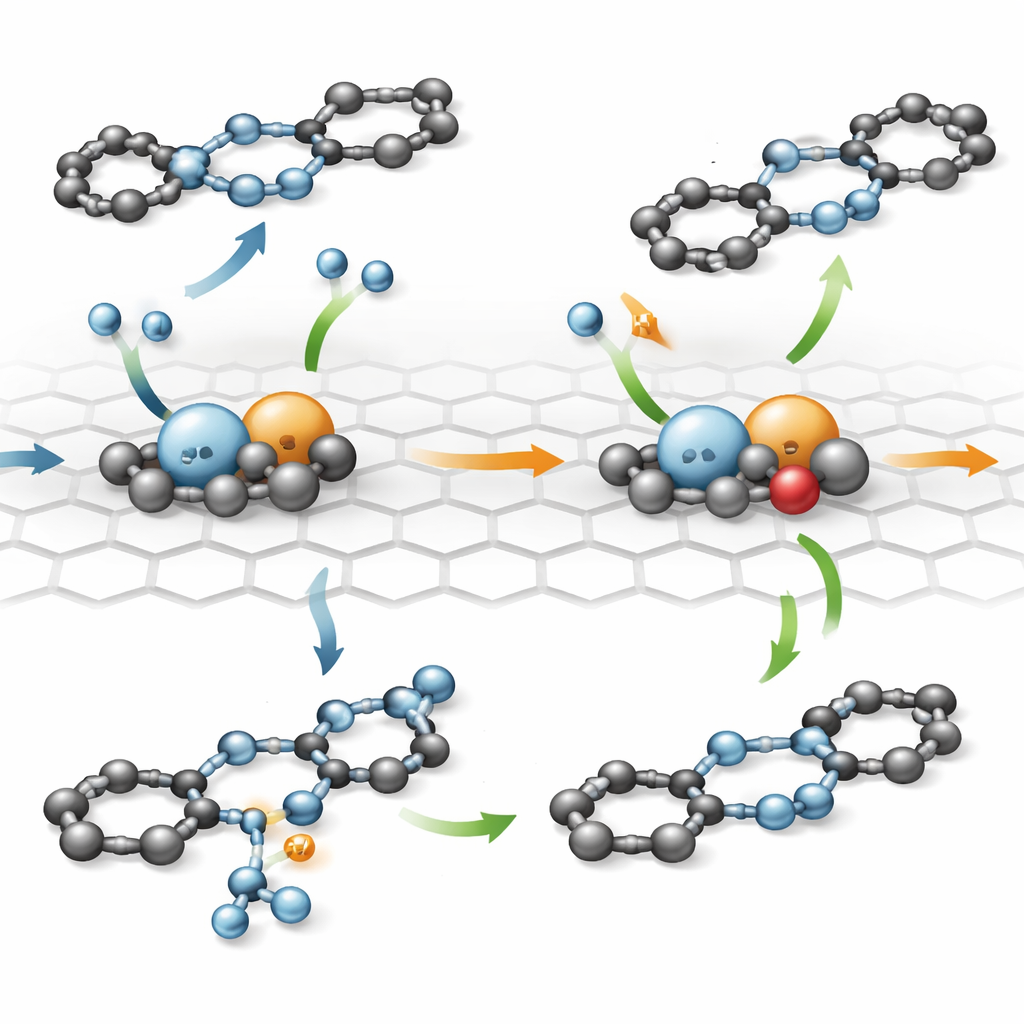
What This Means for Future Green Chemistry
The work shows that not just the choice of metal, but the exact local environment and spacing of atomically precise pairs, can make or break a catalyst’s performance. By engineering an uneven platinum duo at just the right separation, the researchers achieved fast, selective, and robust hydrogenation of azobenzene using a convenient hydrogen source. For non-specialists, the key message is that controlling matter at the level of individual atoms is becoming a practical tool for cleaner, more efficient chemical manufacturing, potentially reducing waste and energy use in the production of dyes, pharmaceuticals, and other fine chemicals.
Citation: Fang, Y., Zhao, W., Xing, Z. et al. Asymmetric Pt1C3-Pt1O1C3 catalytic pairs for efficient transfer hydrogenation of azobenzene. Nat Commun 17, 2239 (2026). https://doi.org/10.1038/s41467-026-68759-9
Keywords: atomic catalysts, platinum pairs, green chemistry, hydrogenation, graphene-supported catalysts