Clear Sky Science · en
Electrographic cue-reactivity co-localizes with accumbens deep brain stimulation in a case of opioid use disorder
Why this story matters
Opioid addiction remains a deadly epidemic, and many people continue to struggle with powerful drug cravings despite medications and counseling. This article tells the story of one young man whose treatment team used a brain implant not just to stimulate his brain, but also to listen to it. By finding a specific electrical signal linked to his opioid cravings, they were able to fine‑tune his deep brain stimulation so that it worked better, with less power, and helped him stay drug‑free.
Listening in on craving
The patient was a 25‑year‑old man with a long history of severe opioid addiction, beginning in early adolescence, along with depression, anxiety, and other complications. Standard treatments, including very high doses of methadone, had not kept his cravings under control. Years earlier he had received deep brain stimulation (DBS) to a region deep in the brain called the nucleus accumbens, a key hub in motivation and reward. With this stimulation on, his cravings dropped and he remained abstinent from opioids for several years. When a surgical complication forced doctors to remove his battery, however, his cravings surged back within weeks, and his risk of relapse increased sharply. This set the stage for a rare opportunity: his brain electrode could be temporarily connected to external recording equipment, allowing researchers to directly measure brain activity while he experienced craving.
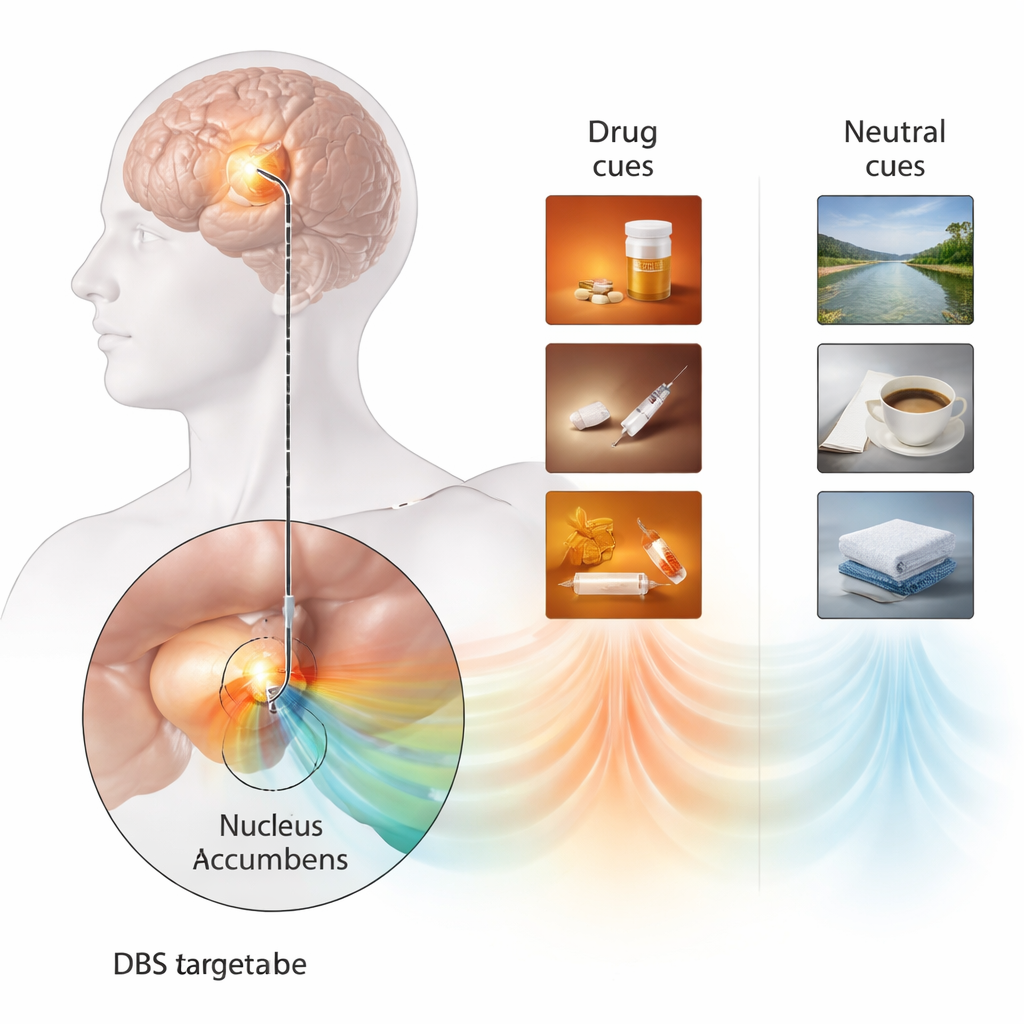
Personalized triggers reveal a brain signal
To capture what craving looks like in the brain, the team built a highly personalized cue‑reactivity task. Instead of generic drug images, they created short videos that closely matched his own past drug use—his preferred opioid, injection tools, and preparation rituals—and contrasted them with calm nature scenes. After each six‑second clip, he rated how strong his urge to use opioids felt. At the same time, the DBS lead in his nucleus accumbens was used like a tiny microphone, recording the local electrical rhythms of his brain. When the drug videos played, a distinctive low‑frequency rhythm (in the 1–6 Hz range) appeared in the ventral part of the nucleus accumbens, an area thought to correspond to the “shell” subregion. This signal was much weaker for neutral videos and was not seen in other parts of the electrode. Importantly, higher power in this rhythm lined up with higher self‑reported craving, suggesting it was a direct brain marker of drug‑cue reactivity rather than a general signal of arousal or attention.
Turning stimulation into a targeted tool
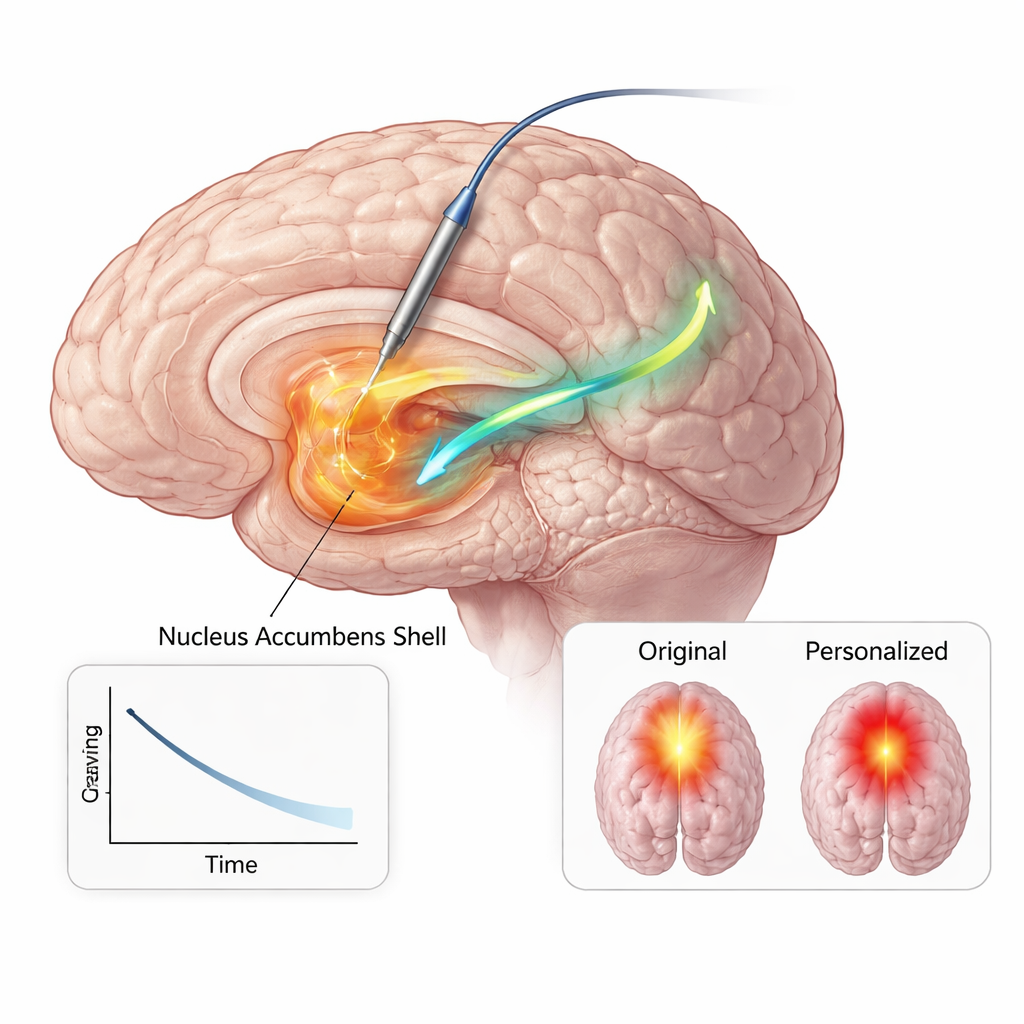
The crucial next question was whether stimulation at that very spot could dampen the craving‑related brain rhythm and the urge it reflected. The team systematically tested different electrode contact pairs, asking in real time about cravings, mood, and side effects while gradually increasing the stimulation strength. The ventral contact pair sitting in the nucleus accumbens shell emerged as the sweet spot: it produced strong craving relief and mood improvement at relatively low current, with minimal side effects. More dorsal contact pairs, in a nearby fiber bundle called the anterior limb of the internal capsule, also helped but required much stronger stimulation and produced uncomfortable sensations. Guided by these tests, the researchers then repeated the cue‑reactivity task while delivering short bouts of nucleus accumbens shell stimulation before each video. Under this “stimulated” condition, the previously elevated 1–6 Hz rhythm during drug cues was pushed down toward neutral levels, and his average craving ratings dropped by more than half.
Lasting change with a lighter touch
Armed with this converging evidence—from anatomy, brain signals, and behavior—the clinicians permanently reprogrammed his implanted device to stimulate focally within the nucleus accumbens shell at a much lower current than before. Over the following six months, he reported sustained reductions in both the frequency and intensity of opioid cravings, maintained abstinence, and fulfilled criteria for early remission, without major changes in mood, anxiety, or energy. Brain‑network modeling showed that the new settings activated a smaller, more focused volume of tissue while engaging a key pathway between the nucleus accumbens and a region beneath the frontal lobes involved in emotion and valuation. Because the stimulation was more efficient, the battery demands were markedly reduced, an important benefit for a young patient likely to need long‑term therapy.
What this could mean for future care
This N‑of‑1 case does not prove that the same approach will work for every person with opioid addiction, but it serves as a proof of principle. It shows that in at least one individual, a specific electrical rhythm in a reward hub of the brain closely tracked opioid cravings and could be quieted by precisely targeted stimulation, leading to durable symptom relief with lower electrical dose. In simple terms, the team learned to “tune” the stimulator to the patient’s own craving signal. If similar brain markers can be found and tracked in more people, and if devices that both sense and stimulate become more widely available, future treatments might personalize brain stimulation for addiction—responding when craving circuits flare up and potentially helping people stay ahead of relapse.
Citation: Qiu, L., Nho, YH., Seilheimer, R.L. et al. Electrographic cue-reactivity co-localizes with accumbens deep brain stimulation in a case of opioid use disorder. Nat Commun 17, 1708 (2026). https://doi.org/10.1038/s41467-026-68758-w
Keywords: opioid addiction, deep brain stimulation, nucleus accumbens, brain biomarkers, drug craving