Clear Sky Science · en
Meta-amplified dark-field interferometric scattering microscopy
Seeing the Smallest Building Blocks of Life
Many of the most important players in biology—such as proteins, viruses, and tiny membrane bubbles called exosomes—are far smaller than the wavelength of visible light. Watching these nanoscale actors in action, without attaching fluorescent tags that might disturb them, has been a long‑standing goal. This paper introduces a new kind of microscope that makes these nearly invisible particles stand out sharply against an almost perfectly dark background, opening the door to gentler, faster, and more sensitive measurements in biology and medicine.
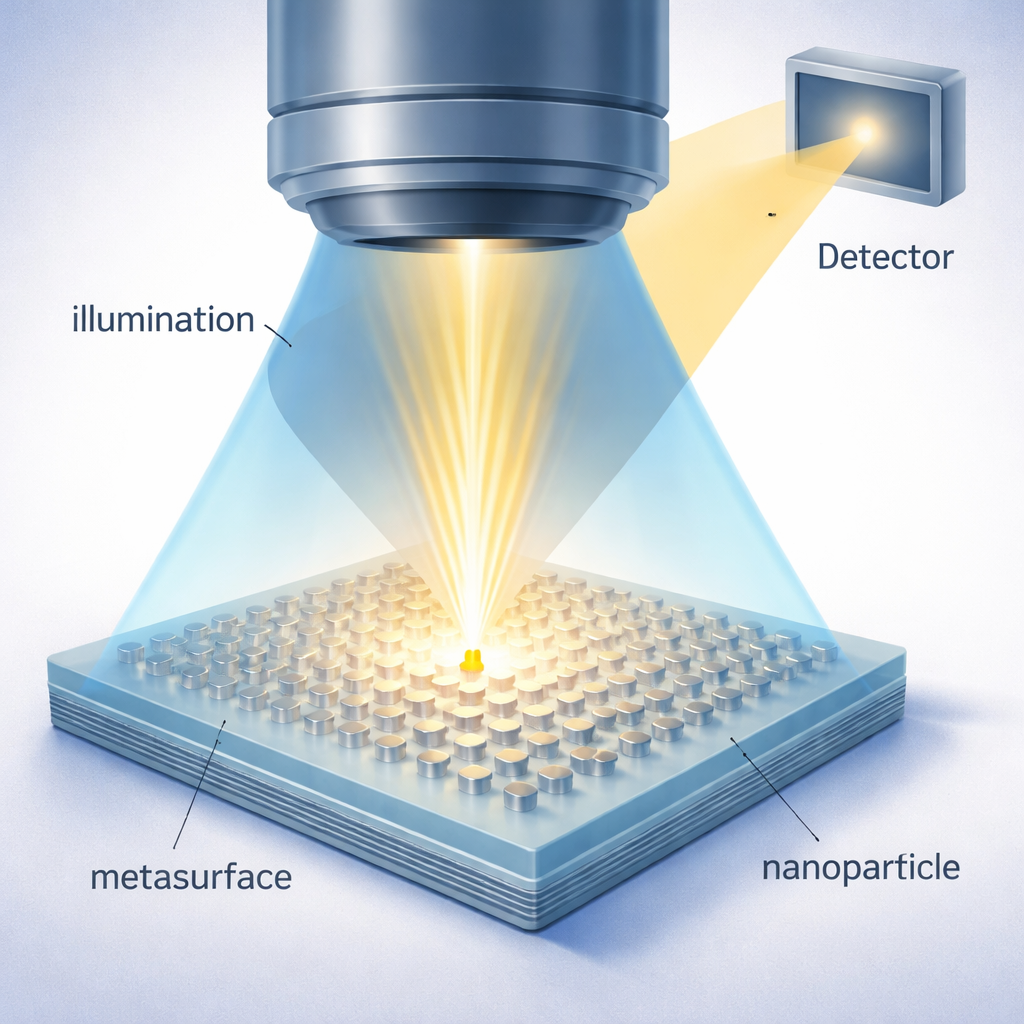
Why Tiny Particles Are So Hard to Spot
Ordinary light microscopes struggle with nanoscale objects because such small particles scatter only a vanishingly small amount of light—the scattering strength falls dramatically as the particle gets smaller. Interferometric scattering microscopy (iSCAT) partly solves this by recording the interference between a weak signal from the particle and a stronger, flat reference beam reflected from a surface. This boosts sensitivity enough to reveal single proteins and viruses. But there is a trade‑off: if you dim the reference beam to improve contrast, you also reduce the total number of photons, so the image becomes noisy. Pushing iSCAT to reliably detect ever smaller particles has therefore become increasingly difficult.
Turning a Flat Surface into an Active Light Antenna
The authors tackle this problem by replacing the usual flat glass slide with a carefully engineered “metasurface” made of dense, hexagonally arranged silver nano‑pillars, each only tens of nanometers in size. These tiny metal structures behave collectively like an antenna array for light. In their normal state they are designed to cancel one another’s scattering into the microscope’s collection cone, creating a very dark background—this is called the dark mode. However, when a nanoparticle comes close to the metasurface, it disturbs the local electromagnetic balance. That disturbance shifts the phases and strengths of the nano‑pillars’ oscillations so that they now radiate strongly into the detector, switching locally into a bright mode centered around the particle.

Amplifying Signals from Nanoparticles and Biomolecules
This new technique, called meta‑amplified dark‑field interferometric scattering microscopy (MAD‑iSCAT), effectively uses the metasurface as an active amplifier of the particle’s presence. Instead of relying mainly on the particle’s own weak scattering, MAD‑iSCAT measures how the particle reshapes the much stronger light waves produced by the metasurface. Because those waves are intense and highly sensitive to tiny environmental changes, even very small particles can trigger a detectable bright patch in the image. Simulations and experiments show that the signal grows far more gently with particle size than in conventional “Rayleigh” scattering, meaning that the method remains effective down to very small diameters where traditional approaches would fail.
Putting the New Microscope to the Test
To prove that MAD‑iSCAT works in practice, the researchers fabricated their silver metasurfaces using nanoimprint techniques and coated them with a thin protective polymer layer. They then imaged polystyrene spheres with diameters between 45 and 200 nanometers and compared the brightness to the same particles on a plain polymer film. The metasurface boosted the apparent scattering intensity by more than one to two orders of magnitude, depending on size and color of light. In watery environments, where many biological samples live, the team directly compared MAD‑iSCAT with a state‑of‑the‑art iSCAT setup. For particles just a few tens of nanometers across, MAD‑iSCAT delivered tens of times higher image contrast, and did so using only two frames instead of hundreds, indicating much higher throughput.
Watching Real Biological Nanoparticles
Beyond plastic test beads, the authors demonstrated that MAD‑iSCAT can visualize single exosomes released by breast cancer cells and even individual ferritin protein complexes. By tracking the motion of exosomes in solution, they estimated their sizes and found that MAD‑iSCAT provided signal levels 10 to 100 times stronger than expected from simple scattering. For ferritin, a large protein complex of about 440 kilodaltons, they observed clear spots with significantly improved signal‑to‑noise ratio compared with standard interferometric approaches. These results show that the new method can reach down to the scale of single biomolecules while still working in realistic liquid environments.
What This Means for Future Biosensing
In everyday terms, MAD‑iSCAT converts an otherwise ordinary microscope slide into a smart surface that lights up only when a nanoscale object touches it. By combining a nearly black background with strongly amplified signals around each particle, the technique makes it far easier to detect and measure tiny biological structures without labels. While the current devices still face challenges in fabrication precision and field‑of‑view, the concept promises faster, more sensitive tools for weighing individual molecules, monitoring disease‑related vesicles like exosomes, and potentially pushing label‑free optical imaging into the realm of super‑resolution.
Citation: Lee, H., Zhao, J., Hu, P. et al. Meta-amplified dark-field interferometric scattering microscopy. Nat Commun 17, 1977 (2026). https://doi.org/10.1038/s41467-026-68697-6
Keywords: label-free microscopy, nanoparticle detection, plasmonic metasurfaces, biosensing, interferometric scattering