Clear Sky Science · en
TREM2 expression level is critical for microglial state, metabolic capacity and efficacy of TREM2 agonism
Why brain cleanup cells matter in Alzheimer’s
Alzheimer’s disease is best known for sticky protein clumps in the brain, but an equally important story is how the brain’s own immune cells respond to this damage. These cells, called microglia, act as janitors and paramedics: they clear debris, manage inflammation, and help neurons survive. This study asks a deceptively simple question with big implications for future treatments: how much of a key microglial receptor, TREM2, is “just right” for protecting the brain—and when might drugs that boost TREM2 help or harm?
A dimmer switch, not an on–off button
Past genetics research showed that people with certain loss-of-function changes in the TREM2 gene face a higher risk of late-onset Alzheimer’s disease. Yet TREM2 was often treated as an on–off switch: either present or missing. The authors instead built a special “reporter” mouse in which microglia glow in proportion to how much TREM2 they make. This allowed them to sort microglia into low, medium, and high TREM2 groups and examine what each group was doing. They studied these cells both in healthy mice and in mice that develop amyloid plaques, a hallmark of Alzheimer’s-like pathology. 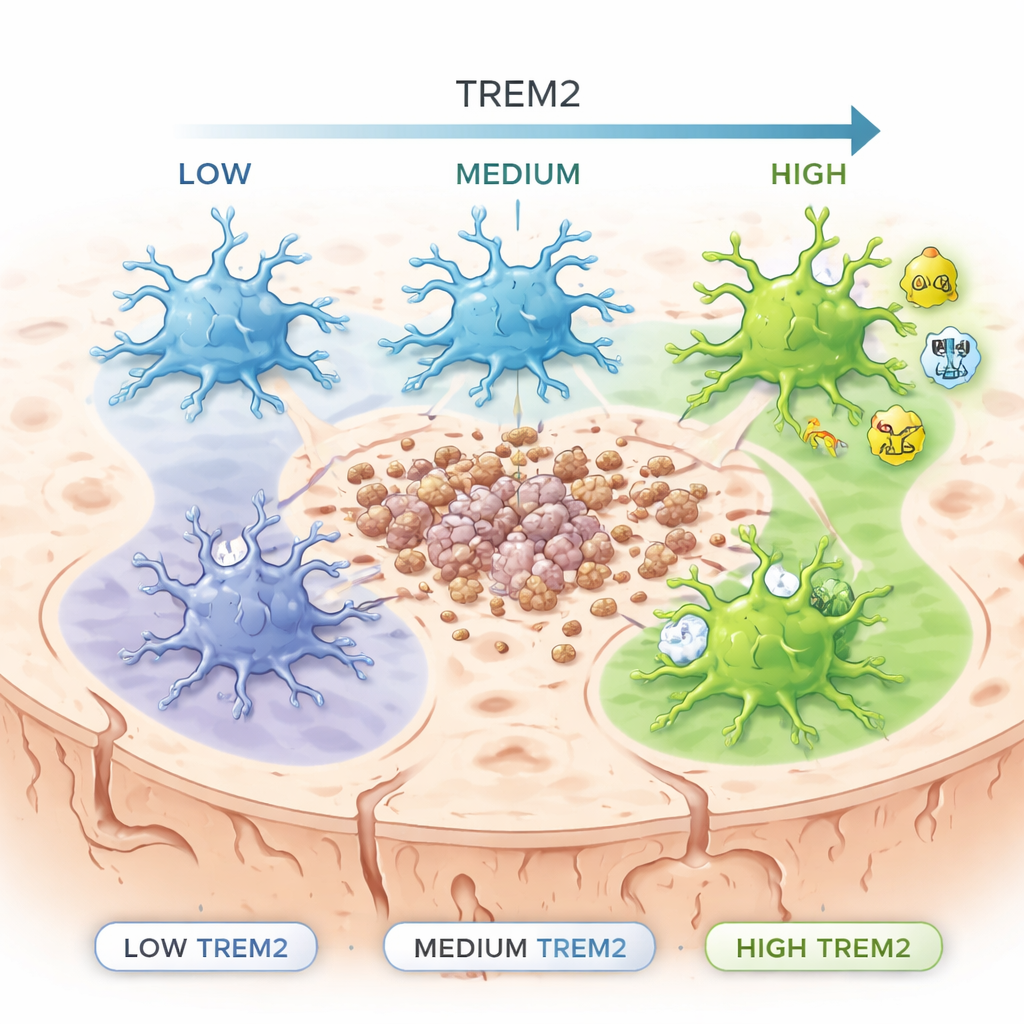
Energy use and cholesterol handling shift with TREM2 level
Once they could separate these microglial subgroups, the researchers analyzed which genes were turned on, which metabolites were present, and how actively the cells were using energy. Microglia with more TREM2 showed stronger activity in pathways linked to oxidative phosphorylation (the cell’s main energy-generating system) and to cholesterol and lipid metabolism. Measurements of labeled glucose uptake confirmed that cells with higher TREM2 drew in more sugar, especially in diseased brains. Detailed metabolite and lipid profiles revealed that high-TREM2 microglia had more of the building blocks for cellular energy and antioxidant defense, and less free cholesterol and certain fatty molecules, pointing to more efficient fuel use and better cholesterol disposal.
From metabolism to cleanup power
These molecular signatures translated into real functional differences. In controlled lab tests, the team exposed microglia to fluorescently labeled myelin fragments—a stand-in for the kinds of debris microglia must clear in diseased brain tissue. Microglia with low TREM2 took up the least debris; those with medium levels did better; and the high-TREM2 cells were the most voracious. The same pattern appeared in two different Alzheimer’s-like mouse models, underscoring that TREM2 level is tightly linked to the cell’s “cleanup capacity.” At the same time, gene network analyses showed that higher TREM2 was associated not only with improved metabolism but also with certain immune response programs, including interferon-related signals that may be beneficial in some contexts and harmful in others. 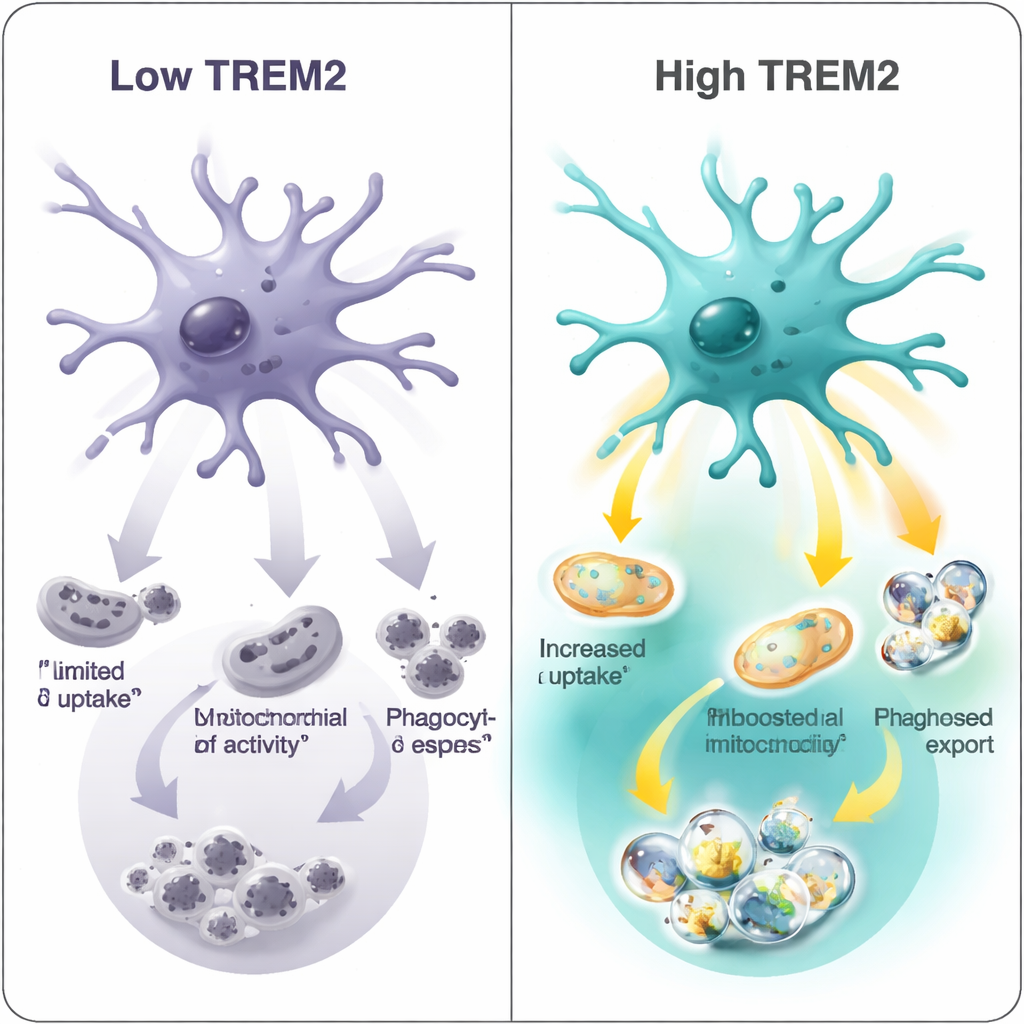
When boosting TREM2 helps—and when it may overshoot
Because pharmaceutical companies are testing antibodies that activate TREM2, the authors asked how such a drug affects microglia with different baseline TREM2 levels. They treated reporter mice carrying amyloid pathology with a TREM2-activating antibody that can cross the blood–brain barrier, then again sorted microglia by TREM2 expression. Strikingly, the cells with medium TREM2 responded most strongly: their metabolic markers showed boosted glycolysis, better lysosomal and peroxisomal activity, and increased levels of certain protective membrane lipids. Low-TREM2 microglia barely changed, likely because they did not provide enough target for the drug. In high-TREM2 microglia, however, the same treatment seemed to nudge metabolism back down toward a more moderate state, hinting at a “ceiling effect” where excessive activation is dampened rather than amplified.
What this means for future Alzheimer’s treatments
For a lay reader, the core takeaway is that microglia with the right amount of TREM2 are better fueled, better at managing cholesterol, and better at clearing harmful material around amyloid plaques. But the balance is delicate. Drugs that stimulate TREM2 will likely work best within a specific window of receptor levels—too little TREM2 and there is not enough to activate; too much and the system may already be near or past its optimal point. This helps explain why an early clinical trial of a TREM2-boosting antibody failed and suggests that future trials may need to measure TREM2 activity and microglial state over time, perhaps with brain imaging, to pick the right patients and treatment timing. In short, this work reframes TREM2 as a finely tunable “dimmer” for microglial metabolism and cleanup, rather than a simple on–off switch, and offers a roadmap for smarter, more targeted immunotherapies in Alzheimer’s disease.
Citation: Feiten, A.F., Dahm, K., Schlepckow, K. et al. TREM2 expression level is critical for microglial state, metabolic capacity and efficacy of TREM2 agonism. Nat Commun 17, 2002 (2026). https://doi.org/10.1038/s41467-026-68706-8
Keywords: microglia, TREM2, Alzheimer’s disease, brain metabolism, immunotherapy