Clear Sky Science · en
Cancer cachexia in STK11/LKB1-mutated non-small cell lung cancer is dependent on tumor-secreted GDF15
Why some lung cancers cause dangerous weight loss
Cancer does not always just grow silently in one spot. In many people, it slowly robs the body of fat, muscle, strength, and energy in a condition called wasting or cachexia. This study asks a pressing question for patients with a common form of lung cancer: why do some tumors trigger this severe weight loss while others do not? The researchers trace the problem to a stress signal made by certain tumors, and show that blocking this signal in animals can largely stop or even reverse the wasting.
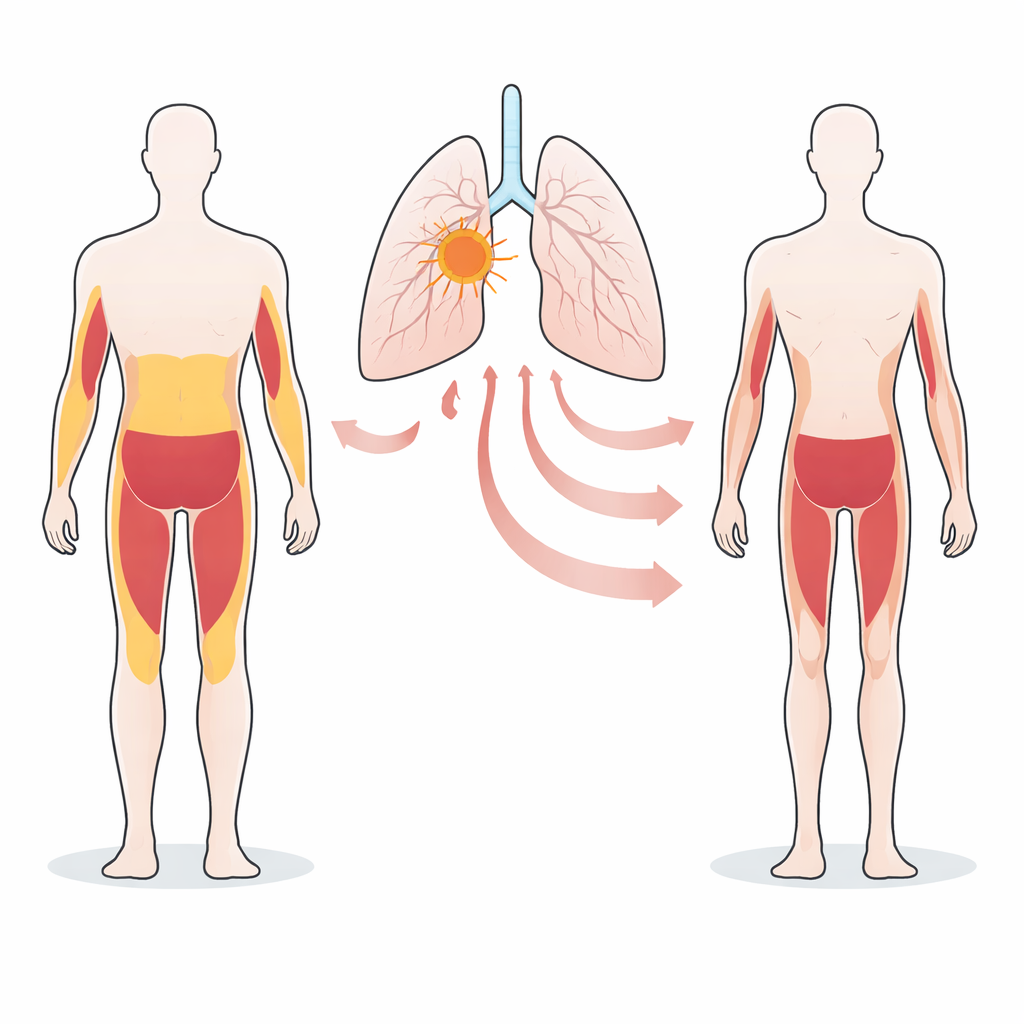
A hidden link between tumor genes and body wasting
The team focused on non-small cell lung cancers that carry faulty versions of a gene called STK11/LKB1. Earlier work showed that tumors with this defect often go hand in hand with major loss of weight, fat, and muscle in both mice and patients. Here, the scientists compared many human lung tumor lines that either did or did not cause wasting when grown in mice. They found that wasting tumors with STK11/LKB1 defects produced much higher amounts of a stress hormone-like protein called GDF15, and that this protein spilled into the bloodstream. In contrast, cells from the host animals contributed little to the GDF15 found in blood, pointing to the tumor itself as the main source.
Proving the tumor signal drives weight, fat, and muscle loss
To test whether tumor-made GDF15 actually causes wasting, the researchers used genetic tools to switch off GDF15 only in the cancer cells, leaving all other genes untouched. In several different STK11/LKB1‑mutant lung cancer lines, turning off GDF15 did not slow tumor growth, but it powerfully protected mice from losing body weight, fat mass, muscle mass, and grip strength. This protection appeared in both males and females and in models where tumors were placed under the skin or directly into the lung, suggesting the effect comes from factors released into the blood rather than from where the tumor sits. Even when tumors produced only modest amounts of GDF15, cutting off this signal still blunted wasting, showing that relatively low levels can be biologically important.
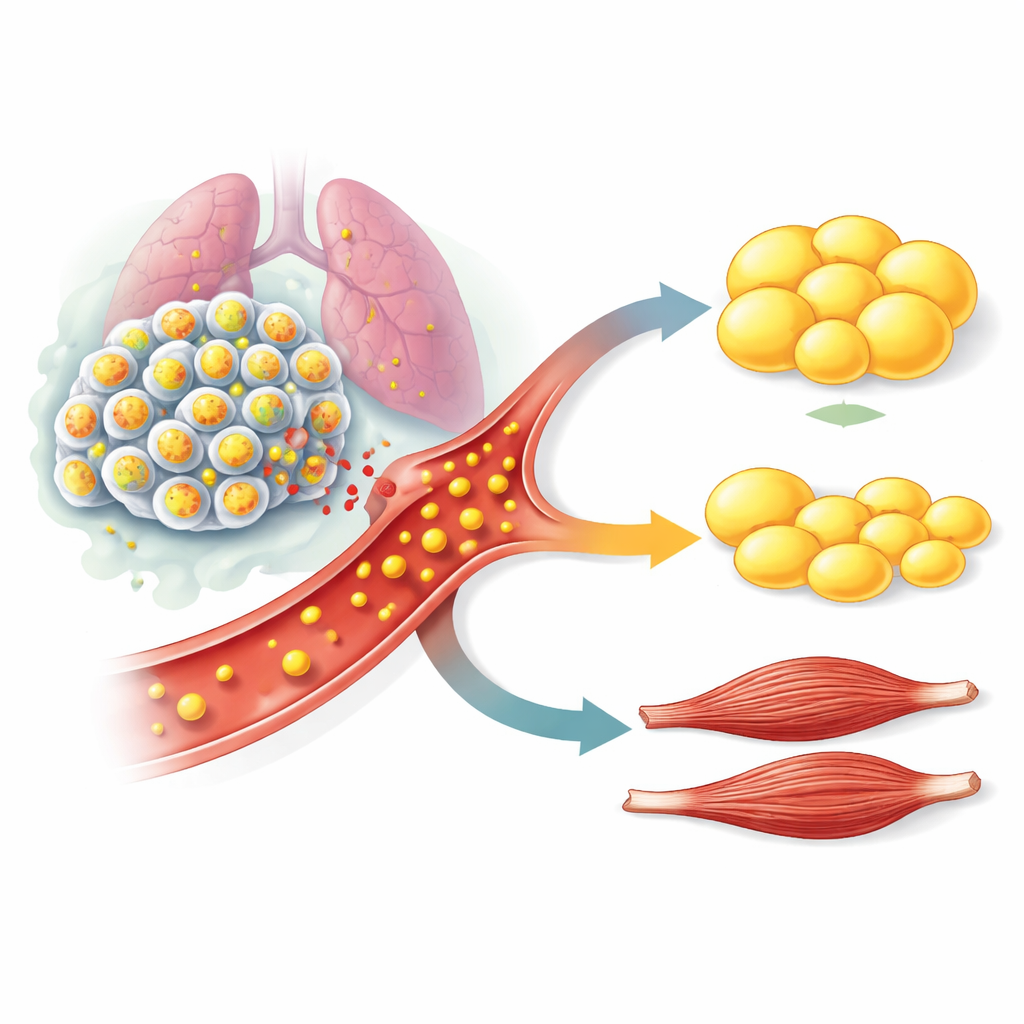
Antibodies that block the signal show treatment promise
Because drug companies are already developing antibodies that neutralize GDF15, the team asked whether such an approach could help in their models. They treated mice bearing STK11/LKB1‑mutant tumors with a GDF15‑blocking antibody similar to one now in clinical trials. When given early, the antibody prevented the usual loss of fat, muscle, and strength without affecting how fast tumors grew. When given later—after the animals had already lost a large share of their fat—the same antibody could stop further decline and partially restore body reserves and muscle weight. Similar results were seen in a mouse lung cancer model with an intact immune system, suggesting the findings are not limited to immune‑deficient animals.
How a stressed tumor turns on the wasting switch
The study also explores why STK11/LKB1‑mutant tumors make so much GDF15. Cells use an internal alarm system, called a stress response, to cope with low nutrients and other threats. In tumors lacking STK11/LKB1, this alarm is chronically active, boosting production and processing of GDF15 into its mature, secreted form. When the researchers re‑introduced a working STK11/LKB1 gene into a human lung cancer line, the stress response quieted down. Levels of GDF15 inside the tumor and in the blood dropped sharply, food intake improved, and the animals no longer developed cachexia, even though the tumors were still present.
What this means for patients with lung cancer
For people with non-small cell lung cancers that carry STK11/LKB1 mutations, this work suggests that the tumor itself can act like an endocrine organ, releasing GDF15 into the bloodstream and driving whole-body wasting. In mice, blocking GDF15—either by removing it from the tumor or by using an antibody in the circulation—greatly reduced fat and muscle loss without directly shrinking the tumor. That raises the possibility that, in the roughly 15 percent of lung cancer patients with STK11/LKB1 defects, anti‑GDF15 therapies could help preserve strength and body weight across a wide range of blood GDF15 levels. In other words, turning down this single tumor-made signal may one day offer a practical way to ease one of cancer’s most debilitating complications.
Citation: Yu, J., Guo, T., Gupta, A. et al. Cancer cachexia in STK11/LKB1-mutated non-small cell lung cancer is dependent on tumor-secreted GDF15. Nat Commun 17, 2182 (2026). https://doi.org/10.1038/s41467-026-68702-y
Keywords: cancer cachexia, non-small cell lung cancer, GDF15, STK11 LKB1 mutation, muscle and fat loss