Clear Sky Science · en
Modulating thermo-diffusion/galvanic coupling via ion speciation engineering enables high-performance ionic thermoelectric cells
Turning Gentle Warmth into Useful Power
Every day, huge amounts of gentle warmth—from warm pipes, electronics, factory equipment, and even our own skin—escape into the air as waste. This paper explores a new kind of soft, salt-filled gel that can turn these small temperature differences directly into electricity. By carefully arranging copper and chloride ions inside a flexible polymer, the researchers show how to get both high voltage and steady power from low-grade heat, opening doors for self-powered wearables and tiny sensors that run on nothing but warmth.
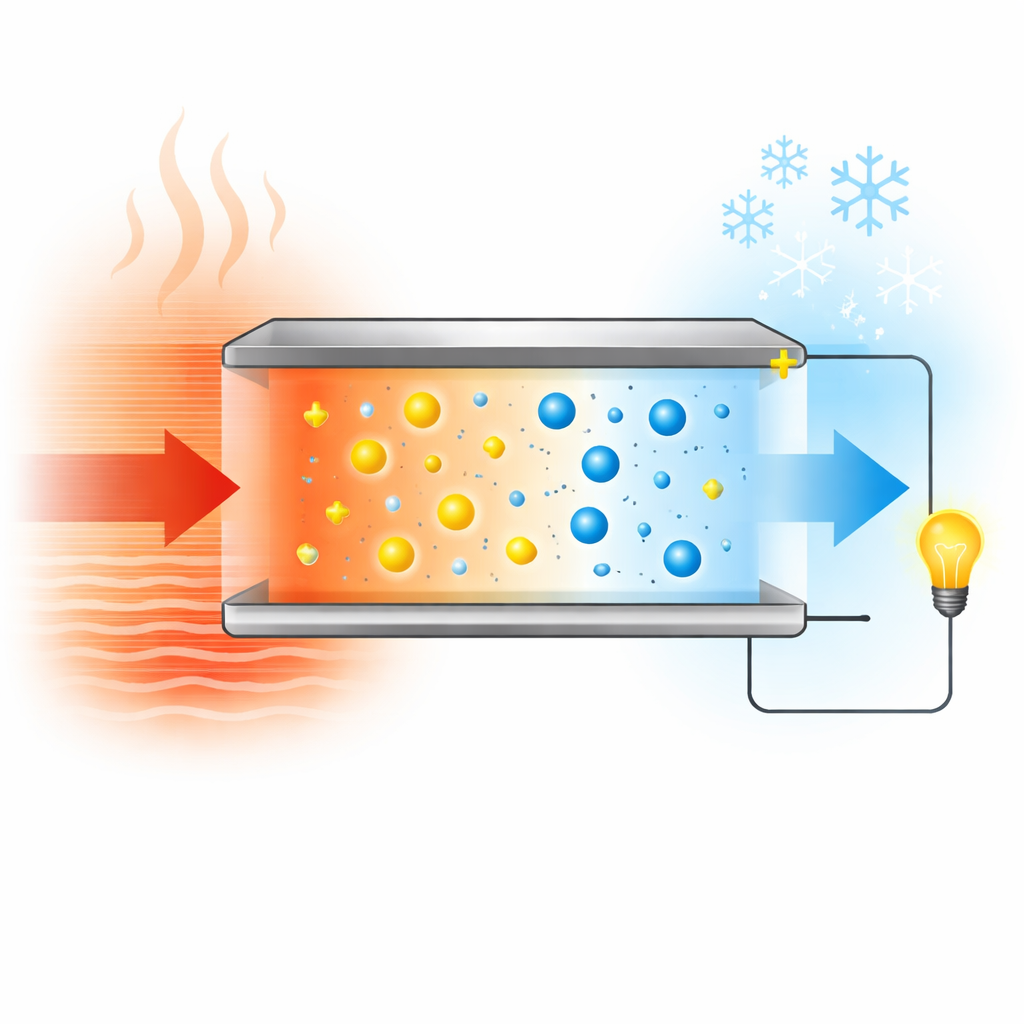
Why Waste Heat Is Hard to Use
Most technologies that convert heat to electricity rely on electrons flowing through rigid crystals made with rare or costly metals. These conventional devices work best at high temperatures and usually deliver only modest voltages per degree of temperature difference. That makes them poorly suited for harvesting mild warmth below about 100 °C, such as body heat or room-temperature equipment. In contrast, ionic thermoelectric gels use moving ions in a soft, water-rich network to create a voltage when one side is warmer than the other. Some of these gels generate huge bursts of voltage but fade quickly, while others give a steady output but at low voltage. The central challenge has been achieving both strong voltage and lasting power in the same simple material, especially for negatively charged (n-type) systems needed for practical devices.
A Simple Gel with a Hidden Advantage
The team focused on an uncomplicated recipe: a common water-soluble polymer (polyvinyl alcohol) soaked with copper chloride salt. At first glance, this kind of gel was known mainly for an effect where ions drift from hot to cold, building up charge temporarily. The authors discovered that copper ions in the presence of chloride can also quietly undergo a reversible chemical change between two charge states, Cu²⁺ and Cu⁺, whenever a temperature difference is applied. This “thermogalvanic” reaction lets electrons move through the external circuit again and again, sustaining current over long times. Using advanced Raman scattering, X-ray probes, and computer simulations, the researchers directly tracked how copper–chloride complexes form, move, and switch charge state inside the gel while it is working.
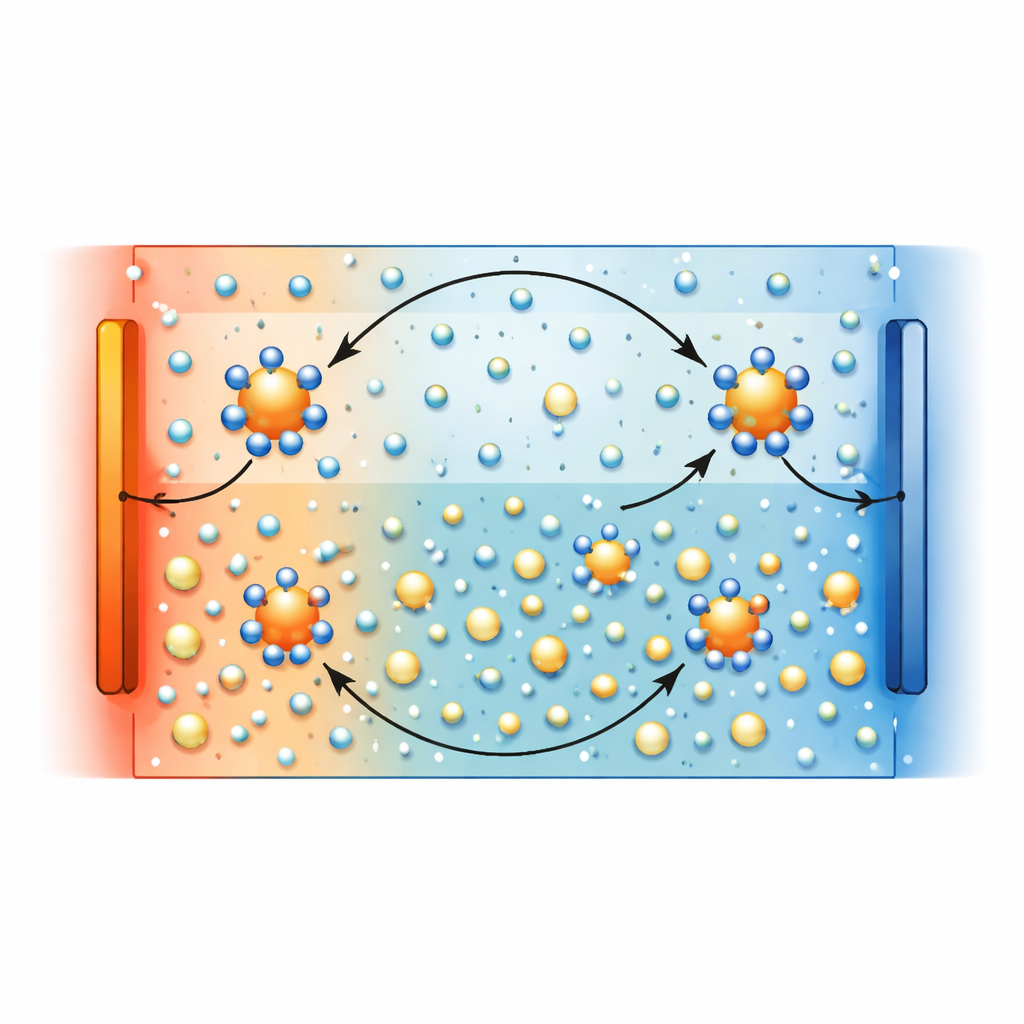
Balancing Two Paths to Electricity
In this gel, electricity comes from two intertwined processes. First, chloride ions and copper complexes drift under the temperature gradient, which can generate a large voltage but tends to be short-lived. Second, copper ions at the electrodes repeatedly gain and lose electrons in a redox cycle stabilized by surrounding chloride ions, which supports a continuous flow of current. Crucially, both processes rely on the same chloride ions, so they compete with each other. At low copper chloride levels, the gel favors ion drift, giving a very large voltage—over 30 millivolts per degree—but limited continuous current. As the copper chloride concentration increases, more copper–chloride complexes appear that boost the thermogalvanic reaction and power output, while the pure drift contribution to voltage is gradually suppressed.
Tuning the Inner Chemistry for Best Performance
By mapping exactly which copper–chloride species exist at different salt concentrations, the authors identified the combinations that give the best balance of high voltage and strong power. Moderate concentrations favor simple copper complexes that support both mechanisms, producing a record-high thermopower of about minus 30.6 millivolts per kelvin—far beyond typical electronic thermoelectrics. Higher chloride content, sometimes aided by adding extra salts like calcium chloride and by improving the electrodes with a thin gold layer, maximizes the number of active redox pairs. This pushes the power density up to 0.6 milliwatts per square meter per kelvin squared and allows continuous current for hours with excellent stability over many cycles. Linking 16 of these cells in series yields a module that can reach 3.5 volts from only a 15-degree temperature difference and power small devices without extra electronics.
From Warm Surfaces to Self-Powered Devices
For a non-specialist, the key message is that the researchers have learned how to “tune the recipe” of a soft copper–salt gel so that it converts gentle warmth into electricity both strongly and steadily. By controlling how copper and chloride ions pair up and move, they overcome a long-standing trade-off between high voltage and long-lasting output. The resulting flexible, low-cost ionic thermoelectric cells and modules can run on small temperature differences found in everyday settings, pointing toward future wearables and sensors that quietly power themselves from the warmth already around us.
Citation: Li, Y., Qiu, YR., Liao, J. et al. Modulating thermo-diffusion/galvanic coupling via ion speciation engineering enables high-performance ionic thermoelectric cells. Nat Commun 17, 2209 (2026). https://doi.org/10.1038/s41467-026-68721-9
Keywords: ionic thermoelectrics, waste heat harvesting, copper chloride gel, flexible energy devices, thermogalvanic cells