Clear Sky Science · en
A proteogenomic atlas of 1032 brain metastases identifies molecular subtypes, immune landscapes, and therapeutic vulnerabilities
Why cancers spread to the brain matter
As cancer care improves, more people are living long enough for their tumors to spread to new places, including the brain. These brain metastases are now a common and often devastating complication, yet doctors still treat them mostly by where the original tumor started—lung, breast, skin, colon—rather than by how the cancer has adapted to life inside the brain. This study built a detailed “atlas” of more than a thousand brain metastases to understand their hidden biology, how they interact with the brain’s own cells and immune system, and where new treatment opportunities might lie.
Building a giant map of brain tumors
The researchers collected data from 1,032 brain metastases that had arisen from many different primary cancers, along with dozens of matching primary tumors and some aggressive primary brain tumors for comparison. They combined several cutting-edge approaches: DNA and RNA sequencing, large-scale protein and metabolite measurements, single-cell and spatial mapping of tumor and immune cells, and miniature patient-derived organoids grown in the lab. By integrating all of these layers, they aimed not just to list genes, but to see how whole biological systems behave inside brain metastases.
Four recurring “personalities” of brain metastases
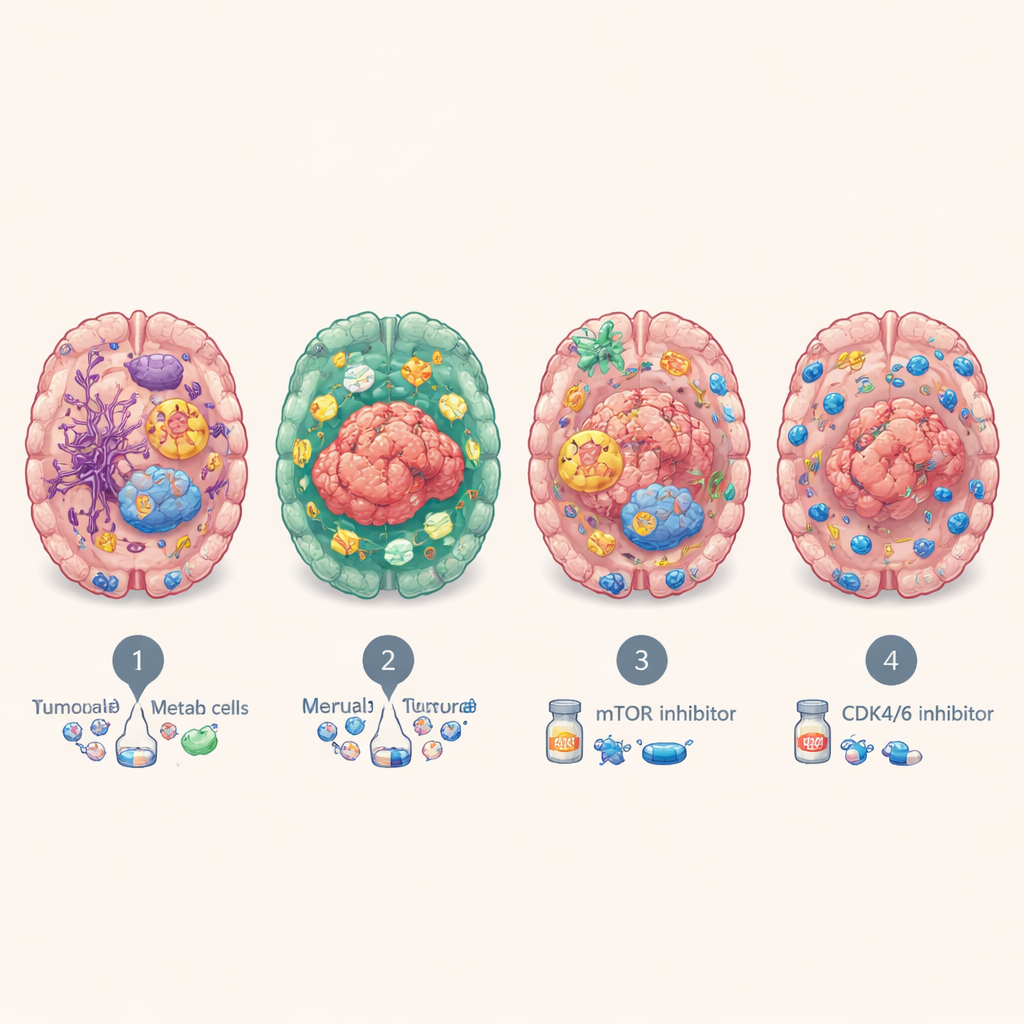
Despite coming from many organs, the brain metastases repeatedly fell into four major molecular subtypes. One, called BrMS1, looks “neural-like”: tumor cells and surrounding tissue express many brain and nerve cell programs, and show relatively active immune surveillance. BrMS2 is “immune-infiltrated,” packed with immune cells, especially T cells, and shows a program known as epithelial–mesenchymal transition, which loosens cell attachments and reshapes the local environment. BrMS3 is “metabolic,” dominated by highly active cancer cells that rewire their energy use—burning fuel through pathways like fatty acid metabolism and oxidative phosphorylation. BrMS4 is “proliferative,” marked by rapid cell division, stem-like features, and high genomic instability, but strikingly few immune cells.
What the tumor’s environment reveals
By zooming in at single-cell and spatial resolution, the team showed that each subtype lives in a distinct neighborhood within the brain. BrMS1 lesions are rich in brain support cells such as astrocytes and neurons, suggesting the tumor closely mimics and co-opts normal brain tissue. BrMS2 tumors, in contrast, are dotted with dense clusters of T cells, including many “exhausted” T cells expressing checkpoint molecules like PD-1, and have activated blood vessels and inflammatory signals. BrMS3 lesions sit in metabolically busy zones, while BrMS4 lesions form islands of rapidly dividing cells surrounded by relatively immune-poor territory. Across the board, the study found that, in brain metastases—but not in primary tumors—strong activation of epithelial–mesenchymal transition tends to go hand in hand with higher T-cell infiltration, hinting that this program may help open the door for immune cells to enter.
Clues to treatment and patient outcomes
These molecular personalities were not just academic. Patients with the immune-infiltrated BrMS2 subtype tended to live longer than those with the proliferative BrMS4 subtype, which consistently had the worst outcomes. By testing drugs on patient-derived organoids, the team found that the metabolic BrMS3 tumors were especially sensitive to blocking the mTOR signaling pathway, which helps control cell growth and metabolism, and that the fast-dividing BrMS4 tumors were more vulnerable to CDK4/6 inhibitors, drugs that slow the cell cycle. Separate analyses suggested that BrMS1 tumors may respond better to radiotherapy, while BrMS2’s inflamed, checkpoint-rich environment could make it more likely to benefit from immunotherapies that reinvigorate exhausted T cells—although this needs confirmation directly in patients with brain metastases.
What this means for people with brain metastases
This atlas shows that once cancer cells reach the brain, they tend to converge into a few shared biological states that cut across where they originally came from. Understanding whether a person’s brain metastasis is neural-like, immune-rich, metabolic, or highly proliferative could eventually guide more tailored treatments—such as choosing between radiotherapy, immunotherapy, or drugs that target metabolism or cell division. While the work does not yet change standard care, it provides a detailed roadmap for future research and clinical trials aimed at turning these molecular vulnerabilities into better, more precise therapies for patients facing brain metastases.
Citation: Yang, Z., Wei, S., Duan, H. et al. A proteogenomic atlas of 1032 brain metastases identifies molecular subtypes, immune landscapes, and therapeutic vulnerabilities. Nat Commun 17, 2038 (2026). https://doi.org/10.1038/s41467-026-68748-y
Keywords: brain metastasis, tumor subtypes, tumor microenvironment, cancer immunotherapy, cancer metabolism