Clear Sky Science · en
Nickel-catalyzed divergent sulfonations of propargylic carbonate
Why this chemistry matters beyond the lab
Sulfur-containing molecules sit at the heart of many modern medicines, crop-protection agents, and advanced materials. Yet making these compounds in a precise, efficient, and flexible way is often difficult. This paper describes a nickel-based catalytic method that can turn simple starting materials into several different families of sulfur-rich molecules, all with tightly controlled 3D shapes. That kind of control can speed up drug discovery and help chemists build complex molecules more cleanly and sustainably.
Building useful molecules from simple pieces
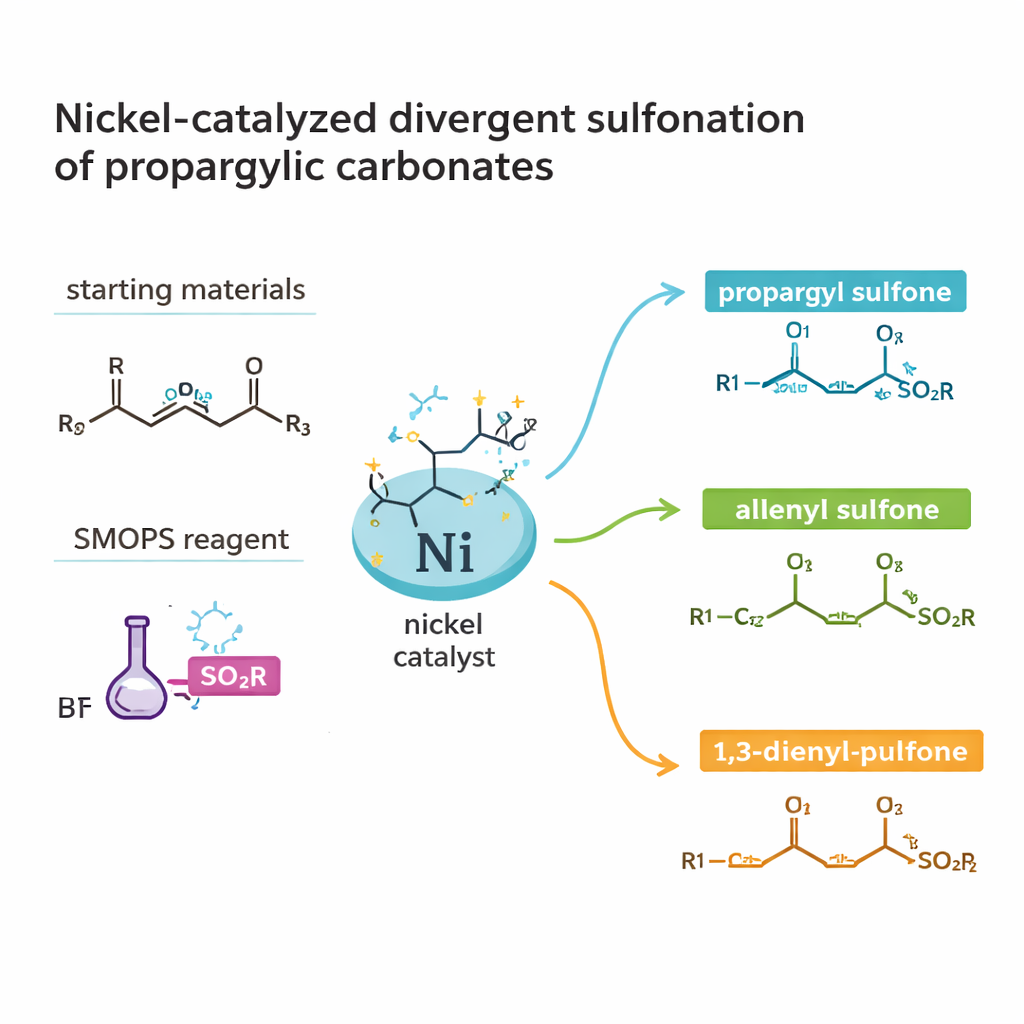
The work centers on a class of molecules called sulfones and sulfinates, which contain sulfur bonded to oxygen and carbon. These structures appear throughout bioactive natural products and pharmaceuticals, but traditional routes to them can be long and wasteful. The authors focus on a particularly valuable subtype: chiral sulfinates, whose mirror-image forms can behave very differently in the body. Instead of starting from already complex building blocks, they use two readily available partners: propargylic carbonates (a kind of small carbon backbone with a built‑in leaving group) and a commercial sulfur source known as SMOPS. By combining these under nickel catalysis, they aim to generate highly valuable sulfur-containing products in just one or two steps.
One catalytic system, three product families
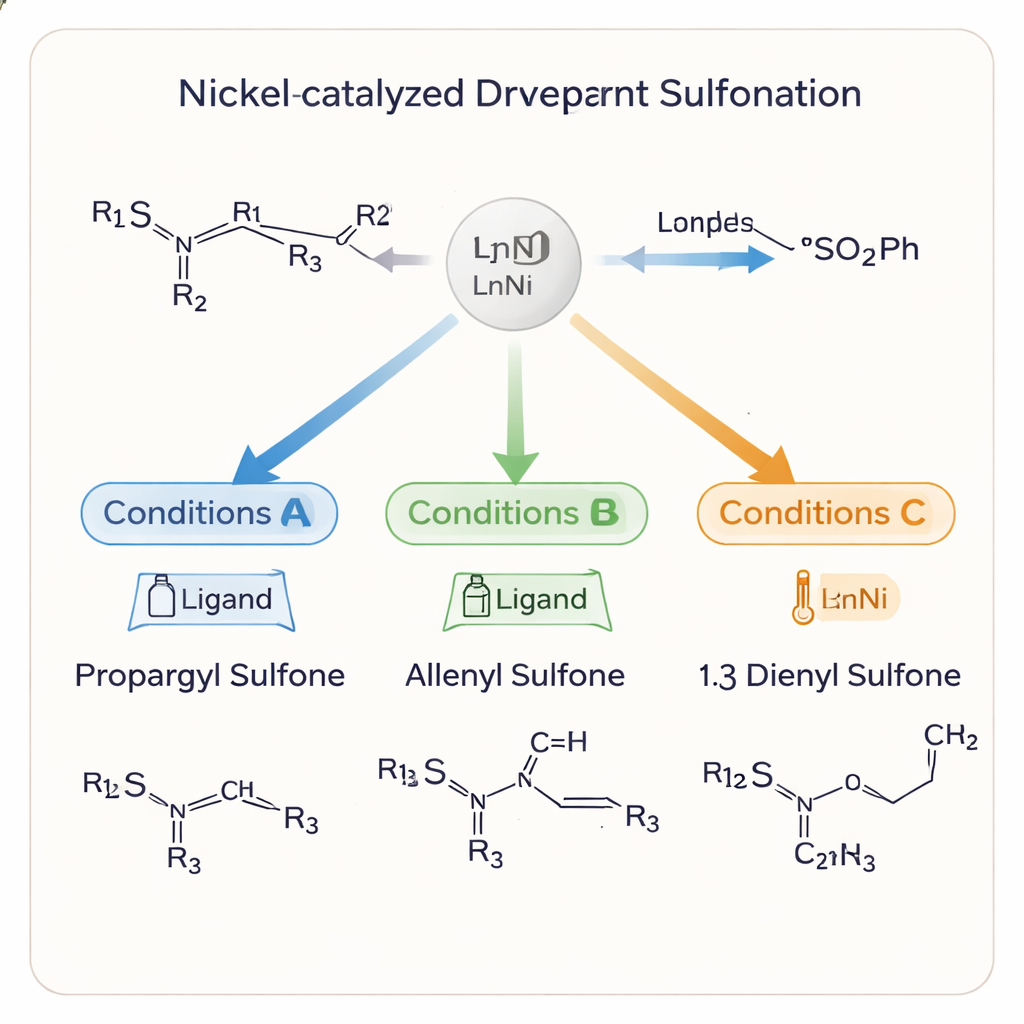
A striking feature of the study is that the same basic ingredients can be directed to form three distinct types of products: propargyl sulfones, allenyl sulfones, and 1,3‑dienyl sulfones. Each of these carbon frameworks leads to different downstream chemistry and biological activity. By carefully choosing the ligand bound to nickel, the solvent, temperature, and additives, the team can “steer” the reaction toward one outcome or another. Under mild conditions in acetonitrile and with a chiral phosphine ligand, they obtain propargyl sulfones in high yield and with excellent control over handedness. Treating these products with aluminum oxide gently reshapes the carbon–carbon triple bond into an allene, again without losing the chiral information. Switching to a different ligand and solvent instead channels the process into 1,3‑dienyl sulfones, extending the carbon framework.
Testing scope and flexibility
To see how general this approach is, the researchers varied both reaction partners. They showed that many different sodium sulfinates, including simple alkyl, aryl, and more complex ring systems, all participate cleanly and give chiral products with high enantiomeric excess. Likewise, a broad set of propargylic carbonates bearing different substitutions on the aromatic ring or carbon chain work well, although some substrates with very bulky groups or terminal alkynes fall outside the current method. The team also demonstrates that the quantities can be scaled up without sacrificing efficiency or selectivity, an essential step toward practical use. This broad scope means chemists can plug in many different fragments and rapidly access a library of related sulfur-containing molecules.
Turning building blocks into complex targets
Beyond simply making these sulfones, the authors showcase how easily they can be transformed into other useful structures. Hydrogenation converts the triple bond into alkanes or alkenes while preserving the chiral center next to sulfur. Simple follow-up reactions turn the sulfone units into sulfinates, sulfonamides, and sulfonyl fluorides—motifs that commonly appear in medicinal chemistry and in “click-like” bond‑forming reactions. As a highlight, they use their method as the key step in a concise synthesis of a chiral β‑sulfinylhydroxamic acid, a type of molecule known to inhibit bacterial enzymes. This route avoids several older, more cumbersome steps and underscores the synthetic power of having chiral sulfinates available on demand.
How the reaction chooses its path
The team also probes how and why the reaction switches between products. Time‑course experiments show that the propargyl sulfone usually forms first, then can rearrange into the allene and eventually into the 1,3‑diene under certain conditions. The nickel catalyst, together with the chosen ligand and additive, controls which intermediates are favored and how the sulfur group shifts along the carbon chain. Aluminum oxide, for example, allows the alkyne‑to‑allene reshaping to proceed at very low temperature while preserving the 3D arrangement of atoms. A simplified mechanism suggests that subtle changes in reaction conditions redirect a common nickel intermediate down different pathways, explaining how one system can yield multiple, precisely defined products.
What this means for future medicines and materials
For non-specialists, the key takeaway is that this nickel-catalyzed method provides a versatile chemical “switchboard”: from the same simple starting pieces, chemists can dial in which sulfur-rich skeleton they want and obtain it in a single, highly selective step. Because these products are excellent building blocks for drugs and other functional molecules, the approach could shorten the path from concept to candidate compound. It also demonstrates how careful catalyst and condition design can turn a once‑challenging transformation into a routine tool, opening the door to faster, cleaner construction of complex, chiral sulfur-containing molecules.
Citation: Gu, W., He, Z., Wang, H. et al. Nickel-catalyzed divergent sulfonations of propargylic carbonate. Nat Commun 17, 1882 (2026). https://doi.org/10.1038/s41467-026-68720-w
Keywords: nickel catalysis, chiral sulfones, asymmetric synthesis, organic reaction design, drug-like building blocks