Clear Sky Science · en
The H-Pd···N-H metal-ligand dual-atom sites synergistically catalyzed alkyne semi-hydrogenation with complete Z-selectivity
Turning Tough Chemical Knots into Useful Building Blocks
Chemists often need to turn rigid, triple-bonded molecules called alkynes into more flexible double bonds, which are key pieces of many medicines and materials. The challenge is to stop the reaction at just the right point and in just the right 3D shape, without going too far. This study reports a light-driven catalyst built from single metal atoms that can stop with near-perfect precision, offering a cleaner and more efficient way to make valuable chemical ingredients.
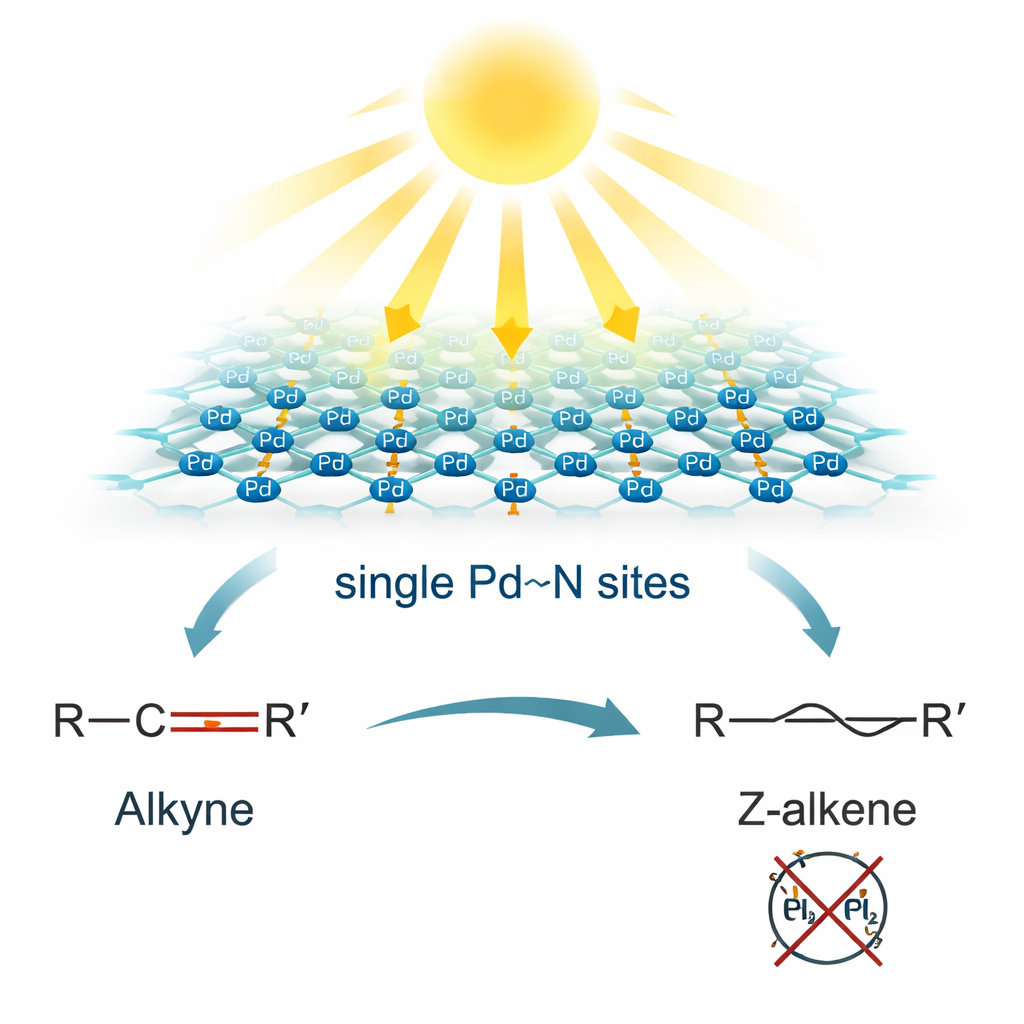
Why the Shape of a Double Bond Matters
Many drugs and bioactive molecules contain “Z-shaped” double bonds, where attached groups sit on the same side of the bond. Their mirror-like “E-shaped” counterparts, with groups on opposite sides, often behave very differently in the body—even though the two versions are almost identical in size and polarity, making them extremely hard to separate. Traditional catalysts, such as the classic Lindlar catalyst, can turn alkynes into Z-alkenes, but they almost always produce a mix of shapes and may push the reaction too far, converting useful alkenes into less useful alkanes. Industry therefore needs catalysts that not only react selectively but also stop at the Z-alkene stage every time.
Designing a Single-Atom Workbench
The researchers tackled this problem by anchoring isolated palladium atoms onto thin sheets of graphitic carbon nitride, a light-absorbing semiconductor made by heating common chemicals like urea. Using a light-driven ligand-exchange method, each palladium atom became neatly surrounded by four nitrogen atoms, forming uniform Pd(II)–N4 sites. Advanced microscopy and X-ray techniques confirmed that the metal atoms were truly dispersed one by one, not clumped into nanoparticles. Tests also showed that adding palladium improved how efficiently the material separated electric charges under light, a key requirement for any photocatalyst that aims to harvest energy from water and light to drive chemical reactions.
How Light and Dual-Atom Sites Steer the Reaction
Under blue light in water, with a common amine acting as an electron donor, these Pd–N4 sites transform into special dual-atom centers described as H–Pd···N–H. At these spots, the palladium holds a hydrogen atom ready to bond, while a neighboring nitrogen carries another hydrogen that can move as a proton. When an alkyne approaches, it inserts into the Pd–H bond, and then the nearby N–H delivers a proton directly to the same intermediate. The rigid carbon nitride framework crowds out the unwanted reaction pathway that would create the E-shaped product, so the Z-shaped alkene is formed along a lower-energy, less hindered route. Calculations support this picture, showing that the Z pathway has a significantly smaller energy barrier than the E route, and that the step where the N–H proton transfers internally is likely the slow, controlling step of the cycle.
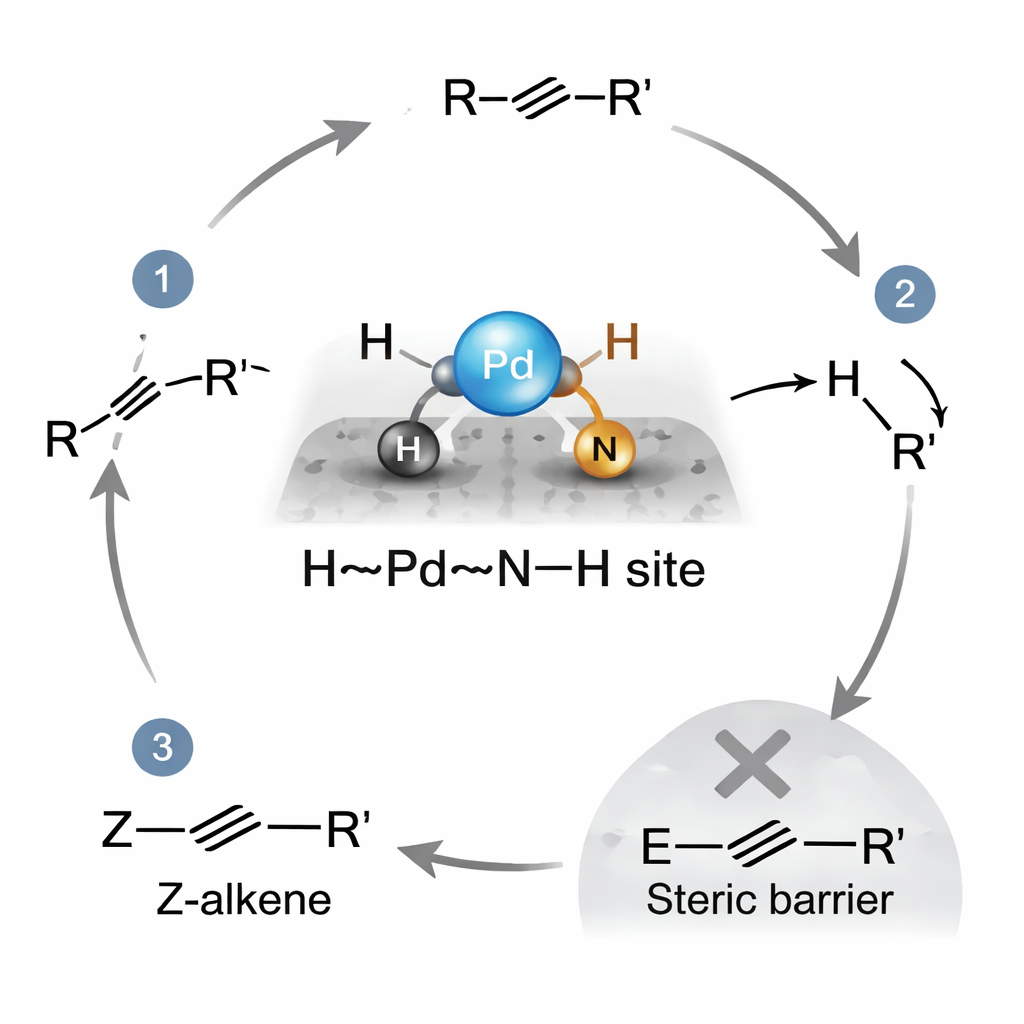
Stopping at Just the Right Point
Beyond shape control, the catalyst also avoids overreacting. Measurements of how strongly molecules stick to the surface reveal that alkynes bind much more tightly to the H–Pd···N–H sites than the resulting alkenes do. That means the starting materials are held in place for reaction, while the products are gently released before they can be further reduced to alkanes. In model reactions, a variety of internal alkynes, even those carrying fragile chemical groups like halogens, carbonyls, and amides, were converted to Z-alkenes in high yield with no detectable E-alkenes or over-hydrogenated products. Impressively, when the team treated a mixture containing only 5% alkyne impurity in 95% valuable Z-alkene, the catalyst selectively cleaned up the alkyne without damaging the desired product, illustrating a powerful tool for product purification.
What This Means for Cleaner Chemistry
This work shows that carefully designed single-atom catalysts can mimic the subtle control seen in sophisticated molecular catalysts, while retaining the robustness of solids. By pairing palladium and nitrogen in a cooperative H–Pd···N–H arrangement and embedding them in a rigid, light-harvesting scaffold, the authors achieved essentially perfect control over when the reaction stops and which 3D shape is produced. For non-specialists, the key message is that chemists are learning to build catalytic “lock-and-key” environments at the level of individual atoms, opening the door to cleaner, more selective manufacturing of pharmaceuticals and fine chemicals using light and water as gentle driving forces.
Citation: Ma, H., Wang, L., Wang, J. et al. The H-Pd···N-H metal-ligand dual-atom sites synergistically catalyzed alkyne semi-hydrogenation with complete Z-selectivity. Nat Commun 17, 1972 (2026). https://doi.org/10.1038/s41467-026-68755-z
Keywords: alkyne semi-hydrogenation, Z-alkenes, single-atom catalysis, photocatalysis, palladium carbon nitride