Clear Sky Science · en
Kynurenine mediates the chemotherapy-induced intestinal toxicity through modulation of gut microbiota
Why this matters for people on chemotherapy
Chemotherapy saves lives, but it often comes with grueling side effects in the gut—pain, diarrhea, weight loss—that can force doctors to cut doses or stop treatment early. This study uncovers a hidden chain reaction between a blood-borne chemical, immune cells, and gut microbes that helps explain why a common cancer drug, oxaliplatin, can be so harsh on the intestines, and points to new ways to protect patients without weakening their anticancer therapy.
A chemical link between treatment and gut damage
The researchers began with colorectal cancer patients receiving oxaliplatin-based chemotherapy. They compared blood samples from people who developed severe intestinal side effects with those who tolerated treatment better. Using targeted chemical profiling, they found that several breakdown products of the amino acid tryptophan were higher in the high-toxicity group, with one molecule—L‑kynurenine—standing out as the most strongly elevated. Patients with more L‑kynurenine in their blood also showed more signs of inflammation and lower white blood cell counts, hinting that this molecule might be tied directly to treatment-related harm.
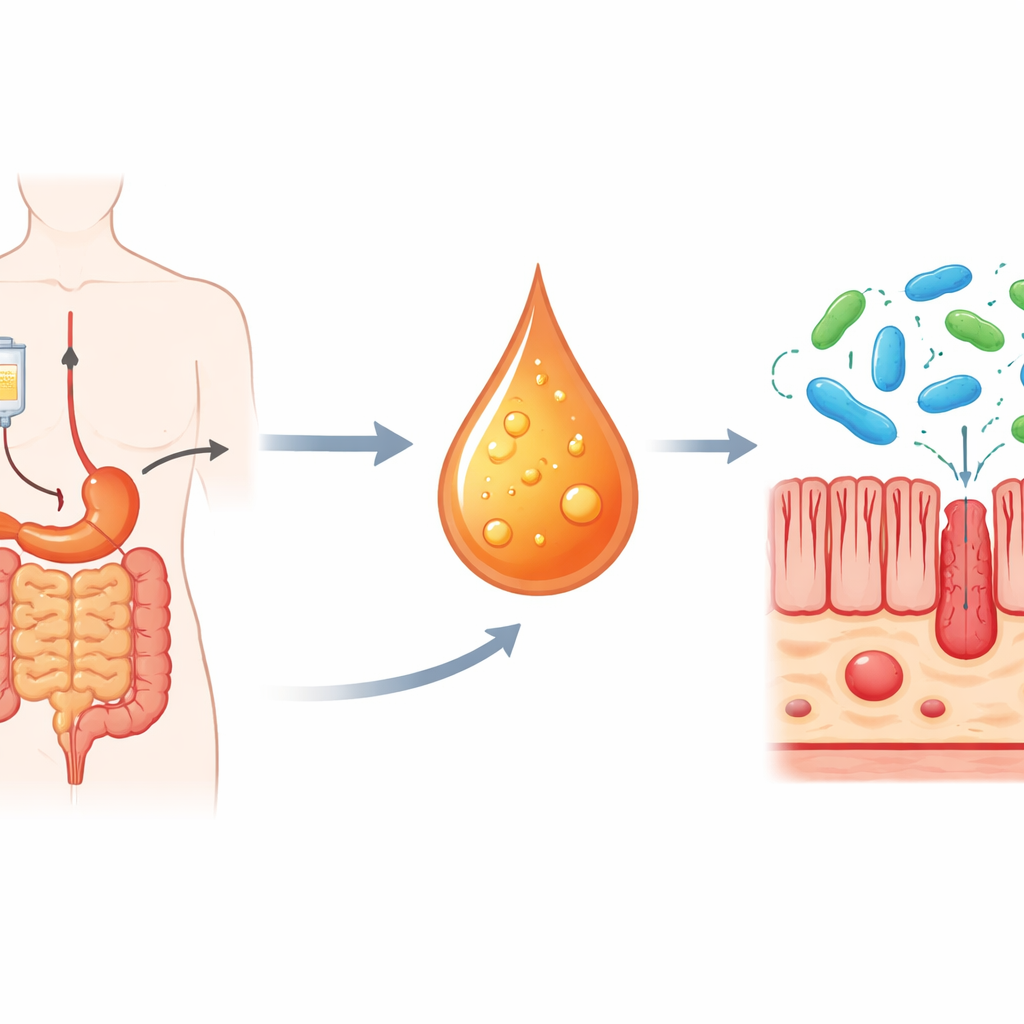
Proving cause and effect in mice
To move beyond correlation, the team turned to mouse models. When healthy mice were given high doses of oxaliplatin, they developed classic signs of intestinal injury: weight loss, shortened colons, swelling of the gut wall, fewer dividing cells, and more dying cells in the intestinal lining. These mice also showed a sharp rise in L‑kynurenine in their blood, mirroring the human data. When the researchers fed additional L‑kynurenine to oxaliplatin-treated mice, the intestinal damage became even worse—yet the cancer-killing power of the drug against tumors remained intact. Conversely, when they removed or blocked the enzyme IDO1, which the body uses to convert tryptophan into L‑kynurenine, the mice were far more resistant to gut toxicity while still benefiting from chemotherapy.
Immune cells and gut microbes in the crossfire
Diving deeper, the scientists asked which cells were responsible for making excess L‑kynurenine during treatment. They found that oxaliplatin spurs certain immune cells (CD8 T cells) to release the signaling protein interferon‑gamma, which then switches on IDO1 in nearby myeloid cells—a class of white blood cells. Mice engineered so that only their myeloid cells lacked IDO1 produced much less L‑kynurenine and were protected from intestinal injury, whereas deleting IDO1 only in gut lining cells had little effect. At the same time, the composition of the gut microbiota shifted. In normal mice, oxaliplatin and high L‑kynurenine levels were linked to a loss of a helpful bacterium, Lactobacillus johnsonii. In contrast, mice with reduced L‑kynurenine maintained higher levels of L. johnsonii. When gut bacteria were wiped out with antibiotics, the protective effect of low L‑kynurenine vanished, and when bacteria from low‑kynurenine mice were transplanted into normal mice, those recipients became more resilient to oxaliplatin’s gut damage.
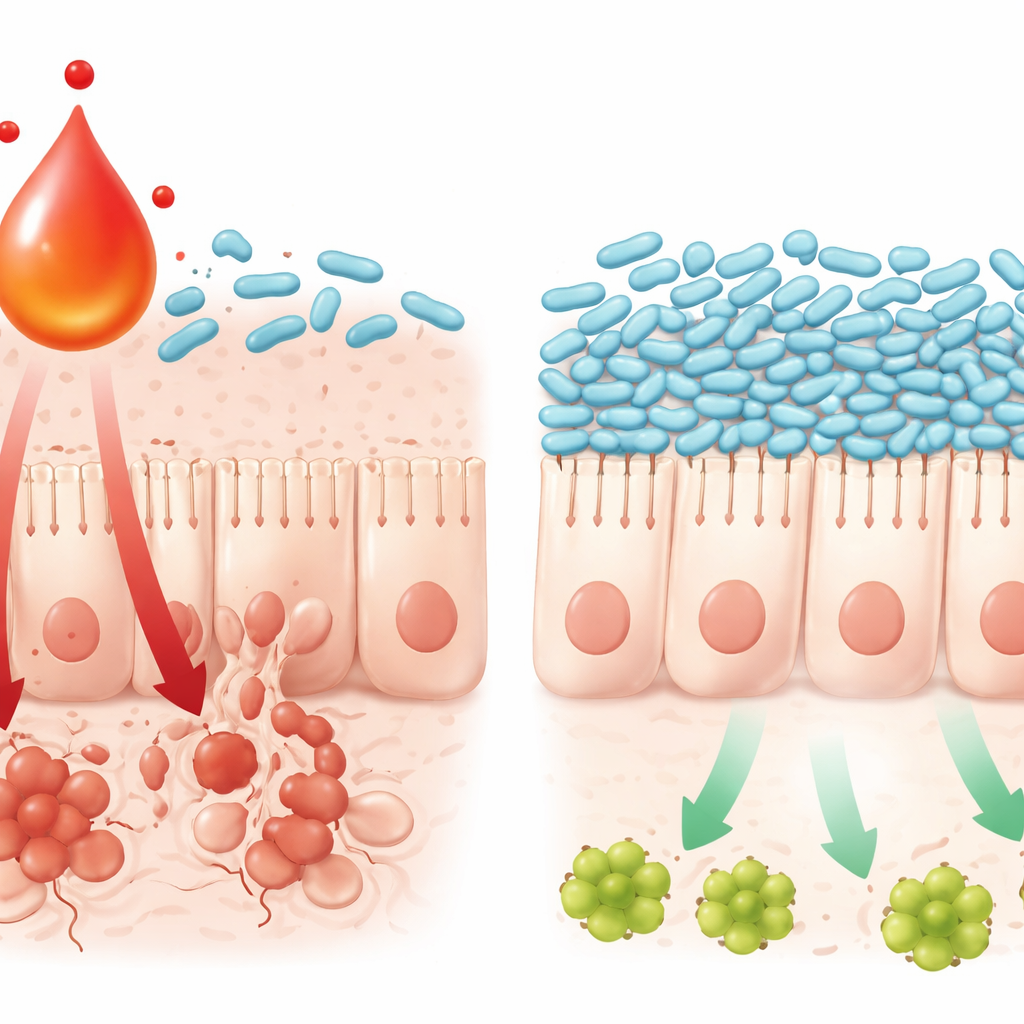
How one metabolite drives cell death in the intestine
The study also traced how this chemistry–microbe interaction injures the gut lining. High L‑kynurenine levels were associated with increased activity of a well-known inflammatory route in intestinal tissue, the TNFα/JNK pathway, which pushes cells toward programmed death. In cell culture, combining oxaliplatin with L‑kynurenine made gut cells more likely to activate this pathway and die, while adding L. johnsonii bacteria blunted the signal and preserved cell survival. L‑kynurenine itself could slow the growth and undermine the survival machinery of L. johnsonii in lab dishes, helping explain why the bacterium disappears when the metabolite builds up. Together, these findings outline a self-reinforcing loop: chemotherapy triggers immune signaling, immune cells ramp up L‑kynurenine, this disrupts friendly bacteria like L. johnsonii, and the altered microbial community in turn amplifies inflammatory signals that kill off gut cells.
New ways to protect patients during treatment
Encouragingly, the work also points to practical strategies. Treating mice with a drug called Epacadostat, which inhibits IDO1, lowered L‑kynurenine levels and markedly reduced oxaliplatin-induced intestinal injury without diminishing tumor control. In a complementary approach, the team engineered a harmless strain of E. coli to overproduce an enzyme that breaks down L‑kynurenine in the gut. Mice given these engineered bacteria were similarly protected from gut toxicity, again without compromising chemotherapy’s anticancer effects. Supplementing L. johnsonii itself also eased symptoms and preserved intestinal structure.
What this means for people with cancer
Taken together, this study reveals that a single treatment-driven metabolite—L‑kynurenine—acts as a key middleman between chemotherapy, the immune system, gut microbes, and intestinal injury. By showing that blocking its production, enhancing its breakdown, or restoring protective bacteria can all soften the blow of chemotherapy on the gut, the research opens a path toward add-on therapies that keep patients stronger and more comfortable while they fight cancer.
Citation: Xie, H., Yang, J., Wu, J. et al. Kynurenine mediates the chemotherapy-induced intestinal toxicity through modulation of gut microbiota. Nat Commun 17, 2087 (2026). https://doi.org/10.1038/s41467-026-68741-5
Keywords: chemotherapy side effects, gut microbiome, tryptophan metabolism, L-kynurenine, colorectal cancer