Clear Sky Science · en
A heterogeneous population code at the first synapse of vision
Why the first steps of seeing are more surprising than we thought
Every glance you take, from a road sign to a passing fish in a stream, starts with tiny cells in the eye called cones. These cells turn light into signals that the brain can understand. It might be tempting to think that cones of the same kind all behave in the same way, like identical pixels on a screen. This study shows that, in fact, even cones of a single type form a diverse team: each member handles light a little differently, and that variety helps the eye cope with the messy, changing world we actually see.
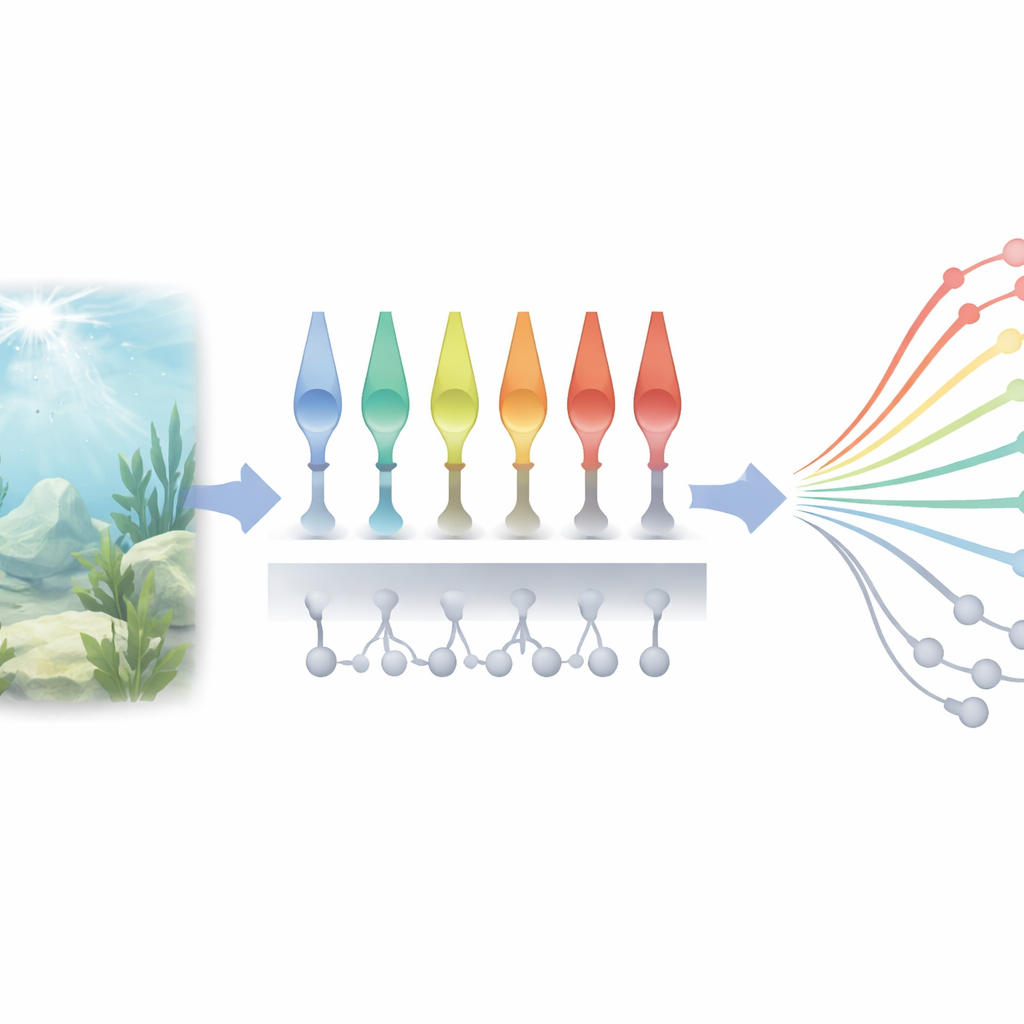
Many cones, one scene, different stories
The researchers studied a particular cone type in young zebrafish that is closely related to the red- and green-sensitive cones dominating human daytime vision. Using an advanced optical sensor that glows when the chemical messenger glutamate is released, they watched, in living animals, how individual cone endings pass on visual information to the rest of the retina. Rather than recording the electrical "input" inside the cone, they focused on the "output" at the very first synapse of vision, where the cone talks to the next cells in the circuit. This let them see directly how light is translated into a chemical signal that drives the entire visual system.
Reliable timing, but different levels of sensitivity
One of the most striking findings is that each cone is extremely reliable. When the same brief darkening of light was presented almost a hundred times, a given cone produced responses with very similar size and timing, with only a few milliseconds of variation. That means this first synapse adds remarkably little noise to the visual message. Yet when the researchers compared many cones of the same type, they discovered that their sensitivities varied widely. Some cones responded strongly to very small light changes; others needed larger changes to react. Some could track rapidly flickering light up to around twenty times per second, while others dropped out at much lower speeds. So the population was not a uniform grid of identical detectors, but more like a mixed group with different thresholds and reaction speeds.
Dark flashes, steady glow, and split channels of information
Vision is not just about whether light is present, but about how it changes over time. The team found that each cone’s output can be broken down into a fast, brief burst and a slower, more continuous component. The fast bursts were strongly biased toward sudden decreases in light – brief dark flashes – and hardly appeared for equally strong light increases. The slower component, in contrast, could represent both brightening and dimming in a more balanced way, at least for modest contrast changes. By feeding natural underwater movies through a simple model that mimicked these two components, the authors showed that the slow component preserves most of the overall scene, while the fast, dark-biased bursts selectively highlight sharp, dark edges in the foreground. In other words, the same cone can simultaneously provide a steady view of the world and a spotlight on striking dark events.
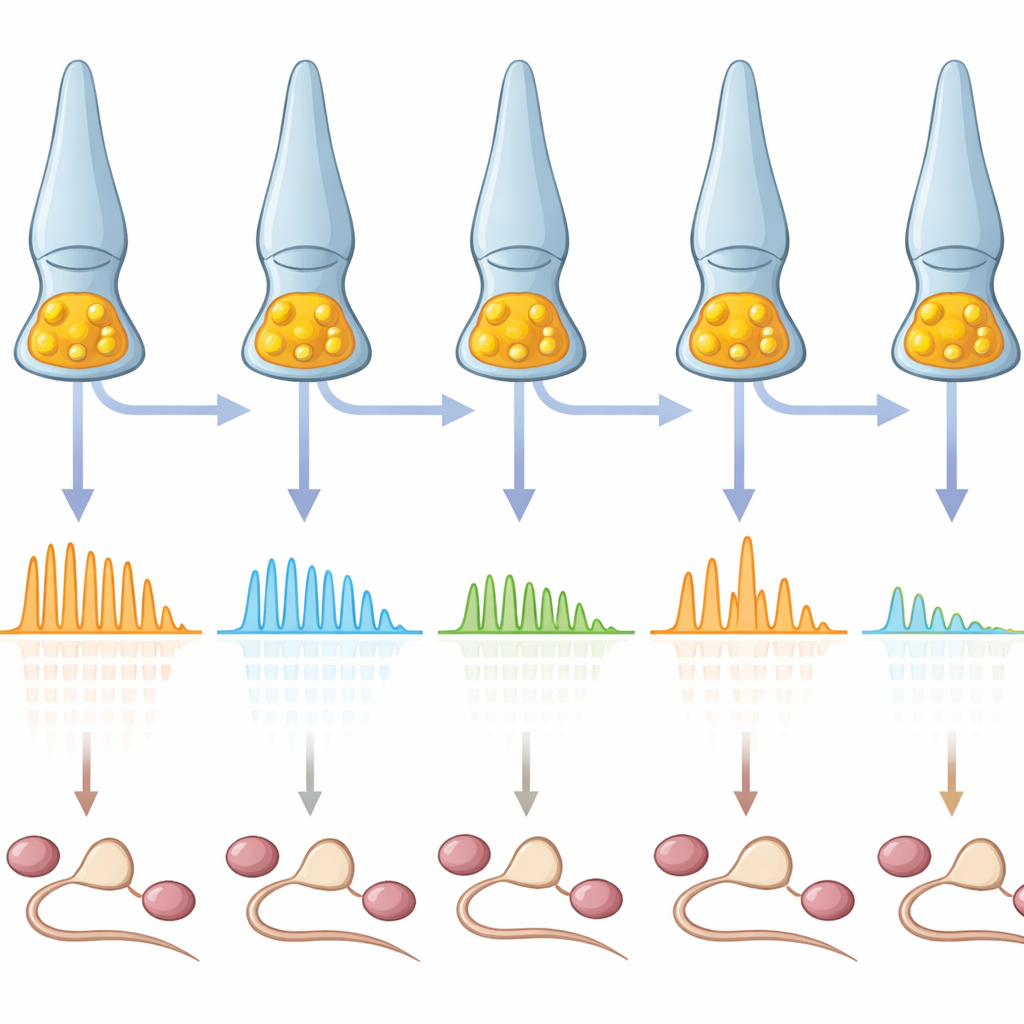
How neighbor signals and network feedback shape diversity
Where does this diversity come from? Cones do not work in isolation: they interact with a mesh of neighboring cells called horizontal cells, which send feedback onto many cones at once. When the researchers pharmacologically blocked this feedback, cones became more alike. Their responses shifted to be more strongly driven by dark changes, and their preferred flicker speeds dropped. This indicates that the surrounding network tunes each cone’s operating point—how much it is already releasing in steady light—and thereby sets how it splits its effort between bright and dark, slow and fast signals. Subtle differences in this feedback from cone to cone help create the observed spread of sensitivities and time courses, even among closely spaced neighbors that view nearly the same part of the scene.
Why a mixed team of cones helps the brain see the real world
To ask why such diversity might be useful, the authors built a simple model of downstream cells that sum signals from several cones while "swimming" through a natural zebrafish habitat. They compared two scenarios: one where all cones behaved identically, and another where cones had the measured variety of responses. The heterogeneous case consistently represented the changing contrast pattern of the natural scene better, on average by about eight percent and sometimes by much more. For a lay observer, the takeaway is that the eye does not aim for perfect uniformity at its first synapse. Instead, cones of the same apparent type form a varied ensemble that collectively stretches the range of brightness, contrast, and speed that can be encoded. This built-in diversity helps the visual system capture both the stable structure of the world and fleeting dark events that may signal nearby objects or looming threats.
Citation: Herzog, T., Yoshimatsu, T., Moya-Diaz, J. et al. A heterogeneous population code at the first synapse of vision. Nat Commun 17, 2174 (2026). https://doi.org/10.1038/s41467-026-68757-x
Keywords: photoreceptors, retina, visual coding, synaptic transmission, natural scenes