Clear Sky Science · en
Phospholipid composition strongly affects the assembly of β barrel proteins into purified bacterial outer membranes
Why Bacterial Coats Matter
Drug-resistant “superbugs” are a growing threat because many antibiotics simply cannot get past their tough outer defenses. This study looks closely at one key part of that defense in Gram‑negative bacteria like E. coli: a protective outer coat packed with barrel‑shaped proteins and specialized fats. By teasing apart how the mix of these fats controls the building of the outer coat, the authors reveal weak points that could be exploited when designing future antibiotics.
The Double-Walled Shield
Gram‑negative bacteria are unusual because they have two membranes. The inner membrane handles everyday cell functions, while the outer membrane acts like a rugged raincoat that keeps out many toxins and antibiotics. This outer layer is made of two types of lipids: common phospholipids on the inner side and a stiff sugar‑fat called lipopolysaccharide (LPS) on the outer side. Threaded through this shell are many “beta‑barrel” proteins—hollow cylinders that form pores and gates for nutrients. A molecular machine called BAM (short for barrel assembly machine) is responsible for folding these proteins into shape and inserting them into the outer membrane.
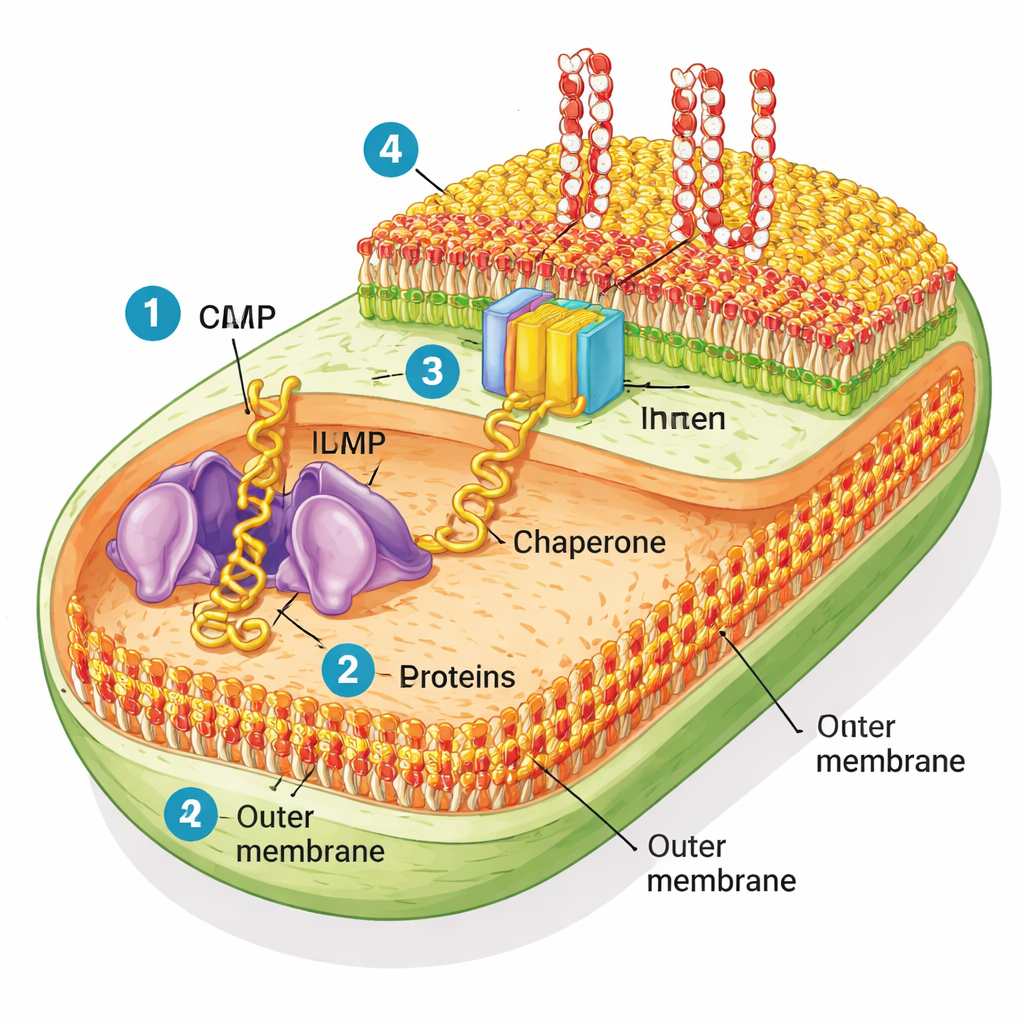
Recreating the Bacterial Coat in a Test Tube
To see how well BAM works in a realistic setting, the researchers purified pieces of E. coli outer membrane, creating tiny bubbles they call “native OMs.” Unlike simple artificial membranes used in many lab studies, these native membranes keep their full mix of proteins and lipids. The team then used a cell‑free protein factory to build new outer membrane proteins from scratch and watched whether BAM in these native bubbles could fold and insert them. They focused on a model beta‑barrel protein called EspP, along with a few others. When the helper protein SurA was present, BAM in the native membranes efficiently folded EspP, and this process was blocked by a known BAM‑targeting antibiotic called darobactin—clear evidence that the natural machine remained active.
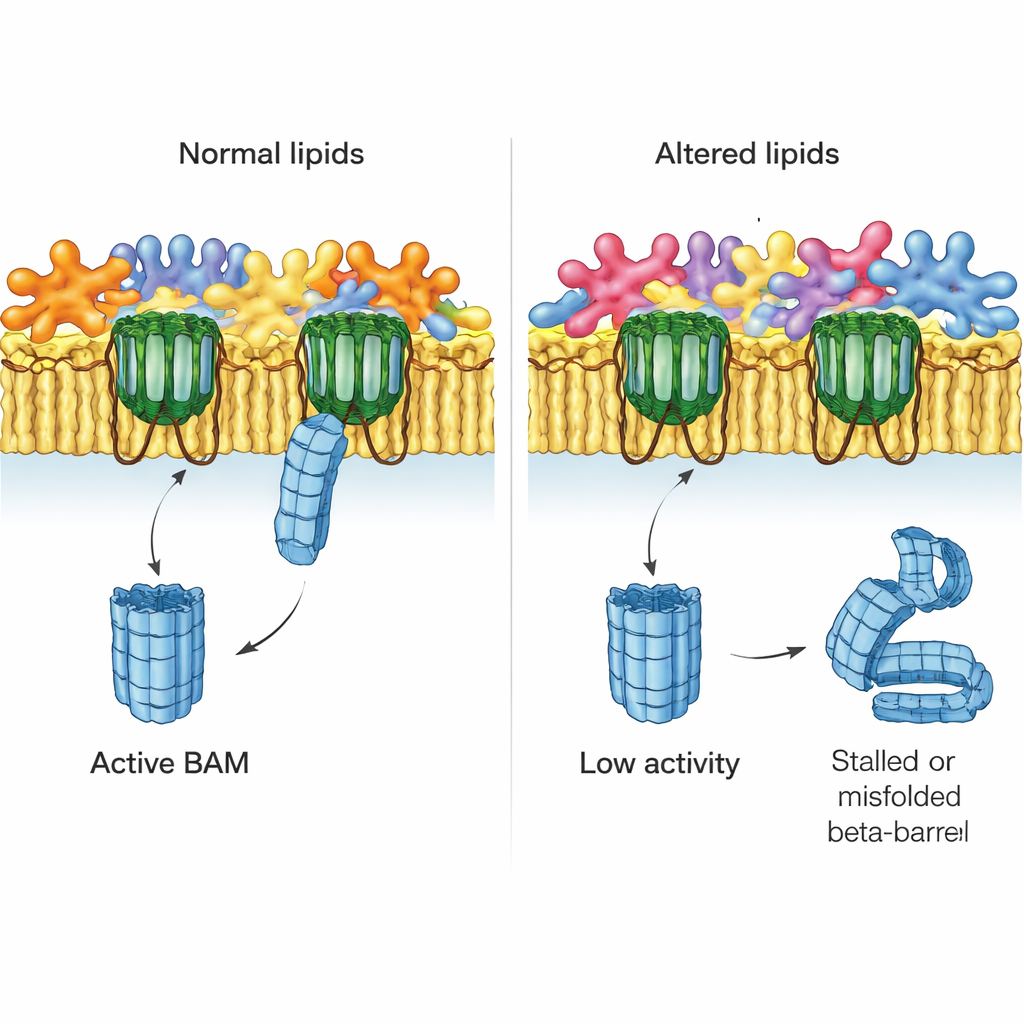
When the Fat Mix Goes Wrong
The authors next asked what happens when the lipid balance of the outer membrane is disturbed. They studied mutant strains of E. coli lacking key systems that normally correct misplaced phospholipids in the outer leaflet. One system, MlaA, hauls wayward phospholipids back to the inner membrane; another, the enzyme PldA, chops them up. Using native membranes from these mutants, the team found that BAM‑driven EspP assembly dropped noticeably in strains missing MlaA and was severely crippled when PldA was absent. These effects did not stem from losing BAM itself or other major proteins, which were still present at normal levels, pointing instead to the altered lipid environment as the culprit.
Mapping the Lipid Landscape in Detail
To link function to chemistry, the researchers developed a sensitive mass‑spectrometry method to catalog hundreds of individual phospholipids in whole cells and in purified outer membranes. In normal E. coli, they confirmed that one lipid type (phosphatidylethanolamine, or PE) is enriched in the outer membrane, while others (phosphatidylglycerol and cardiolipin) are relatively depleted there. They also found that “lyso‑lipids,” which have only one fatty tail and tend to bend membranes, are surprisingly concentrated in the outer membrane. In mutant strains, especially those lacking PldA, these patterns were scrambled: the usual differences between inner and outer membranes shrank or even reversed, and the lengths and types of fatty tails shifted. Strains with the most disturbed lipid profiles were the same ones where BAM worked poorest.
Implications for Antibiotic Strategies
Taken together, the work shows that the outer membrane is not just a passive scaffold for proteins. Its precise phospholipid makeup strongly influences how well BAM can build the beta‑barrel pores that keep the bacterium alive. When lipid balance is disrupted, fewer BAM complexes function correctly, and some work more slowly. Although live bacteria can compensate for such changes in ways that a test‑tube system cannot, this study highlights how tuning or disturbing specific lipids could weaken the bacterial coat. That insight opens new possibilities for drugs that either target BAM directly or subtly unbalance the outer membrane’s lipids, making even the toughest Gram‑negative bacteria more vulnerable.
Citation: Nilaweera, T.D., Brandes, N.T., LaCroix, I.S. et al. Phospholipid composition strongly affects the assembly of β barrel proteins into purified bacterial outer membranes. Nat Commun 17, 1915 (2026). https://doi.org/10.1038/s41467-026-68743-3
Keywords: Gram-negative bacteria, outer membrane proteins, beta-barrel assembly, bacterial lipids, antibiotic resistance