Clear Sky Science · en
Ammonia oxidizers offset acidification stress via adaptive substrate affinity in aquatic ecosystems
Why tiny ocean workers matter
From mountain lakes to the open ocean, invisible microbes quietly manage much of the planet’s nitrogen, a key nutrient that feeds fisheries, influences water quality, and helps control greenhouse gases. As human-caused carbon dioxide makes waters more acidic, scientists have worried that this chemical shift could slow these microbial “workers,” disrupting food webs and boosting pollution. This study asks a deceptively simple question with big consequences: when waters become more acidic, do the microbes that burn ammonia—the first step in turning waste nitrogen into safer forms—shut down, or do they find ways to cope?
A global change problem in a drop of water
The researchers focused on ammonia oxidation, the process by which specialized microbes convert ammonia into nitrite, eventually leading to nitrate and nitrous oxide. This pathway helps strip excess nitrogen from water, but it also produces a powerful greenhouse gas. Earlier studies had painted a confusing picture: some experiments showed that acidification slows ammonia oxidation, while others showed little change—or even faster rates. To untangle this, the team sampled a wide sweep of aquatic environments, from a nutrient-rich freshwater reservoir and busy estuaries in southern China to the nutrient-poor Northwest Pacific. They also grew a representative ammonia-oxidizing archaeon, Nitrosopumilus maritimus, in the lab to watch its response under tightly controlled conditions.
Microbes under acid stress
As pH was experimentally lowered, the availability of ammonia in its usable form dropped, just as basic chemistry predicts. In many places dominated by ammonia-oxidizing bacteria, oxidation rates steadily declined with acidification, especially when substrate was scarce. But in other waters, particularly those where ammonia-oxidizing archaea were more abundant, the story differed. There, rates often stayed steady or even peaked under moderate acidification before falling off at stronger pH decreases. The same pattern appeared in the lab-grown archaeal strain. This suggested that some microbes were not simply being harmed by acidification—they were adjusting in a way that compensated for the loss of easily usable ammonia.
A hidden lever: grabbing scarce fuel more efficiently
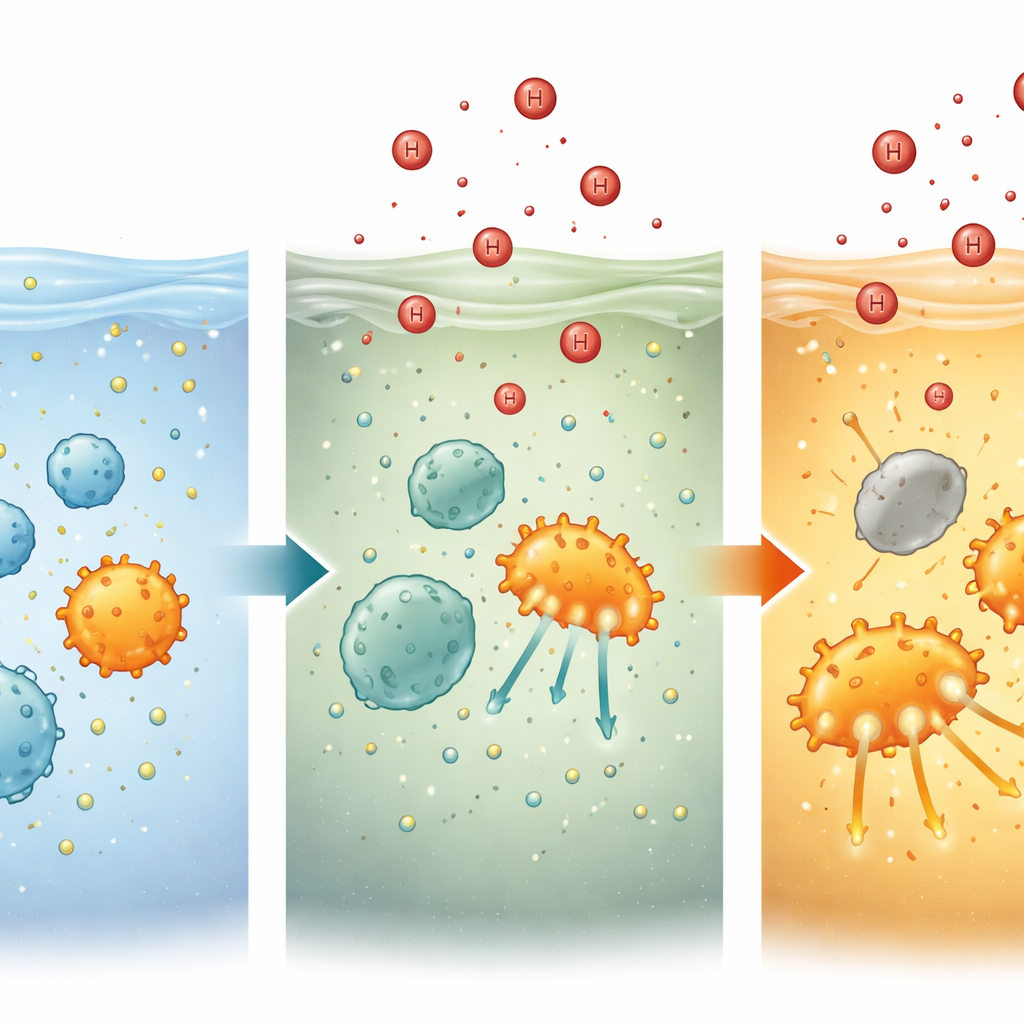
To understand how, the team turned to “substrate kinetics,” a quantitative way of describing how efficiently microbes take up and use their food. Instead of lumping all ammonium forms together, they tracked the uncharged ammonia molecules that the microbes actually consume. Across field sites and in the lab, they found that as pH dropped, the microbes needed less ammonia to maintain the same activity, meaning their effective “grip” on the substrate became stronger. This increase in substrate affinity was especially striking for archaea, which already excel at scavenging low levels of ammonium. In high-salinity estuaries and open-ocean waters where archaea dominate, the gain in affinity often more than offset the loss of available ammonia, allowing oxidation rates to hold steady under moderate acidification.
Different winners in different waters
By combining their measurements with ecological models, the researchers showed that two opposing forces act simultaneously under acidification: less usable ammonia pushes rates down, while higher affinity pulls them back up. In bacteria-dominated freshwater and inner estuary zones, the negative effect of reduced availability wins; even when extra substrate is added, acidification still tends to suppress activity. In archaeal-dominated outer estuaries and offshore waters, the situation can reverse. There, the affinity boost is so strong that it can balance, or even outweigh, the loss of substrate—at least until acidification becomes severe enough that overall metabolic capacity starts to decline. Molecular evidence from earlier work suggests that archaea achieve this resilience through high-affinity transport systems and active control of their internal pH, investments that bacteria often lack.
What this means for future oceans
Seen together, these results help reconcile years of conflicting observations and point to a simple organizing idea: under acidification, what matters is not just how much ammonia is present, but how well local microbes can grab it. In nutrient-rich, bacteria-heavy waters—such as many lakes and estuaries—acidification is likely to slow ammonia oxidation, allowing reactive nitrogen to build up and potentially worsening eutrophication problems. In contrast, in vast, nutrient-poor ocean regions dominated by archaea, rising acidity may not weaken this key nitrogen-processing step and could even speed it up under moderate pH declines. By highlighting substrate affinity as a key trait of microbial resilience, the study offers a new framework for forecasting how the marine nitrogen cycle—and related greenhouse gas emissions—will respond as the planet’s waters continue to acidify.
Citation: Tong, S., Shen, H., Han, LL. et al. Ammonia oxidizers offset acidification stress via adaptive substrate affinity in aquatic ecosystems. Nat Commun 17, 2083 (2026). https://doi.org/10.1038/s41467-026-68747-z
Keywords: ocean acidification, nitrogen cycle, ammonia oxidation, marine microbes, aquatic ecosystems