Clear Sky Science · en
Machine-learning-guided tungsten single atoms promote oxyhydroxides for noble-metal-free water electrolysis
Turning Water into Fuel More Efficiently
Splitting water into hydrogen and oxygen using electricity is one of the most promising ways to store clean energy from the sun and wind. But today’s best devices still waste a lot of power and often rely on rare, expensive metals like iridium. This study shows how combining artificial intelligence with clever chemistry can uncover a cheaper, more efficient material for making oxygen—the hardest half of the water-splitting reaction—bringing large-scale green hydrogen a step closer.
Why Oxygen Is the Hard Part
In water-splitting devices, the oxygen-forming reaction demands extra voltage beyond what simple chemistry would predict, acting like a stubborn toll gate that drains efficiency. Iridium-based materials are excellent at pushing this reaction forward, but they are scarce and costly, and their mining raises environmental concerns. More common metal compounds made from nickel, iron, and cobalt are strong candidates, yet their natural activity is limited. Scientists have learned that adding tiny amounts of other elements, or even isolating single atoms, can dramatically boost performance—but the possible combinations are almost endless, making trial-and-error experiments too slow.
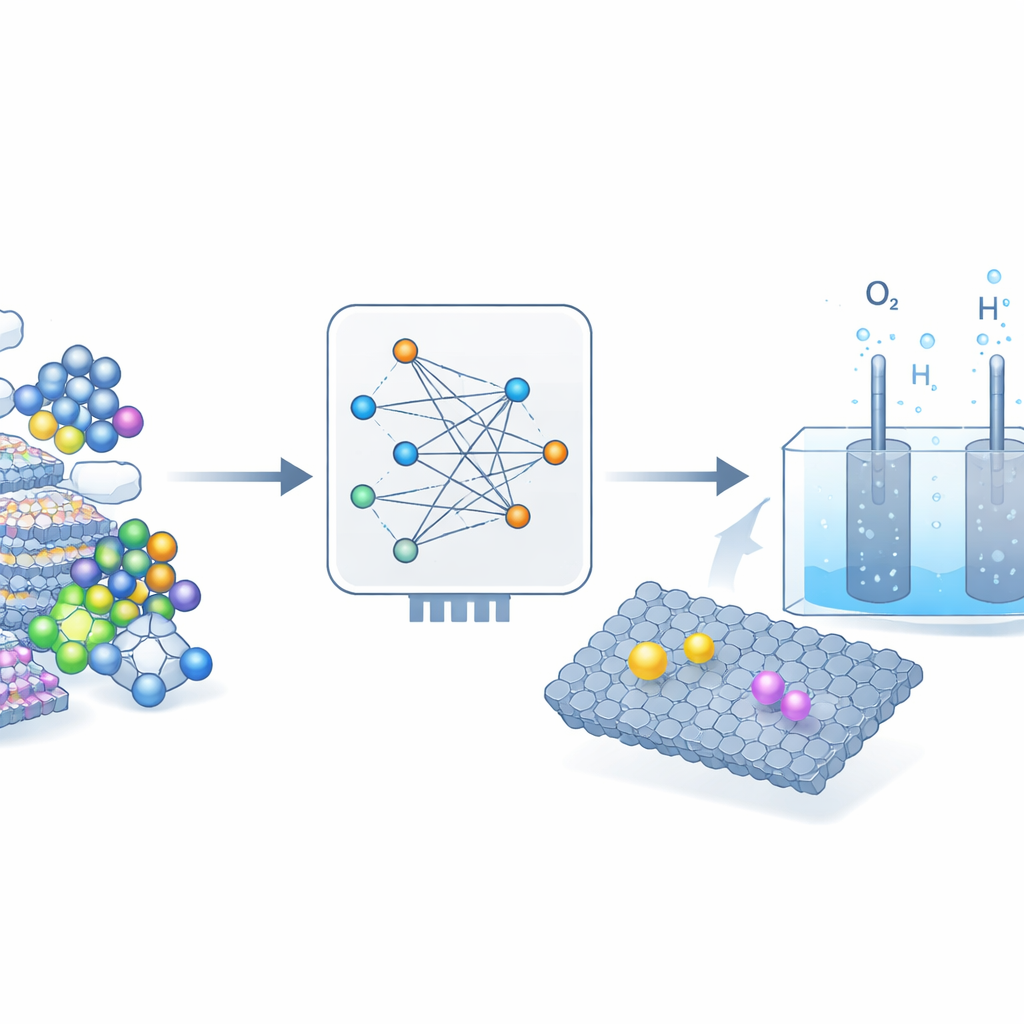
Letting Machine Learning Search the Lab
The researchers tackled this challenge by turning to a powerful machine-learning model, EquiformerV2, trained to predict how atoms interact on catalyst surfaces. They fed the model nearly 4,000 different designs where single metal atoms were introduced into layered metal oxyhydroxides—materials already known to work in alkaline water-splitting. For each design, the model rapidly estimated how strongly key reaction fragments would bind, something that would normally require heavy quantum-mechanical calculations. From this virtual screening, one standout emerged: a nickel–iron oxyhydroxide with isolated tungsten atoms hidden just below the surface, dubbed W1–NiFeOOH.
Building and Probing the New Catalyst
Guided by the computer results, the team developed a rapid electrodeposition method to grow ultrathin W1–NiFeOOH sheets directly on electrode supports in just minutes at room temperature. Advanced microscopy showed bright individual tungsten atoms dispersed within the nickel–iron lattice, without forming larger particles, and X-ray techniques confirmed that the original crystal framework remained intact. When tested in alkaline solution, this material needed substantially less extra voltage to drive the oxygen-forming reaction than standard nickel–iron oxyhydroxide and even a commercial iridium catalyst. In a full device using a commercial membrane, the tungsten-enhanced anode reached industrially relevant current densities—over 13 amps per square centimeter at 2.0 volts—while remaining stable for more than 500 hours.
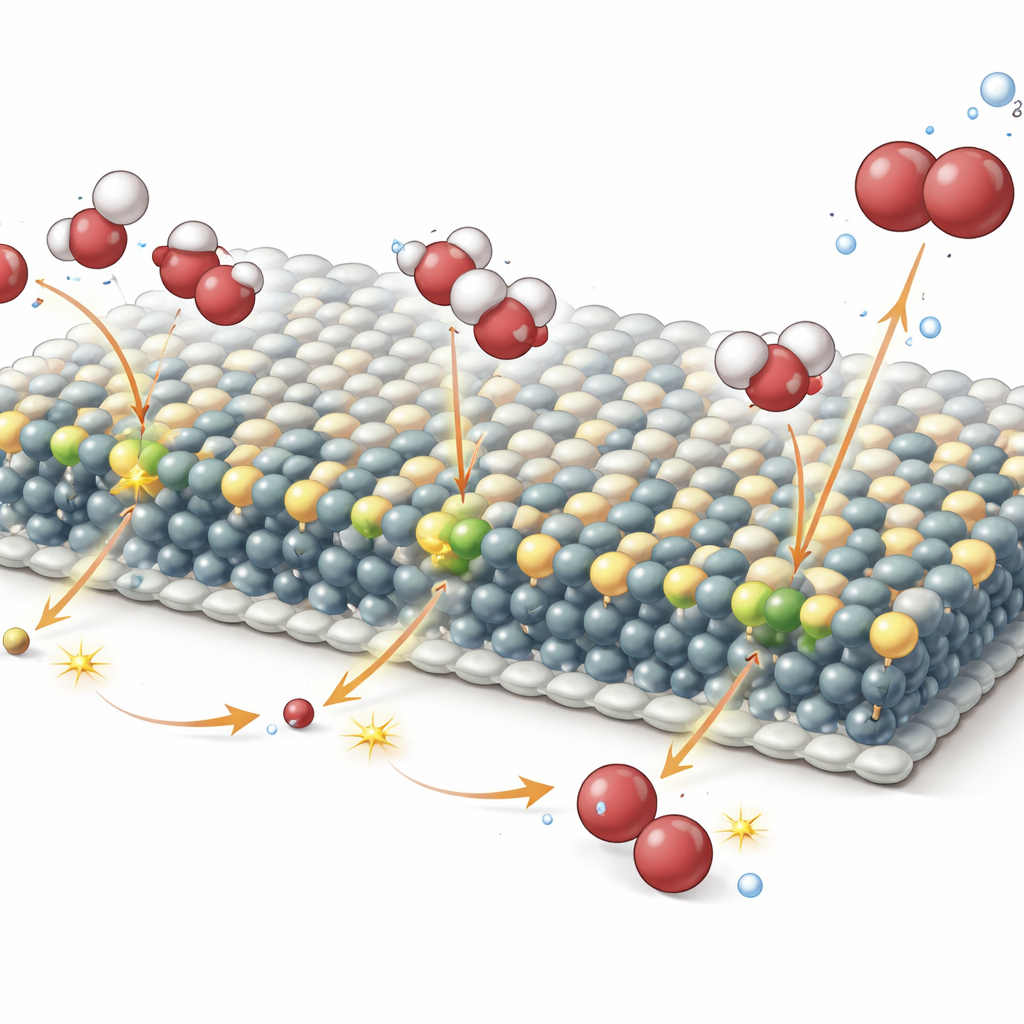
How Hidden Tungsten Atoms Do the Heavy Lifting
To understand why tungsten helps so much, the team watched the catalyst operate in real time using X-ray absorption and Raman spectroscopy. These measurements revealed that the tungsten atoms themselves barely change their chemical state during operation, meaning they are not the direct sites where oxygen forms. Instead, they subtly reshape the electronic environment of nearby nickel and iron atoms at the sheet edges, where the reaction actually takes place. This tuning makes it easier for the surface to give up protons and rearrange oxygen-containing groups, pushing the material into a more active “gamma” phase at lower applied voltages. Computer simulations backed this picture, showing that tungsten lowers the energy barrier for a critical reaction step by adjusting how electrons are shared between metal and oxygen atoms.
What This Means for Clean Hydrogen
By fusing fast machine-learning searches with careful experiments, the study delivers both a practical advance—a robust, iridium-free catalyst for oxygen generation—and a clear picture of how it works. Instead of acting as a star performer, each tungsten atom plays the role of a skilled coach, quietly boosting the abilities of the existing nickel–iron sites. This strategy of using single “promoter” atoms to fine-tune common materials could guide the design of many future catalysts, helping to cut costs and improve efficiency in devices that turn water and renewable electricity into clean hydrogen fuel.
Citation: Kim, J., Kwon, I.S., Lim, J. et al. Machine-learning-guided tungsten single atoms promote oxyhydroxides for noble-metal-free water electrolysis. Nat Commun 17, 2344 (2026). https://doi.org/10.1038/s41467-026-68735-3
Keywords: water electrolysis, oxygen evolution reaction, single-atom catalysts, machine learning materials discovery, green hydrogen