Clear Sky Science · en
Electrochemical C–N coupling via adsorption modulation: selective synthesis of amines from biomass-derived 5-hydroxymethylfurfural
Turning Plant Sugars into Useful Medicines
Modern medicines, agrochemicals, and advanced materials rely heavily on amines—nitrogen‑containing molecules that are everywhere from heartburn drugs to crop protectants. Today, many amines are made using high temperatures, high pressures, and hazardous reducing agents derived from fossil fuels. This study explores a cleaner route: using electricity and silver catalysts to turn a plant‑based sugar derivative called 5‑hydroxymethylfurfural (HMF) into a valuable amine used in pharmaceutical production, while carefully steering the reaction away from wasteful side products.
A Cleaner Path from Biomass to Amines
HMF can be obtained from carbohydrates in biomass, making it an attractive starting point for sustainable chemistry. Combining HMF with methylamine can yield a key intermediate known as MAMF, important in making the drug ranitidine and related compounds. Conventional “reductive amination” of HMF uses molecular hydrogen or other strong chemical reductants, which are energy‑intensive and generate unwanted by‑products. Here, the authors replace these reagents with electrons supplied by an external power source, performing the transformation electrochemically in water‑based solutions at near‑room temperature. Their central question is how to design the metal electrode so that the desired C–N bond forms efficiently while rival reactions—simple hydrogenation and carbon–carbon dimerization—are suppressed.
Why Surface Shape Matters
The team focuses on silver (Ag) as the electrode material because it binds HMF neither too weakly nor too strongly, an important balance for selectivity. But silver is not just one surface: at the atomic scale, it can expose different “facets” with distinct arrangements of atoms. The researchers synthesize two well‑defined silver catalysts: nearly spherical nanoparticles that mainly present the (111) facet, and nanocubes dominated by the (100) facet. Using electron microscopy and X‑ray techniques, they confirm these shapes and surfaces. When these catalysts are tested in an electrochemical cell containing HMF and methylamine, the difference is striking. The (111)‑rich nanoparticles achieve a Faradaic efficiency of about 89% and a selectivity around 90% for the target amine at modest voltages, clearly outperforming the (100)‑rich nanocubes, which tend to generate more hydrogenated and dimerized side products.
Watching Molecules Land and React
To understand why one shape works better, the authors watch how HMF and reaction intermediates bind to the silver surfaces in real time. They use in situ Raman spectroscopy, which tracks vibrational fingerprints of molecules at the electrode interface, and compare these experiments with detailed quantum‑chemical (DFT) calculations. On the (111)‑dominated nanoparticles, HMF adopts a configuration where its reactive carbonyl carbon sits close to the silver surface without being over‑stabilized. This arrangement makes the carbonyl group more positively polarized and easier for methylamine to attack, forming a short‑lived “imine” intermediate that is then rapidly reduced to the desired amine. On the (100)‑dominated nanocubes, by contrast, HMF binds through both carbon and oxygen of the carbonyl, locking the bond too tightly and channeling electrons into simple hydrogenation or dimer formation instead of C–N coupling.
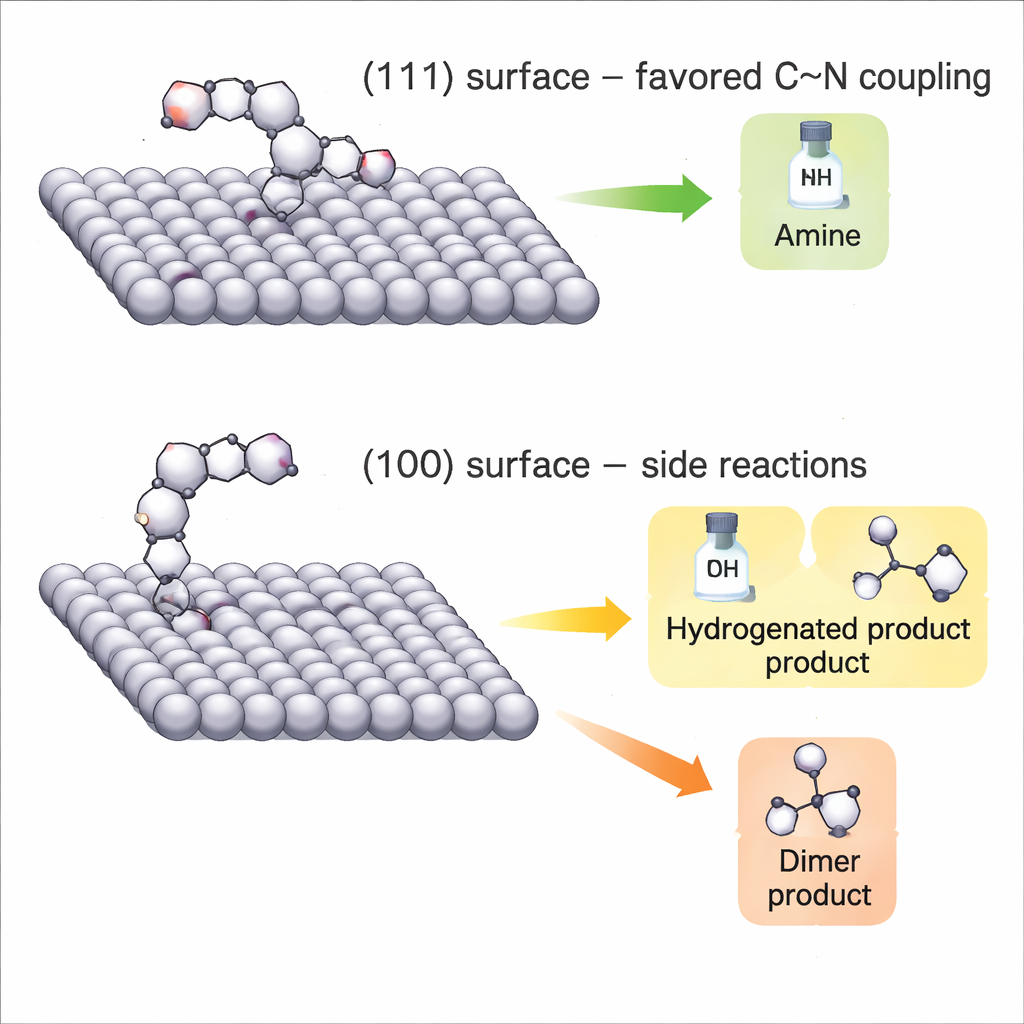
Balancing Hydrogen and Avoiding Detours
Electrochemical measurements shed further light on the reaction choreography. Kinetic analysis shows that C–N bond formation on the (111) nanoparticles proceeds more readily than competing hydrogen evolution from water. Isotope experiments using heavy water reveal that coupled proton‑and‑electron transfers are central to turning the imine into the final amine. Impedance tests indicate faster charge transfer on the (111) surfaces, and time‑resolved NMR confirms that the imine never accumulates in solution because it is either quickly reduced at the surface or hydrolyzed under the locally alkaline conditions near the cathode. By tuning the surface so that imine hydrogenation is fast—but not so fast that other bonds are reduced first—the authors keep the reaction on the desired path. They also show that the same facet preference improves amine formation from other furfural‑based molecules and even when ammonia replaces methylamine, suggesting a broadly useful design rule.
Design Rules for Greener Electrochemistry
For non‑specialists, the core takeaway is that the microscopic “texture” of a metal surface can decisively steer which products form in electricity‑driven reactions. By crafting silver nanoparticles that expose mostly (111) facets, the researchers guide a plant‑derived building block toward a specific amine used in pharmaceuticals with high efficiency and minimal waste. This work demonstrates how controlling how molecules sit and move on a catalyst—rather than just which metal is used—can unlock greener routes to important chemicals and potentially help translate biomass and renewable electricity into everyday products more sustainably.
Citation: Lai, D., Yu, J., Ma, ZA. et al. Electrochemical C–N coupling via adsorption modulation: selective synthesis of amines from biomass-derived 5-hydroxymethylfurfural. Nat Commun 17, 1892 (2026). https://doi.org/10.1038/s41467-026-68734-4
Keywords: electrochemical amination, biomass valorization, silver nanoparticles, catalyst surface facets, green chemistry