Clear Sky Science · en
Managing electrolyte flow boosts the efficiency of continuous oxime electrosynthesis to over 95%
Turning Plastic Precursors Green
Nylon-6 is woven into our daily lives, from clothing and carpets to car parts. Yet one of its key ingredients, cyclohexanone oxime, is usually made by routes that burn fossil fuels and generate hazardous by-products. This study explores how to replace those routes with an electricity-driven process that can run continuously, waste less material, and reach very high efficiency, pointing toward cleaner production of everyday plastics.
Why Today’s Nylon Building Blocks Are a Problem
To make nylon-6, industry first produces cyclohexanone oxime, which is then transformed into caprolactam, the direct nylon precursor. Conventional factories obtain a crucial intermediate, hydroxylamine, by reducing nitrogen oxides with sulfur dioxide and hydrogen. This approach has a large carbon footprint, poor use of atoms, and serious safety and pollution concerns. Alternative chemistry based on hydrogen peroxide avoids some hazards but relies on a costly, unstable oxidant. With global nylon-6 capacity projected in the millions of tonnes per year, finding a safer, low-carbon path to cyclohexanone oxime is an urgent challenge.
Using Electricity to Drive Cleaner Chemistry
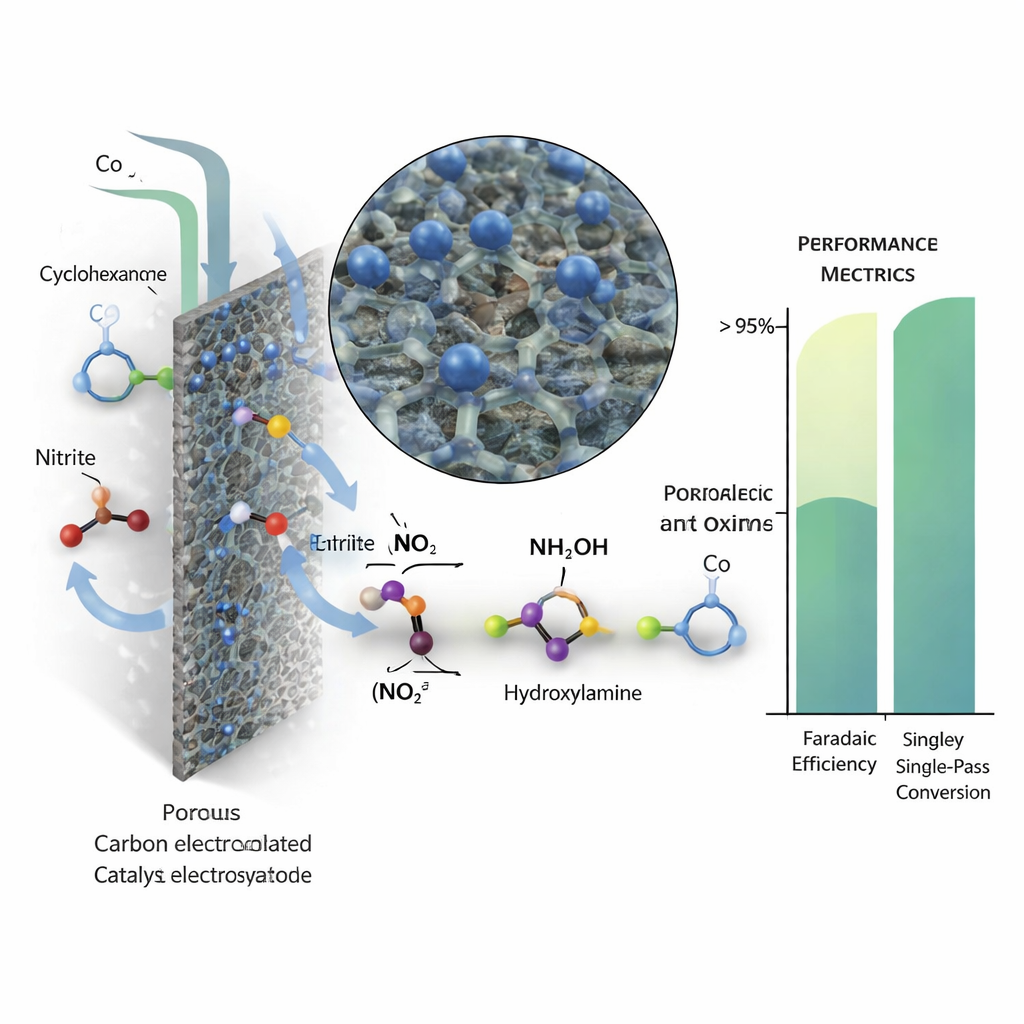
The authors tap into the growing availability of renewable electricity to redesign how cyclohexanone oxime is made. Instead of shipping in hydroxylamine, they generate it directly from nitrite ions in water at an electrode, where electrons supplied by a power source reduce nitrite stepwise to hydroxylamine. This freshly formed hydroxylamine then reacts on its own with cyclohexanone to give the desired oxime. Earlier lab demonstrations showed this route can work, but they relied on small batch cells with limited scalability and left a mismatch between how fast hydroxylamine is formed and how fast it reacts, leading to mediocre overall efficiency in continuous-flow devices.
Single-Atom Catalysts as Precision Tools
To boost performance, the team first searched for a highly selective catalyst. They created a family of “single-atom” materials, where isolated metal atoms (cobalt, iron, or manganese) are anchored on a nitrogen-doped carbon support. Detailed X-ray and electron microscopy studies confirmed that the metals are atomically dispersed, not clumped into particles. When tested in a standard electrochemical cell, the cobalt version stood out: it produced cyclohexanone oxime with Faradaic efficiency above 80 percent and almost perfect carbon selectivity, meaning nearly every carbon atom from cyclohexanone ended up in the target product. Advanced spectroscopy and computer simulations revealed why: cobalt binds key nitrogen-containing intermediates just strongly enough to steer the reaction toward hydroxylamine, but not so strongly that it over-reduces them to ammonia or wastes electrons on hydrogen gas.
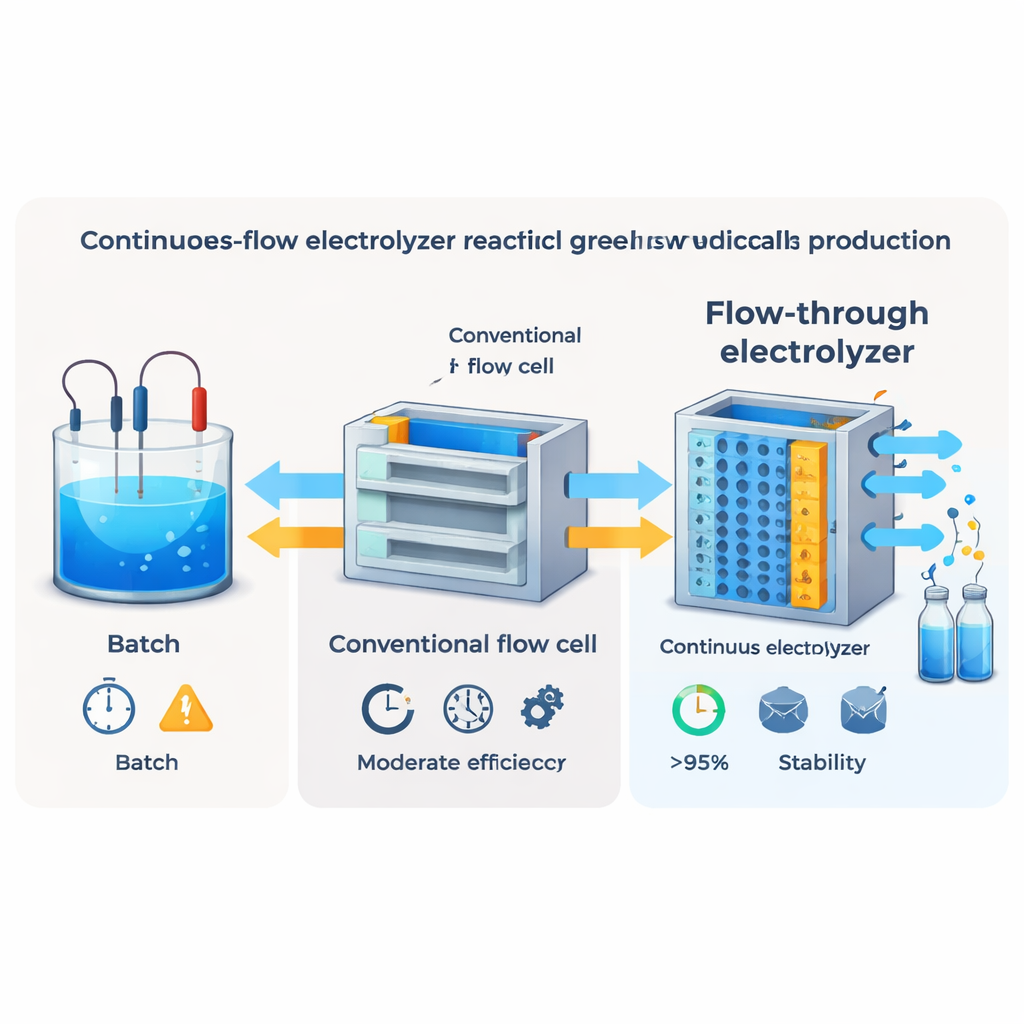
Rethinking How the Liquid Flows
Even an excellent catalyst cannot compensate for poor delivery of reactants. In conventional membrane-based flow cells, the liquid sweeps past rather than through the porous electrode, so molecules must slowly diffuse to the active sites. The researchers used fluid-dynamics simulations and experiments to redesign the cell so that the electrolyte is forced through the electrode itself. This “flow-through” architecture greatly shortens diffusion distances and creates strong convective flow across the catalyst. Compared with standard “flow-by” layouts, the new design increased the velocity of liquid inside the electrode by orders of magnitude and pushed the Faradaic efficiency for cyclohexanone oxime above 95 percent, even at industrially relevant currents.
Making Every Pass Count
For a continuous process to be practical, a single pass of liquid through the cell must convert most of the cyclohexanone, avoiding complex recycling loops. The team showed that by carefully tuning the flow rate and nitrite concentration, they could balance the rate of hydroxylamine formation with the supply of cyclohexanone. Under optimized conditions, single-pass conversion exceeded 95 percent while Faradaic efficiency remained high. The system ran stably for 110 hours, producing over 16 grams of crude oxime with high purity, and the cobalt catalyst retained its atomic structure. A techno-economic analysis suggests that, with further gains in efficiency and scale plus access to low-cost renewable electricity, the process could make cyclohexanone oxime at a cost compatible with large-volume nylon production.
What This Means for Everyday Materials
For non-specialists, the core message is straightforward: by carefully managing how liquid moves through an electrochemical reactor and by using a finely tuned single-atom catalyst, the authors turn a polluting, fossil-based step in nylon production into an efficient, electricity-driven one. Their flow-through design squeezes more product out of every electron and every drop of liquid, while their analysis shows a realistic path toward economic viability. Beyond this specific molecule, the same strategy—combining smart catalysts with smart flow architectures—could help electrify and clean up many other large-scale chemical processes that underpin modern life.
Citation: Li, J., Wang, X., Yang, X. et al. Managing electrolyte flow boosts the efficiency of continuous oxime electrosynthesis to over 95%. Nat Commun 17, 1970 (2026). https://doi.org/10.1038/s41467-026-68738-0
Keywords: electrosynthesis, flow-through electrolyzer, single-atom catalyst, nylon precursor, green chemistry