Clear Sky Science · en
Gemcitabine plus selinexor in selective advanced sarcomas: a phase I of the Spanish group for research on sarcoma study
New Hope for a Rare and Tough Cancer
Sarcomas are rare cancers that often strike younger adults and can be very hard to treat once they spread. For many patients, current drugs only keep the disease under control for a short time. This study tests a new drug combination—an old chemotherapy medicine called gemcitabine plus a newer targeted pill, selinexor—to see whether working together they can better slow or shrink advanced sarcomas while keeping side effects manageable.
Why Combining Two Medicines Might Work Better
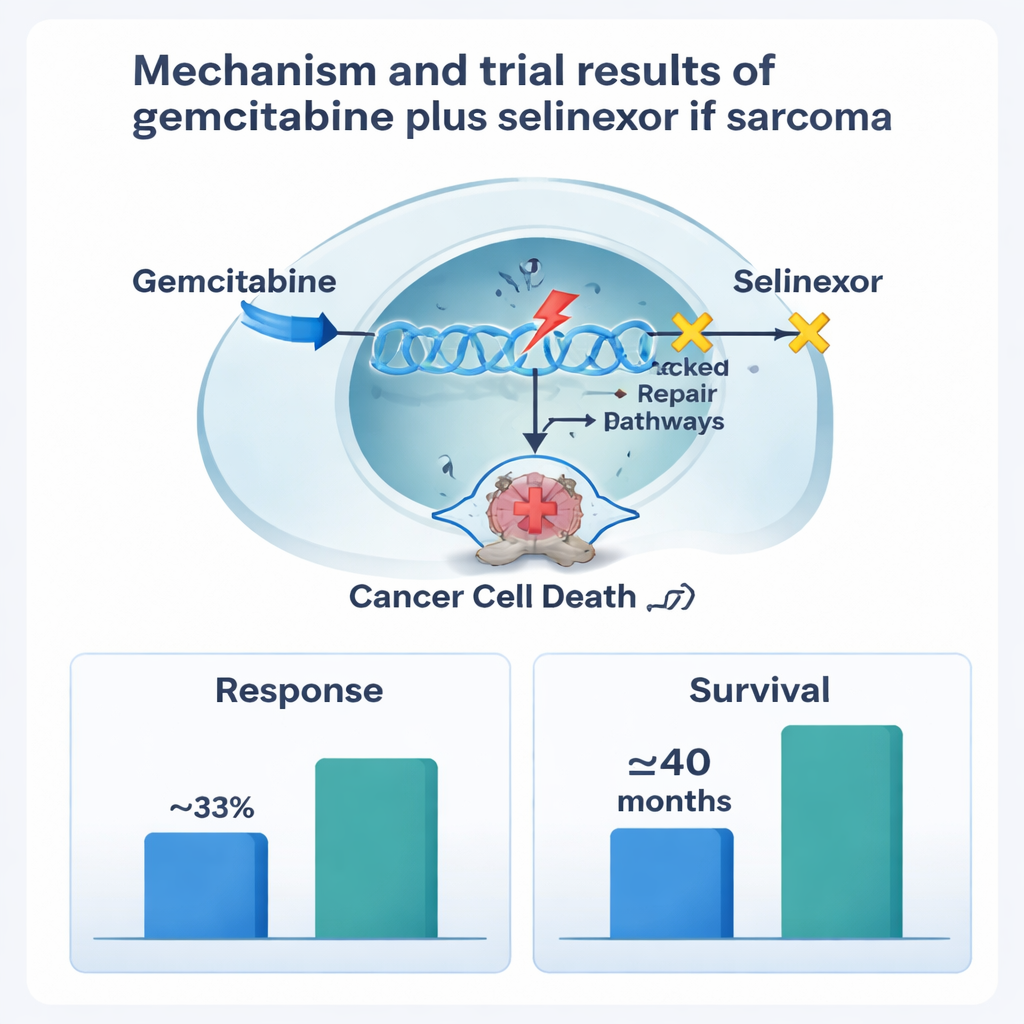
Gemcitabine is a standard chemotherapy that damages the DNA of rapidly dividing cells, including cancer cells. Selinexor works in a very different way: it blocks a protein called exportin-1, which normally helps move important control proteins out of the cell nucleus. When exportin-1 is overactive, as it is in many tumors, cancer cells can dodge the body’s internal “stop signals” and resist drugs. By trapping tumor-suppressing proteins inside the nucleus, selinexor can push cancer cells toward self-destruction. Laboratory work suggested that giving gemcitabine first to damage DNA, followed by selinexor to block repair and survival pathways, might be especially powerful in some sarcomas.
Promising Signals in Lab Experiments
Before treating patients, the researchers tested the two drugs together in cancer cells grown in the lab, focusing on three sarcoma types: leiomyosarcoma, osteosarcoma, and malignant peripheral nerve sheath tumors. In several leiomyosarcoma and nerve sheath tumor cell lines, the combination killed more cancer cells than either drug alone, showing true synergy. These cells showed more DNA damage and higher rates of programmed cell death when both drugs were used together. The team also studied proteins that help cells survive and divide, such as one called survivin. When selinexor drove survivin levels very low and altered another protein, IκBα, the combination worked particularly well. In contrast, osteosarcoma cells often showed the opposite effect: the two drugs interfered with each other, suggesting this approach might help only selected sarcoma types.
How the Study Was Run in Patients
The clinical trial enrolled 17 adults in Spain with advanced sarcomas that had already grown despite at least one round of standard treatment. Most had leiomyosarcoma, and a smaller number had osteosarcoma or two other rare sarcomas. Doctors used a careful stepwise design to find the highest dose that patients could take safely. All received gemcitabine by vein on days 1 and 8 of a three-week cycle, followed by weekly selinexor pills. The team closely watched for severe side effects in the first treatment cycles to decide the recommended dose for future studies.
Safety, Side Effects, and Early Benefits
The biggest side effects were drops in blood counts—especially white cells and platelets—which are common with chemotherapy. These were frequent but generally manageable using growth-factor injections, dose adjustments, and treatment delays. Nausea, vomiting, and fatigue were also common but mostly mild to moderate, and no patient had to quit the study because of toxicity. The final recommended dose kept gemcitabine at a relatively high level while using a slightly lower weekly selinexor dose to improve tolerability. Among 16 patients whose tumors could be measured, five had clear tumor shrinkage and another five achieved stable disease, leading to an overall response rate of about 31 percent. For the whole group, the cancer stayed under control for a median of about 5 to 6 months. Patients with leiomyosarcoma did particularly well, with disease control lasting close to 8 months on average and overall survival approaching three and a quarter years, although the small number of patients means these results must be interpreted cautiously.
Clues to Who Might Benefit Most
Because not all patients responded, the researchers searched for markers in tumor samples that might predict benefit. High levels of survivin and strong nuclear staining of IκBα were both linked to shorter periods before the cancer grew again, especially in leiomyosarcoma. This fits with the lab finding that complete loss of survivin is associated with stronger drug synergy. If confirmed in larger studies, these proteins could help doctors identify which patients are most likely to respond to the gemcitabine–selinexor combination and which might need different strategies.
What This Means for Patients With Sarcoma
For people facing advanced sarcomas, treatment options are limited and often only briefly effective. This early-phase study shows that pairing gemcitabine with selinexor is feasible, has side effects that can be handled with careful support, and offers encouraging signs of benefit, particularly for leiomyosarcoma. The work also begins to uncover biological markers that might guide more personalized care. Larger phase II and III trials are now under way to confirm whether this combination truly improves survival and quality of life, but for patients and clinicians, it represents a realistic new avenue in the search for better sarcoma treatments.
Citation: Martin-Broto, J., Casado, A., Marquina, G. et al. Gemcitabine plus selinexor in selective advanced sarcomas: a phase I of the Spanish group for research on sarcoma study. Nat Commun 17, 1873 (2026). https://doi.org/10.1038/s41467-026-68729-1
Keywords: sarcoma treatment, leiomyosarcoma, gemcitabine, selinexor, cancer clinical trial