Clear Sky Science · en
Ampere-level CO2 electroreduction to multi-carbon oxygenates in acidic electrolyte through surface microenvironment reconstruction
Turning a Climate Problem into Useful Liquids
Carbon dioxide from burning fossil fuels is warming our planet, but it is also a potential raw material. This study explores how to turn CO2 into valuable liquid chemicals, such as ethanol and acetic acid, using electricity. The researchers show that by carefully redesigning the immediate surroundings of a copper electrode, they can make this transformation faster, more efficient, and compatible with harsh acidic conditions that usually sabotage such reactions.
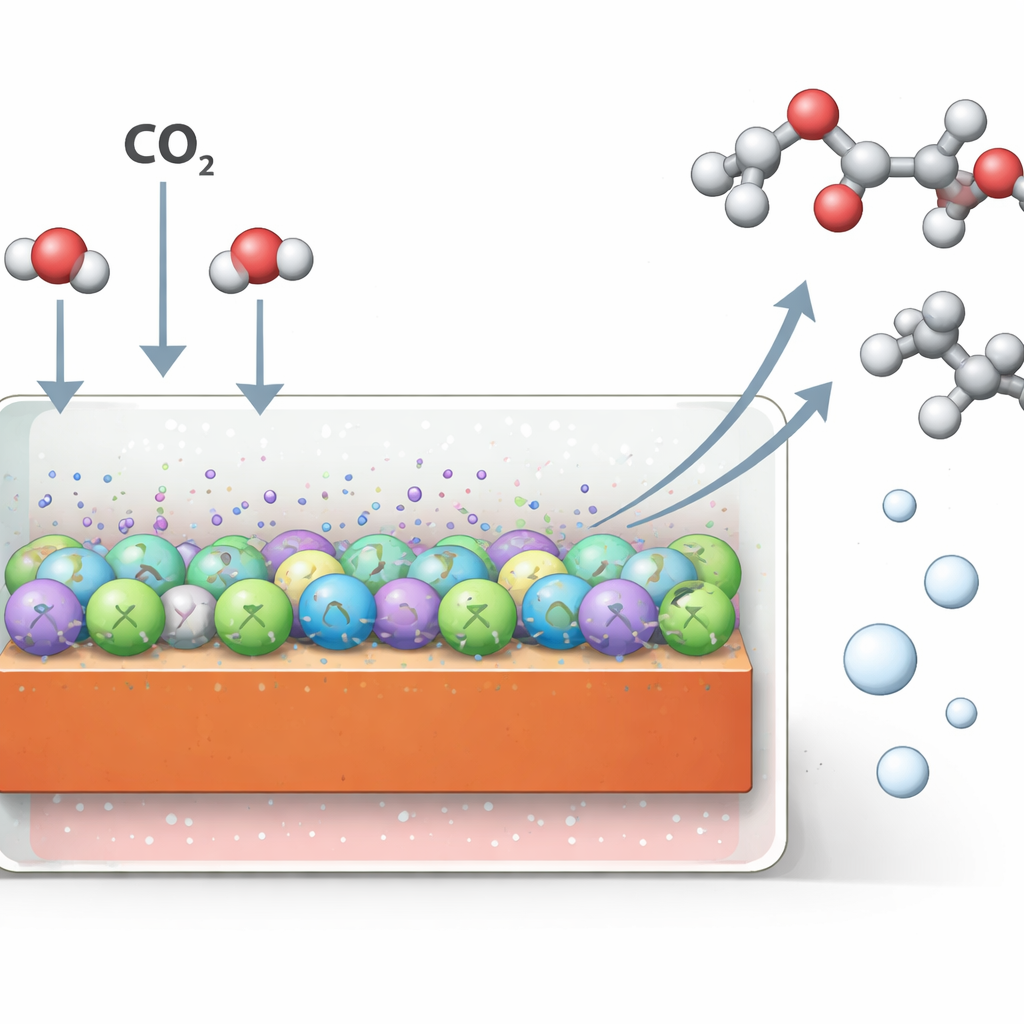
Why Making Liquid Fuels from CO2 Is Hard
Scientists have long dreamed of using surplus renewable electricity to convert CO2 into energy-rich products, storing sunlight and wind in chemical form. Copper is one of the few materials that can stitch CO2 together into multi‑carbon molecules, including alcohols and acids that industry already uses. Most progress so far, however, has relied on alkaline (basic) liquids, which cause CO2 to be wasted as invisible carbonates and clog devices with salt. Acidic liquids avoid these issues, but under such conditions copper tends to strip oxygen away from promising intermediates, favoring simple gases like ethylene and hydrogen instead of oxygen‑containing liquids.
Building a Smarter Copper Surface
To overcome this trade‑off, the team created a modified copper electrode they call IL@Cu. They formed it by reducing copper oxide in a water‑based solution containing a specially chosen ionic liquid, a salt that is liquid near room temperature. Positively charged components of this ionic liquid, based on a molecule called Bmim, attach themselves uniformly to tiny copper nanoparticles, giving the surface a gentle positive charge. Advanced microscopy and X‑ray techniques confirmed that the underlying copper remains metallic while the ionic liquid forms a thin, well‑anchored layer that alters how other ions and water arrange themselves at the interface where CO2 is converted.
Pushing Performance to Industrial Levels
When the researchers tested IL@Cu in a flowing acidic solution of potassium sulfate, they drove very large electric currents—up to two amperes per square centimeter, comparable to industrial electrolysis. Under these demanding conditions, the modified copper produced multi‑carbon products with a Faradaic efficiency of about 83%, meaning most of the electrons were used to make the desired molecules rather than wasteful side reactions. Even more impressively, about 60% of the current went specifically to liquid oxygen‑containing products, and ethanol alone accounted for roughly half of that. The device also used incoming CO2 very effectively: nearly four‑fifths of the gas passing through was converted in a single pass, and the catalyst maintained its activity and structure over 100 hours of operation.
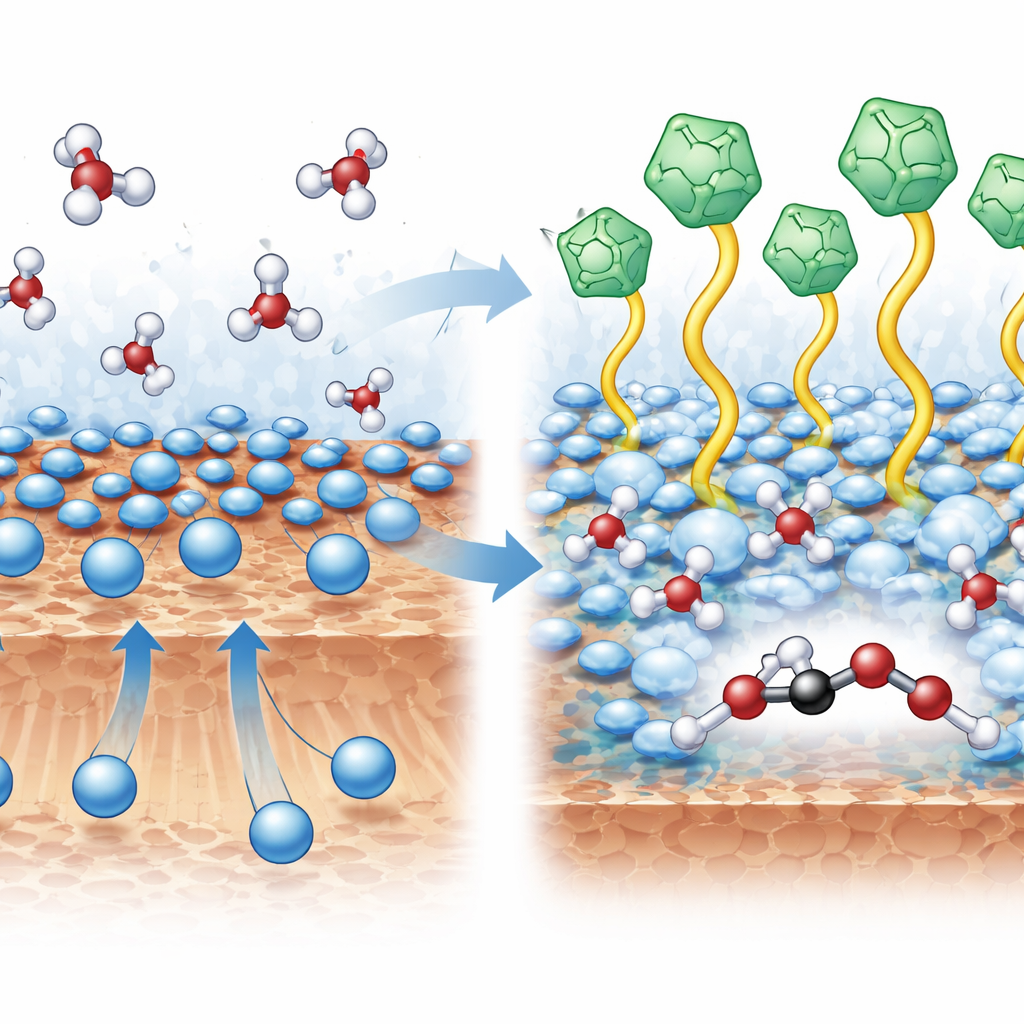
Reordering Water and Ions at the Surface
The heart of the advance lies in the microscopic layout of ions and water where the reaction happens. Spectroscopic measurements and computer simulations revealed that the ionic liquid cations repel nearby potassium ions away from the copper surface. This opens space for water molecules to move closer and form a more connected hydrogen‑bond network around key two‑carbon intermediates. With potassium held at an optimal distance, carbon‑containing fragments can more easily couple to one another rather than detach as carbon monoxide. At the same time, the surrounding water network helps preserve oxygen in the growing molecules instead of allowing bonds to snap and release ethylene gas. Quantum‑mechanical calculations showed that this reorganized environment lowers the energy barrier for carbon–carbon bond formation and tilts the reaction pathway toward oxygen‑rich liquids like ethanol.
Engineering the Invisible Layer That Matters
In essence, the study demonstrates that controlling the "microenvironment"—the nanometer‑scale arrangement of ions and water at an electrode—can be just as important as picking the right metal. By tethering ionic liquid molecules to copper, the authors simultaneously boost the rate of CO2 conversion, favor multi‑carbon products that are easier to store and transport, and keep the device stable in acidic solutions that are more practical for long‑term operation. This strategy of redesigning the thin, invisible layer where reactions occur may guide the development of next‑generation systems that turn waste CO2 into useful chemicals and fuels at scales relevant to mitigating climate change.
Citation: Yin, Y., Ling, Z., Liu, S. et al. Ampere-level CO2 electroreduction to multi-carbon oxygenates in acidic electrolyte through surface microenvironment reconstruction. Nat Commun 17, 2353 (2026). https://doi.org/10.1038/s41467-026-68739-z
Keywords: CO2 electroreduction, copper catalyst, ionic liquids, ethanol production, electrochemical CO2 recycling